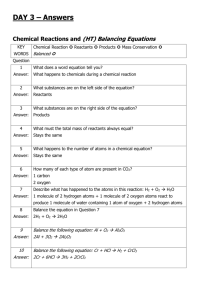
calcium carbonate

Limestone and Building Materials
What is limestone?
What do I need to know?
Must
State that limestone is mainly composed of the compound calcium carbonate, is quarried and can be used as a building material.
Should
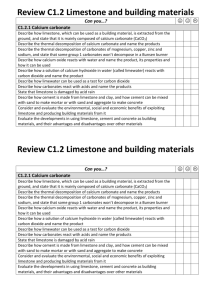
Describe how calcium carbonate can be decomposed by heating to make calcium oxide and carbon dioxide.
Could
Explain the steps in the limestone cycle.
What is limestone?
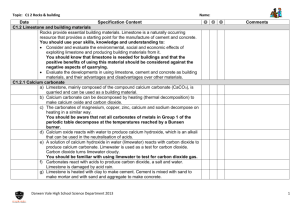
Limestone is mainly composed of the compound calcium carbonate,
We obtain limestone by quarrying
It is a useful building material
Problems with quarrying
• Quarrying is the method that we use to obtain limestone by using explosives to break it into pieces
• It produces dust which may cause breathing difficulties such as asthma
• It is noisy
• It may create more traffic to and from the quarry.
• Destroys the landscape and may impact wildlife.
Benefits of quarrying
• Creates local jobs boosting the local economy
• Provides building materials a valuable raw material for cement
The limestone cycle
Calcium carbonate
Calcium oxide
Calcium hydroxide
Calcium hydroxide solution
Calcium carbonate
• Heat calcium carbonate (limestone) strongly it thermally decomposes to form calcium oxide
• Calcium oxide is another white powder, it reacts exothermically with water to form calcium hydroxide
• Calcium hydroxide reacts with more water to form calcium hydroxide solution .
• Calcium hydroxide solution is known as limewater and is used as a test for carbon dioxide.
• The cloudy substance formed when carbon dioxide is bubbled through limewater is calcium carbonate or limestone.
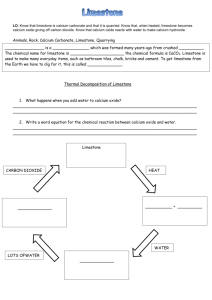
Step 1 - Heating limestone
Thermal Decomposition
• Copy and complete this equation.
CaCO
3
_______ + CO
2
• Write down the word equation for the process
• This is the FIRST STEP in the limestone cycle.
Calculations of mass
This question should be a little bit of revision for you.
CaCO
3
CaO + CO
2
If I heated 100g of calcium carbonate and had 56g of calcium oxide at the end, how much carbon dioxide did I make?
Can you explain why?
The second part of the limestone cycle
• When you react calcium oxide with water it makes calcium hydroxide. The reaction is highly exothermic and produces a lot of heat.
• The reaction has been used to heat self heating cans of hot drinks.
• Can you complete the following word equation?
Calcium oxide + water ______________
The third part of the limestone cycle
• When you blow carbon dioxide into limewater it makes calcium carbonate again. This turns the limewater cloudy.
• Can you complete the following word equation?
Calcium hydroxide + carbon dioxide __________ + water
Summary
State that limestone is mainly composed of the compound calcium carbonate, is quarried and can be used as a building material.
Describe how calcium carbonate can be decomposed by heating to make calcium oxide and carbon dioxide.
Explain the steps of the limestone cycle
Limestone and Building Materials
Other metal carbonates
What do I need to know?
Must
Recall that when calcium carbonate (limestone) is heated it thermally decomposes.
Should
Investigate the decomposition of other metal carbonates
Could
Compare the reactions of different metal carbonates with acids.
Thermal decomposition
Test your knowledge:
When calcium carbonate is _________ it thermally __________ to produce calcium oxide.
