haemostasis new 1
advertisement

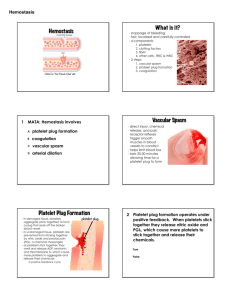
The micrograph shows activated platelets adhering to some damaged cells Hemostasis Content • • • • • Defination Normal vessel wall Stages of haemostasis Anti clotting mechanism Applied Defination • Term haemostasis means prevention of blood flow. • It is a process of forming clots in the walls of damaged blood vessels and preventing blood loss while maintaining blood in a fluid state within the vascular system. HEMOSTASIS • Normal hemostasis is a consequence of tightly regulated process that maintain blood in a fluid state in normal vessels, yet also permit the rapid formation of a hemostasis clot at the site of a vascular injury. HEMOSTASIS The process of hemostasis can be divided into two distinct stages namely: 1. Primary hemostasis – platelet plug formation 2. Secondary hemostasis – coagulation cascade • The goal of both primary and secondary hemostasis is to arrest bleeding from damaged blood vessels (hemo = blood, stasis = standing) • Is counter-balanced by reactions, which prevent blood coagulation in uninjured vessels and maintain the blood in a fluid state – Balance between procoagulants and anticoagulants • 4 overlapping processes or stages – Local vasoconstriction – Formation of a platelet plug – Formation of a web of fibrin proteins that penetrate and surround the platelet plug – blood coagulation or clotting – Clot retraction. 6 LOCAL VASOCONSTRICTION • Results from – Release of vasoconstrictor substances (paracrine & autocrine agents) from • Platelets (i.e., serotonin & thromboxane A2) • Traumatized tissue – Local myogenic spasm initiated by direct tissue damage – Reflex vasoconstriction initiated by activation of nociceptors and other sensory endings • Effects – ↓ blood flow and Pressure in the damaged area Last for many minutes or even hours, during this time the ensuing processes of platelet plugging and blood coagulation can take place 7 FORMATION OF A PLATELET PLUG (temporary hemostatic plug, white plug) Factors that prevent/limit formation of a plug 1. Prostacyclin (prostaglandin I2). Inhibits platelet aggregation; vasodilator 2. Nitric oxide* (NO). Inhibits platelet adhesion, activation and aggregation and stimulates local vasodilation Damaged blood vessel wall Intact blood vessel wall Collagen fibers are exposed to the blood and coated with WF* Secretion of prostacyclin & nitric oxide - + stimulation - inhibition * Von Willebrand factor, a protein synthesized by endothelial cells and megakaryocytes, enhances platelet adherence by forming a bridge between cell surface receptors and collagen in the subendothelial matrix. Adhesion of the platelets Platelet release reaction & activation Platelet aggregation & plug retraction Temporary hemostatic (platelet) plug + + Local vasoconstriction FORMATION OF A PLATELET PLUG (cont.) Stage 1. Platelets adhesion A. vWF - von Willebrand factor (soluble plasma protein) binds to collagen of subendothelial matrix Failure of this step may be due to: - Absence of von Willebrand factor - Malfunction of collagen - Scurvy B. vWF exposes multiple intrinsic binding sites for the platelet specific membrane glycoprotein Ib (GPIb) vWF binds to glucoprotein Ib receptors of platelets and to collagen 9 FORMATION OF A PLATELET PLUG (cont.) Stage 2-3 Platelets release reaction and activation. Binding of the platelets to the collagen → Release of agents from secretory granules (degranulation) – serotonin, adrenaline, several clotting factors, thromboxane A2, tissue factor and ADP Serotonin, adrenaline and ADP act locally → changes in the metabolism, shape, and surface proteins of the platelets. Serotonin and thromboxane A2 stimulate local vasoconstriction 10 Primary Hemostatsis Step 1: Transient vasoconstriction (endothelin) Step 2: Platelet adhesion - von willebrand factor bind to the disrupted blood vessel via GpIb Step 3: Platelet release ADP and thromboxane