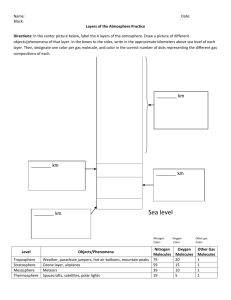
layers of the atmosphere
advertisement

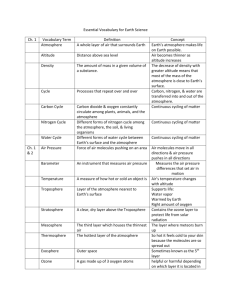
NC Essential Standard: Compare the composition, properties, and structure of Earth’s atmosphere to include: mixtures of gases and differences in temperatures and pressure within layers. All weather and the air you breathe! The lowest and most important layer. Contains 90% of all the gas molecules in the atmosphere! Why? Temperature is highest at the surface of the earth and decreases with altitude. High altitude, colder temperatures Low altitude, warmer temperatures The Sun sends solar radiation through the atmosphere in the form of electromagnetic waves. Rock, soil, and water absorbs the Sun’s light and radiates it back as heat. As you move further from the ground, you are moving further from the source of this reflective heat. Also, there are less molecules in the air as you increase in altitude. Less molecules = less collisions = less heat Second layer of the atmosphere Contains the ozone layer that helps to trap heat in the atmosphere. As you increase in altitude, temperature increases. Warm air sits above cool air = stable = no weather. How is this different than the troposphere? The pilot says, “We are no at our cruising altitude of 30,000 feet.” Why do planes fly so high in the air? Out of the troposphere and into the stratosphere. Fuel costs are lower because there is less friction due to the lower air density…there’s also less turbulence. Middle layer Temperature decreases with altitude (heat is from the stratosphere) Extremely low density of molecules Meteor showers occur here The pressure is so low that liquids would boil at normal body temperature. Your blood would boil without a space suit! Thinking Question! If pressure decreases with altitude…and your blood would boil in the mesosphere…. Does water boil faster in the mountains or at sea level? Answer: The Mountains Fourth layer of atmosphere from the surface of the earth. Very low density of molecules The gases of the thermosphere are increasingly thinner than in the mesosphere. As such, only the higher energy ultraviolet and x-ray radiation from the sun is absorbed. But because of this absorption, the temperature increases with height and can reach as high as 3,600°F (2,000°C) near the top of this layer. However, despite the high temperature, this layer of the atmosphere would still feel very cold to our skin because of the extremely thin air. The total amount of energy from the very few molecules in this layer is not sufficient enough to heat our skin (430 miles to outerspace) Outermost layer of the atmosphere. * Source of all of the energy on Earth. * Energy from the sun must pass through the different layers of the atmosphere before it hits the surface. • The atmosphere is divided into different layers based on the shifts in the temperature increasing or decreasing with altitude. It is a pattern!! Air pressure and density DECREASE as you increase in altitude (less air the further up you go!). Density: the amount of molecules (mass) in a given volume of substance. Gravity pulls molecules down to Earth! = The force of the air weighing down over a unit of area. - - Air molecules constantly bounce off each other and push on all the surfaces they touch. The amount of pressure exerted by our atmosphere is about 14.7 pounds per square inch. Why don’t you feel it? All the air and liquids in your body push outward with the same pressure! = any instrument that measures air pressure. “bar” refers to “air pressure” The flexible chamber on the barometer contracts when the air pressure increases and expands when air pressure decreases. = a line on a map connecting points having the same atmospheric pressure.