Other metal carbonates decompose on heating in a similar way to calcium _________.
Metal carbonate Metal ______ + ________
Word equations
Write the word equations for the decomposition of the following carbonates:
• Magnesium carbonate (MgCO
3
)
• Zinc carbonate (ZnCO
3
)
• Copper carbonate (CuCO
3
)
Check your answers
Magnesium carbonate magnesium oxide + carbon dioxide
MgCO
3
MgO + CO
2
Zinc carbonate zinc oxide + carbon dioxide
ZnCO
3
ZnO + CO
2
Copper carbonate copper oxide + carbon dioxide
CuCO
3
CuO + CO
2
Acid rain and metal carbonates
• Metal carbonates react with acids.
• The products are carbon dioxide, a salt and water.
• This means that limestone buildings erode with acid rain.
Test your knowledge
Metal _________ react with ____ to produce ______
______ a salt and water.
Limestone is __________ by acid rain.
For example:
Calcium carbonate + hydrochloric acid carbon dioxide + calcium chloride + water
Homework
In your book: Title: “Building Materials”
Find out and write down the composition
(make up) of .
Give ONE or more uses for EACH
Summary
Must
Recall that when calcium carbonate (limestone) is heated it thermally decomposes.
Should
Investigate the decomposition of other metal carbonates
Could
Compare the reactions of different metal carbonates with acids.
Limestone and Building Materials
Cement, mortar and concrete
What do I need to know?
Must
State that limestone is heated with clay to make cement
Should
Explain that mixing cement with sand makes mortar and mixing cement with sand and aggregate makes concrete and uses for each of these
Could
Describe the problems of producing cement and the effects of smoke particles from the fuel.
Building materials
1. What is the composition of: a) Cement b) Mortar c) Concrete
2. Give a different use for each
Cement, mortar and concrete
Cement powder + water = cement
Cement powder + water + sand = mortar
Cement powder + water + sand + aggregate = concrete
The facts – cement
Cement powder + water = cement
Strong but brittle
May be used for fence posts
Usually used as mortar or concrete
The facts – mortar
Cement powder + water + sand = mortar
Sand makes it flexible.
Used as a filler between stone and for pointing
Not very strong if hit with a hammer
The facts – concrete
Cement powder + water + sand + aggregate = concrete
Aggregate makes concrete strong.
It can be used as foundations and paving slabs.
Sand content vs. aggregate content
• The more sand you add to mortar the
WEAKER it becomes.
Anomalous results
• A result which does not follow the expected pattern
• Clue – work out the difference between each result to determine the odd one out.
Making cement
• Cement is made by heating calcium carbonate
(limestone) in an oven (called a lime kiln) with
clay. These are two types of lime kiln
What happens in a lime kiln?
• Methane gas is burned (like a Bunsen) to heat the limestone
• This produces CO
2
• Calcium carbonate thermally decomposes
• This produces CO
2 and calcium oxide
• Air is blown through to flush out the CO
2 provide oxygen for the methane to burn and
• Nitrogen from the air is unreacted.
Choosing a fuel
• Burning different fuels at the limekiln may have a number of effects.
• It may be more efficient
• It may cost less or be more cost effective
• However, it may give rise to more dust or particles and this may affect the amount of deposited smoke particles and the health of the local community.
The problem of smoke particles
Examination questions frequently examine the use of a different fuel at a cement works (limekiln). It considers in depth the problems of the smoke particles that can be produced.
In your answer you need to assess the evidence provided and consider concepts such as:
• Carrying out an independent scientific study
• The idea of a fair test
• Assessing data to determine whether it shows the whole picture.
• The difference between particle size (measured in mm) and concentration of particles (measured in ppm or parts per million).
Summary
Must
State that limestone is heated with clay to make cement
Should
Explain that mixing cement with sand makes mortar and mixing cement with sand and aggregate makes concrete and uses for each of these
Could
Describe the problems of producing cement and the effects of smoke particles from the fuel.