A2 which stimulate adhesion of the next layers of platelets (recruitment) through a positive feedback mechanism and formation of a platelet plug inside the vessel ADP induces platelet to express GpIIB-IIIa which is needed to platelets aggregation via fibrinogen Step 4: Platelet aggregation (Platelt plug) Deficiency: Bernard-Soulier syndrome Deficiency: Glanzmann thombasthenia GpIb Platelet GpIIb-IIIa complex GpIb Endothelium Von willebrand factor Subendothelium FORMATION OF A PLATELET PLUG (cont.) Stage 4: Recruitment and loose platelets aggregation Platelet • ADP and thromboxane A2 stimulate adhesion of the next layers of platelets (recruitment) through a positive feedback mechanism and formation of a platelet plug inside the vessel GpIIb-IIIa complex Failure of this step: - Insufficient number of platelets - Dysfunctional platelets (prior activation occurs during cardiopulmonary bypass, storage, exposure to aspirin, uraemia and acute and chronic alcohol exposure) 12 12 Normal Blood Vessel Injured blood vessel Attracts more platelets Aggregate into platelet plug Release of Platelet factors Exposed collagen binds and activates platelets Secondary Hemostasis This involve the conversion of the fibrinogen (solube) in the platelet plug to fibrin (insoluble). Fibrin is then cross-linked to yield a stable platelet-fibrin thrombus. Secondary hemostasis involve the activation of coagulation cascade factors in both intrinsic and extrinsic pathways BLOOD COAGULATION (CLOTTING) • Is the transformation of the blood into a solid gel (a clot or thrombus) • Occurs locally around the platelet plug; supports and reinforces the plug • Requires 12 plasma clotting factors and platelets • Involves a cascade of biochemical reactions in which each factor that is activated in turn activates the next factor • The fundamental reaction is conversion a soluble protein, fibrinogen to an insoluble protein, fibrin In coagulation a series of plasma proteins called blood-clotting factors play major roles. Most of these are inactive forms of proteolytic enzymes. When converted to the active forms, their enzymatic actions cause the successive, cascading reactions of the clotting process. 17 Coagulation System Consists of a cascading system of proteins – Primarily originating from liver (except factor III) – Circulate in inactive form – System includes: • • • • • Enzymatic factors Non-enzymatic factors Tissue thromboplastin (factor III) Calcium (factor IV) Platelet phospholipid (PF 3) - structural component; accelerates factor activation • Anticoagulant factors The coagulation system consists of three pathways (intrinsic, extrinsic and common) PLASMA CLOTTING FACTORS Scientific Name Common Name Main Function Factor I Fibrinogen Converted to fibrin Factor II Prothrombin Enzyme Factor III Tissue thromboplasm Cofactor Factor IV Calcium Cofactor Factor V Proaccelerin Cofactor Factor VII Proconvertin Enzyme Factor VIII Antihemophilic factor Cofactor Factor IX Christmas factor Enzyme Factor X Stuart factor Enzyme Factor XI Plasma thromboplatin antecedent Enzyme Factor XII Hageman factor Enzyme Factor XIII Fibrin stabilizing factor Enzyme 3 PHASES OF BLOOD COAGULATION • Formation of a complex of activated substances prothrombinase (prothrombin activator) • Formation of active thrombin from prothrombin – Is catalyzed by prothrombin activator • Formation of insoluble fibrin from soluble fibrinogen – Is catalyzed by thrombin 20 PHASE 1 – FORMATION OF PROTHROMBINASE INTRINSIC PATHWAY XII EXTRINSIC PATHWAY XIIa XIa XI IX Ca2+ III Ca2+ VIIa VII IXa PF-3 Ca2+ VIII Xa PF-3 Ca2+ V X II X XIII Thrombin XIIIa Fibrinogen Fibrin Stable fibrin polymer PHASE 3 – FORMATION OF FIBRIN • Thrombin catalyses release of 2 pairs of polypeptides from each fibrinogen molecule and formation of fibrin monomers – Ca++ and platelet factors are also required • Monomers join together to form insoluble fibrin polymers – a loose mesh of stands • Stabilization of fibrin – formation of covalent cross-bridges, which is catalyzed by factor XIII (+ Ca++) 22 FINAL EVENTS OF HEMOSTASIS • Fibrin forms a meshwork, which supports the platelet plug • Clot occludes the damaged blood vessel and ↓ or stops bleeding • Retraction of the clot due to contraction of fibrin fibers and contractile proteins of the platelets – ↑ clot density – Occlusion of the damaged vessel – Bringing the edges of wound together → facilitation of wound heeling • Fate of the blood clot – Invasion by fibroblasts → formation of connective tissue through the clot – Fibrinolysis and destruction of the clot 23 Dissolving the Clot Figure 16-14: Coagulation and fibrinolysis Clot Dissolution • Plasmin (fibrin-digesting enzyme) is made from activating plasminogen (blood protein) – Presence of the clot causes endothelial cells to release tissue plasminogen activator – Fibrinolysis begins within 2 days and continues slowly over several days until the clot is dissolved. ra Plasminogen Activator (e.g. t-PA) Fibrin Plasmin soluble fragments Fibrinolysis Clinical applicationHuman t-PA is produced by recombinant DNA technology and available for clinical use. lyses clots in the coronary arteries if given to patients soon after the onset of myocardial infarction. Streptokinase (from bacteria-streptococcci) and urokinase are also fibrinolytic enzymes used in the treatment of early myocardial infarction 26 Clot retraction & repair • Clot retraction occurs within 30-60 minutes. • Platelets contain actin & myosin • As clot is compacted fibroblasts (stimulated by plateletderived growth factor -PDGF) rebuild the wall while endothelial cells (stimulated by vascular endothelial growth factor -VEGF) multiply to restore the lining Overview of Hemostasis and Tissue Repair Damage to wall of blood vessel Collagen exposed Tissue factor exposed Platelets adhere and release platelet factors Vasoconstriction Coagulation cascade Thrombin formation Platelets aggregate into loose platelet plug Reinforced platelet plug (clot) Temporary hemostasis Fibrin slowly dissolved by plasmin Cell growth and tissue repair Clot dissolves Intact blood vessel wall Converts fibrinogen to fibrin ROLE OF VITAMIN K IN CLOTTING • Vitamin K acts as a cofactor of the enzyme γ-glutamyl carboxylase • Is required for γ carboxylation in the liver of – Prothrombin and factors VII, IX and X – Proteins S and C (natural anticoagulants) Vit K is activated by epoxide reductase in the liver • • γ carboxylation (introduction of a carboxylic acid group) of certain glutamate residues in target clotting factors → binding sites for Ca++ and PF3 most of clotting factors are synthesized by the liver. Therefore, liver diseases (i.e., hepatitis, cirrhoses, atrophy) depress the clotting system. Decreased dietary intake of vit K has limited consequences on blood clotting because Vit K is continuously synthesized by the intestinal flora. Note that Vit K is fat soluble and requires fats for absorption. Lack of the bile decreases fat digestion and absorption. 30 ROLE OF Ca++ IN COAGULATION • Ca++ is required for all steps of coagulation (except first 2 steps of the intrinsic pathway) • ↓ in the plasma [Ca++] below the threshold level for clotting → ↓ blood clotting by both pathways 31 ROLE OF THE PLATELETS IN COAGULATION Activated platelets • Display specific plasma membrane receptors that bind several of the clotting factors → several cascade reactions take place on the surface of activated platelets • Display phospholipids (platelet factors), which act as cofactors of the bound clotting factors 32 ROLE OF THE LIVER IN BLOOD COAGULATION • Synthesis of the plasma clotting factors • Synthesis of the bile salts, which are required for intestinal absorption of lipid soluble vitamin K 33 Anti clotting mechanism Anti clotting mechanism 1. 2. 3. 4. 5. 6. 7. 