Chemistry Lab 2007 – Agenda:
advertisement

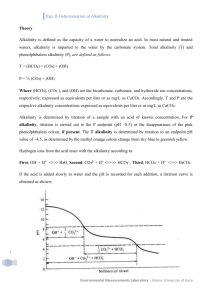
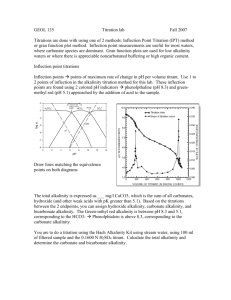
Chemistry Lab 2007 – Agenda: Possible known waters: salt water, DI water, sugar water, water with baking soda (NaHCO3 – sodium bicarbonate) Possible unknown waters: well water, river water, rain water, lake water Before lab: -change the handout to the actual waters that I will be using -get out all materials: -handouts -glassware -gloves and goggles -titrators, ph and conductivity meters -clean all glassware -calibrate all probes -practice all experiments **keep an example of salmon color for titration experiment for students to compare to -draw carbonate/pH diagram on board -draw boxes for answers on board, get averages for the groups before trying out the unknown samples Purpose: -to learn how to measure and interpret pH, conductivity, and alkalinity -illustrate the concept that different waters have different characteristics Background Information and Methods: pH: -the greater the pH, the fewer H+ ions and thus the less acidic -remember, 0 = strong acid, 14 = strong base -lakes and streams vary between 6-9, although exceptions exist (bogs are acidic wetlands) -demonstrate the pH meter and how to use it Meters: -do not submerge below line -allow to stabilize before taking a reading Conductivity: -ability of water to pass an electric current -related to the total amount of dissolved ions in solution (if lots of dissolved ions, the conductivity will be high) -demonstrate 2 kinds of meters that will be used and how they will be used WARNING: do not immerse probe all the way, not water proof NOTE: 1000 microsiemens = 1 millisiemens, make sure to know what you are recording, units are important in this lab Alkalinity: WRITE ON BOARD: CO3 + 2H+HCO3 + H+ CO2 +H20 -also known as ANC (acid neutralizing capacity or a buffering capability, ability of the water to neutralize a strong acid, if extra H+ is put into solution, the CO3 sucks it up and turns into HCO3 so pH would be lowered) (EXPLAIN USING DIAGRAM) -a lake or stream high in alkalinity would resist a reduction in pH -a stream low in alkalinity would drop in pH upon acid addition because it wouldn’t have the ability to resist the change (EXAMPLE: Adirondack mountains, low in alkalinity because of geology and the pH was quickly reduced from the acid rain, now very few fish live in the lakes there because they are so acidic) -major contributor to alkalinity in Michigan is CaCO3 (calcium carbonate) from limestone deposits and MgCO3 (magnesium carbonate) from dolomite -to determine, do a titration experiment -explain how to use titrator Titrator: -get rid of bubbles -set to 0000 -rinse tip with DI water to make sure none extra being added to solution When acid is added, pH is lowered and it will hit these points: 1st Endpoint = pH 8.3: solution will turn pink when phenolphalein indicator is added if the pH is above 8.3, determine how much acid is necessary to bring the pH down to 8.3 (carbonate converted to bicarbonate and pink solution goes clear) 2nd Endpoint = pH 4.5: if pH is less than 8.3 and nothing happens when phenolphalein is added, add bromcresol-green methol-red indicator packet, determine how much acid is necessary to bring the pH down to 4.5 (all bicarbonate converted to CO2, blue/green solution grey/purple pink/salmon color) *at this point the water has no buffering capacity left The amount of acid required to reach the phenolphalein alkalinity + the amount to reach the bromcresol-green methol-red alkalinity is the total alkalinity and represents the total buffering capacity -recorded as mg/L CaCO3 or ppm CaCO3 The Experiment: Safety: -wear gloves and goggles when handling H2SO4 (sulfuric acid when doing alkalinity) Rinse: -DI rinse all meters and titrator before using it on the next sample -shake gently to dry -do not dry with paper towel -rinse glass with DI between samples -acid wash the flasks before titrating next sample Procedure: -using 4 knowns and 4 unknowns -record the pH, conductivity, and alkalinity of all knowns on lab handout and on the board (we will average the known’s numbers just to see a trend but you should use what you got as a group) -obtain 4 unknowns and record pH, conductivity, and alkalinity -try to identify samples based on the measurements you get Assignment: -ask if any questions? -typed questions due at the beginning of lab next week (individual, not group assignment) *Clean glassware and wipe down tables before leaving