8. Smoothness of endothelium (glycocalyx) Thrombomodulin Fibrin fibers and anti thrombin III Heparin Plasmin Prostacyclin 2 macroglobulin Circulating blood • Antithrombin III – inhibits factor X and thrombin • Fibrin acts as an anticoagulant by binding thrombin and preventing its: • Heparin – a natural anticoagulant,potentiates effects of antithrombin III (together they inhibit IX, X, XI, XII and thrombin • Antithromboplastin (inhibits „tissue factors” – tissue thromboplastins) • Protein C and S – degrade factor Va and VIIIa NATURAL ANTICOAGULANTS (cont.) Thrombin/thrombo-modulin/protein C pathway Endothelial cell Thrombomodulin is a thrombin-binding endothelial cell receptor Thrombomodulin Thrombin Binds thrombin and inactivates it Protein C Complex of thrombin+thrombo-modulin binds protein C and activates it Activated Prot C Protein S Protein C in collaboration with protein S inactivates factors Va and VIIIa and activates plasminogen and fibrinolysis ViIIa Note: Mutated factor V cannot be inactivated (switched off) by activated protein C, and this will lead to hypercoagulable state VIII Inactivation of inhibitors of plasminogen activator Plasminogen Thrombin Va V Plasmin Fibrinolysis 37 DRUGS THAT INHIBIT BLOOD CLOTTING (ANTICOAGULANTS) • Heparin: Heparin binds to the enzyme inhibitor antithrombin III (AT), causing a conformational change that results in its activation. The activated AT then inactivates thrombin and other proteases involved in blood clotting such as XIIa, XIa, Xa and IXa • Coumarin derivatives (i.e., warfarin) – Block stimulatory effects of vitamin K on synthesis of clotting factors II, VII, IX, and X by the liver (inhibit epoxide reductase which activates vit K in the liver: K → K1) • Aspirin – Low doses inhibit prostaglandins and thromboxanes synthesis by the platelets → inhibition of platelet release reaction and platelet aggregation – Is effective in preventing of heart attack and reduction of the incidence of sudden death 38 IN VITRO INHIBITION OF BLOOD CLOTTING • Keeping of blood in seliconized containers – prevention of contact activation of platelets and factor XII • Substances that bind ionized calcium to produce un-ionized calcium compound or to form insoluble salts with calcium – Sodium citrate or oxalate – Ammonium or potassium citrate – EDTA (ethylenediaminetetraacetic acid) • Is ability to "sequester" di- and tricationic ions (Ca2+ & Fe3+) • Is widely used as an anticoagulant for blood samples for complete blood count/full blood examination • Heparin 39 Bleeding time This is a test that measures the speed in which small blood vessels close off (the condition of the blood vessels and platelet function) This test is useful for detecting bleeding tendencies The bleeding stops within 1 to 4 minutes. This may vary from lab to lab, depending on how the test is measured The time taken for blood to clot mainly reflects the time required for the generation of thrombin The surface of the glass tube initiates the clotting process. This test is sensitive to the factors involved in the intrinsic pathway The expected range for clotting time is 4-10 mins. Whole blood clotting time Tests of coagulation "Intrinsic" and "extrinsic" coagulation pathways Activated Partial Thromboplastin Time N: 25-35 sec Prothrombin Time N: 9.9 – 13 sec PROTHROMBIN TIME (protime, PT test) • Measures the clotting time of plasma from the activation of factor VII, through the formation of fibrin clot • Assesses the integrity of the extrinsic/tissue factor pathway and common pathways of coagulation (factors VII, X, V, II, I) • The PT test is widely used to monitor patients taking anticoagulants as well as to help diagnose clotting disorders 44 Prothrombin Time Factors Thromboplastin and Calcium Patient’s Plasma Diagnostica Stago training, 2005 I II V VII X 45 Prothrombin time (PT) test – norm 11 -15 sec evaluates extrinsic system (VII, X, V, II, fibrinogen) • prolonged PT indicates a deficiency in any of factors VII, X, V, prothrombin (factor II), or fibrinogen (factor I). • Prolonged PT: - a vitamin K deficiency (vitamin K is a co-factor in the synthesis of functional factors II (prothrombin), VII, IX and X) - liver disease - Warfarin therapy - DIC - excesive heparin PROTHROMBIN TIME (cont.) • Depends on [prothrombin] in the blood • Normal range 12 – 14 sec • Increased – ↓ prothrombin (less than 10% of normal) – Deficiency of fibrinogen or factors V, VII, or X – Therapeutic anticoagulants (i.e., heparin, warfarin, aspirin), some drugs (i.e., antibiotics, anabolic steroids, estrogens, etc.) – Liver diseases – Vit K deficiency – Disseminated intravascular coagulation • Decreased – Vit K supplementation – Thrombophlebitis 47 ACTIVATED PARTIAL THROMBOPLASTIN TIME (aPTT) • Assesses the integrity of the intrinsic and common pathways of coagulation • Measures the clotting time of plasma, from the activation of factor XII by a reagent through the formation of fibrin clot • Normal range 25 – 38 sec • Prolonged time – Use of heparin – Antiphospholipids antibodies – Coagulation factors deficiency (intrinsic and common pathways; i.e., hemophilias) 48 Activated Partial Thromboplastin Time Factors Ca++ Phospholipid and Activator Patient’s Plasma Diagnostica Stago training, 2005 I II V VIII IX X XI XII 49 Activated Partial Thromboplastin Time test (aPTT) – norm: 25-35 s; evaluates intrinsic system (VIII, IX, XI, XII, X, V, II, fibrinogen) • an isolated prolongation of the aPTT (PT normal) suggests deficiency of factor VIII, IX, XI or XII • prolongation of both the APTT and PT suggests factor X, V, II or I (fibrinogen) deficiency, all of which are rare • aPTT is normal in factor VII deficiency (PT prolonged) and factor XIII deficiency Most common case of prolonged aPTT – heparin!!! Thrombin Clotting Time Low concentration of thrombin Screens for effects of • Heparin • FDPs Undiluted patient’s plasma Diagnostica Stago training, 2005 52 Thrombin time (TT) – norm: 14-15 sec Prolonged TT: • Heparin (much more sensitive to heparin than aPTT) • Hypofibrinogenemia Selected causes of abnormal coagulation tests Partial Thromboplastin Time (aPTT) Prothrombin Time (PT) Thrombin Time (TT) Bleeding Time (BT) Factor deficiency (except VII) VII, X, V, II, fibrinogen deficiency Low or absent fibrinogen Thrombocytopenia Antibodies to clotting factors Antibodies Dysfibrinogenemia, Von Willebrand’s disease Heparin Warfarin; Vit K defficiency (mild to severe) Heparin Drugs (Aspirin, NSAIDs, high dose penicillins, etc.) Excessive Warfarin Excessive Heparin hypofibrinogenemia Cirrhosis, Uremia, PLTs dysfunction 2 TYPES OF ABNORMALITIES OF HEMOSTASIS • Excessive bleeding (hemorrhagic disease) caused by deficiency of a clotting factor/s or platelets • Excessive clotting: thrombosis, embolism, disseminated intravascular coagulation 56 CONDITIONS THAT CAUSE EXCESSIVE BLEEDING • Vitamin K deficiency • Deficiency of clotting factors (i.e., hemophilia) • Deficiency of thrombocytes – thrombocytopenia • Deficiency of von Willebrand factor 57 VITAMIN K DEFICIENCY • Results from – ↓ intestinal absorption of fats due to ↓ bile secretion (i.e., liver disease or obstruction of the bile ducts) – ↓ dietary intake of vit K (limited importance) • Results in – ↓ hepatic gamma carboxylation of • Prothrombin (II) • Factors VII, IX and X • Protein C and S – Bleeding tendency • Prolonged prothrombin time and partial thromboplastin time • Normal platelets count and serum fibrinogen split products 58 HEMOPHILIA • Is a hemorrhagic disease that results from deficiency of – Factor VIII (the smaller component) - hemophilia A or classical – Factor IX – hemophilia B, Christmas disease – Factor XI – hemophilia C • Is a genetic disease – Hemophilia A and B are sex linked (X chromosome) • Occur in males • Females are hemophilia carriers • Results in ↑ aPTT (PT, thrombocytes count, fibrin split products are normal) 59 Hemophilia A (lack of F VIII; 85%) • Spontaneous or traumatic subcutaneous bleeding • Blood in the urine • Bleeding in the mouth, lips, tongue • Bleeding to the joints, CNS, gastrointestinal tract Mild hemophilia after injection in buttock HEMOPHILIA Deficienc Clinical Syndrome y Factor Cause Factor I Afibrinogenemia Depletion during pregnancy with premature separation of placenta: Congenital Factor II Hypoprothrombinemia (Hemorrhagic tendency in liver diseases Decreased hepatic synthesis (secondary to vitamin K deficiency) Factor V Parahemophila Congenital Factor VII Hypoconvertinemia Congenital Factor VIII Hemophilia A (classical hemophilia) Congenital recessive sex-linked defect due to abnormalities of the gene that codes for factor VIII (X chromosome) Factor IX Hemophilia B (Christmas disease) Congenital recessive trait carried on X chromosome Factor X Stuart-Prower factor deficiency Congenital Hemophilia C (PTA deficiency Congenital Hageman trait Congenital THROMBOCYTOPENIA • Low thrombocytes count (below 50 000/m l) → poor plug formation, deficient clot retraction, deficient platelet phospholipids, poor constriction of ruptured vessels → bleeding tendency from many small venules and capillaries • Multiple hemorrhages in the skin and mucous membranes – thrombocytopenic purpura – Petechiae – small punctate hemorrhages(1-3 mm) – Echymoses - large hemorrhages (bruises) • Other causes of purpura – ↓ plasma level of 1 or more clotting factors – ↑ fragility of capillary walls (congenital, Vit C deficiency, adrenal failure, toxins, drugs, allergic reactions) 62 Severe reduction in the number of PLTs thrombocytopenia this causes spontaneous bleeding as a reaction to minor trauma in the skin - reddish-purple blotchy rash it may result from: - decreased production (toxins, radiation, infection, leukemias) - increased destruction (autoimmune processes) - increased PLTs consumption (DIC) Thrombocytopenia Hemorrhagic spots (petechiae) von Willebrand’s disease • Is the most common genetic bleeding disorder • Results from defect in vWF ( quantitative or functional) • Results in combination of – Platelet function abnormality (vWF) - impaired adhesion – Clotting factor deficiency (factor VIII) - ↑ aPTT (PT is normal) 64 THROMBO-EMBOLIC CONDITIONS • Thrombosis - blood clotting within the CVS which obstruct the blood flow through the CVS (Should be distinguished from extravascular clotting, clotting in wounds and clotting that occurs in the CVS after death). Thrombosis is rather a pathological condition. • Common causes – Roughened endothelial surface (i.e., atherosclerosis, infections, traumas) – Slow blood flow – Hypercoagulobility – Acquired refers to transient or acquired conditions that increase the tendency to clot. This might include antiphospholipid antibodies or a temporary hypercoagulable state such as pregnancy. Also, advanced carcinomas of the pancreas or lung may produce a hypercoagulable state. – Congenital refers to hereditary conditions that increase the tendency to clot. These include Factor V Leiden, prothrombin ,protein C, protein S and antithrombin deficiencies 65 Consequences Formation of emboli (thromboembolism) – braking down of the thrombus and spreading of its particles particles throughout the CVS Thrombosis in the left side of the heart and large arteries → emboli in the brain, kidneys, etc Thrombosis in the venous system and in the right side of the heart → emboli in the pulmonary circulation Disseminated intravascular coagulation (DIC) • Widespread coagulation thrombosis in small blood vessels increased fibrinolysis, and depletion of coagulating factors generalized bleeding • It may result from: - bacterial infections damage) - disseminated cancers (release of procoagulants) - complications of pregnancy - severe catabolic states (endothelial Disseminated cervical cancer metastases (PET imaging) DESSIMINATED INTRAVASCULAR COAGULATION • Reasons – Large areas of necrotic tissue (release of tissue factors into the blood) – Septicemia (activation of clotting by circulation bacteria and bacterial toxins) • Consequences – Consumption coagulopathy • ↓ fibrinogen, thrombocytopenia • ↑ fibrin split products • ↑ PT and PTT 68 CHALENGE YOURSELF 1/5 A baby is born prematurely at 28 weeks gestational age with a birth weight of 1200 g. A few weeks after birth his mother noticed a bleeding tendency in the infant. Blood test revealed a low prothrombin level. Which vitamin can be given to the baby to reduce or to prevent the bleeding tendency? a. Vitamin B12 b. Vitamin B6 c. Vitamin K d. Folic acid e. Vitamin A Answer is C 69 CHALENGE YOURSELF 2/5 • A 72-year-old African-American man undergoes hip surgery. On his third hospital day he experiences chest pain, tachycardia, dyspnea, and a low-grade fever. The man goes into cardiac arrest, and efforts to resuscitate him are unsuccessful. On autopsy a massive pulmonary embolus is discovered. Which of the following, if present, would most likely predispose the patient to this event? (A) Factor VIII defi ciency (B) Low serum homocysteine levels (C) Mutation in the Factor V gene (D) Overproduction of protein C (E) von Willebrand factor defi ciency Answer is C CHALENGE YOURSELF 3/5 Taking aspirin every day can reduce the risk of heart disease because a.it is a powerful vasodilator b it stimulates fibrinolysis c. it prevents atherosclerosis d.it loosens atherosclerotic plaque on arterial walls e. it prevents platelet aggregation Answer is E CHALENGE YOURSELF 5/5 • A 65-year-old man presented with history of acute chest pain that radiates to his left arm. Coronary angiography demonstrates more than 75% occlusion of his coronary artery. He was administered a thrombolytic agent for reestablishment of blood flow to the dying myocardium. The thrombolytic agent activates: The Answer is C A. Heparin B. Throbin C. Plasminogen D. Kininogen E. Prothrombin Fill in the gap please PT Hemophilia A Hemophilia B vWF disease Vit K Def Liver Disease N PTT Platelet Count Bleeding Time Reticulo cyte Megakyr D-Dimer yocyte Schicocy te N N N N No No What is the proper pathway for the extrinsic clotting pathway? A) Contact of blood with collagen, formation of prothrombin activator, conversion of prothrombin into thrombin, conversion of fibrinogen into fibrin threads B) Tissue trauma, formation of prothrombin activator, conversion of prothrombin into thrombin, conversion of fibrinogen into fibrin threads C) Activation of platelets, formation of prothrombin activator, conversion of prothrombin into thrombin, conversion of fibrinogen into fibrin threads D) Trauma to the blood, formation of prothrombin activator, conversion of prothrombin into thrombin, conversion of fibrinogen into fibrin threads What condition leads to a deficiency in factor IX that can be corrected by an intravenous injection of vitamin K? A) Classic hemophilia B) Hepatitis B C) Bile duct obstruction D) Genetic deficiency in antithrombin III A patient suffers from a congenital deficiency in factorXIII (fibrin-stabilizing factor). What would analysis of his blood reveal? A) Prolonged prothrombin time B) Prolonged whole blood clotting time C) Prolonged partial thromboplastin time D) Easily breakable clot A 2-year-old boy bruises easily and has previously had bleeding gums. The maternal grandfather has a bleeding disorder. His physical examination shows several small bruises on the legs. Of which coagulation factor would you suspect this patient to be deficient? A) Prothrombin activator B) Factor II C) Factor VIII D) Factor X An 11-year-old premenstrual female presents with a painful knee after mild trauma. Upon further evaluation you observe soft tissue bruises. The child is an orphan and there is no family history. The foster mother reports no other problems. The aPTT is prolonged and the PT is normal. A complete hematologic workup would yield A) decreased plasma Ca21 B) elevated plasmin C) lack of factor VIII D) decreased platelet number