Document
advertisement

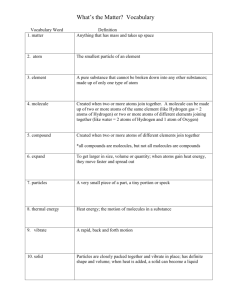
Warm Up: 3/1/13 1)What 2 pieces of information can you identify from a chemical formula? (Ex: H2O) 2)What is the name for the number written to the lower right of a chemical symbol? H2O 3)Determine the types and number of elements in each of the following chemical formulas: a) NH4Br b) Fe2O3 c) KNO3 Chemical Formulas PT 1) Hydrogen 2) H20 3) subscript 4) Chlorine 5) metallic 6) 1 nitrogen and 3 hydrogen - 1 Barium and 2 Chlorine 7) Difference is what they are & their properties, Hydrogen Peroxide has 2 Oxygen PT continued 8) 2 things - what atoms and how many Skillbuilder a. CO2 - 1 carbon 2 oxygen b. Ammonia - NH3 - 1 nitrogen and 3 hydrogen c. calcite - CaCO3 - 1 calcium, 1 carbon, 3 oxygen d. Potassium Hydroxide - KOH - 1 potassium, 1 oxygen, 1 hydrogen e. Baking Soda - NaHCO3 - 1 Sodium, 1 hydrogen, 1 carbon, 3 oxygen Chemistry Unit Andolino & Williams A Quick Review - Atom Defined - smallest building block of a substance or element A Quick Review -Molecule Defined- smallest substance of a compound composed of one or more atoms chemically combined A Quick Review Element Defined - pure substance made up of only one kind of atom All are listed on the Periodic Table of Elements • Examples: Calcium, Sodium, Nitrogen A Quick Review - Compound Defined - 2 or more elements that are chemically combined to make a new substance properties of the elements used to make the compound are changed • Examples: Salt, Water, Alcohol A Quick Review -Mixtures Defined - 2 or more elements physically combined elements used keep their original properties • Examples: salt water, trail mix, rice & beans, Pizza Ways to separate a mixture • • • • Boil to separate something that is dissolved in water Strain (filter)- separate sand/gravel Density differences- scoop off less dense material (salad dressing) Magnet- separate magnetic things from a mixture Chemical Formula Defined - way of writing one molecule of a compound Shows THREE things the elements that make up the compound o metallic element always goes first o the number of atoms of each element called Subscripts o H2O = the 2 is a subscript. ( H= 2 and O= 1) Challenge...Round 1 • • • With your table mates - represent the molecule projected on each slide. Make sure you have the correct elements combined to make the compound Make sure you have the correct number of atoms in each molecule to make the compound Water - H2O 1 molecule of Water has... 2 atoms of Hydrogen 1 atom of Oxygen • • Colorless, Odorless Gas Colorless, Odorless Gas Colorless, Odorless Liquid Salt - NaCl a molecule of salt contains 1 atom of sodium 1 atom of Chlorine • • Shiny, Luster, Silver, Highly Reactive, Solid Yellow, Vaporizes, Poisonous Gas White, Crystals, Edible Ammonia - NH3 Chalk - CaCO3 Methane - CH4 Hydrogen Peroxide - H2O2 Glucose - C6H12O6 Butane - C4H10 Baking Soda - NaHCO3 Citric Acid - C6H8O7 Aspirin - C9H8O4 Caffeine - C8H10N4O2 Citric Acid - C6H8O7 Sugar - C12H22O11 Solutions- is a well stirred mixture Contains a solute and solvent • Solvent- substance which dissolves the solute Example: water, coffee or tea WATER IS THE UNIVERSAL SOLVENT Solute – substance that is dissolved into the solvent Example: powdered drinks or sugar Solute or Solvent? Making a Solution Solubility - how well a solute can be dissolved into a given amount of solvent • Depends on the temperature o Most substances become more soluble as the temperature increases o As temperature increases liquid particles can move faster and dissolve the solute at a faster rate Example: Hot Coffee – sugar dissolves faster compared to an iced coffee Depends on pressure - For gases only Pressure Decreases - allows gas molecules to be farther apart o Gases become less soluble as the temperature increases (hotter). The more energy the particles have the harder they are to trap. This is why soda goes “flat” after the cap has been removed. Pressure Increases – allows more gas molecules to be close together o Gases become more soluble as the temperature decreases (colder). The less energy the particles have the easier they are to trap. o You tend to burp more when soda is cold! Solubility graphs are used to summarize the solubility of different substances at a given mass (grams) of water as temperatures (º C) increase • ABOVE the line the solution is SUPER SATURATED • BELOW the line the solution is UNSATURATED • ON THE LINE the solution is SATURATED Chemical Reaction Defined - process in which new substances with new chemical and physical properties are formed Reactants Products substance changed substance formed What are chemical reactions? http://videos.howstuffworks.com/discovery/29 165-assignment-discovery-chemicalreactions-video.htm Are these chemical reactions? Burning Paper Rusting Burning Gasoline Digestion Conservation of Matter Defined - matter cannot be created or destroyed in a chemical change Mass of Reactants Mass of Products Chemical Equations Defined - describe chemical reactions in a simple way "yield" Reactants Na + Cl Products NaCl Coefficient is the number of units involved in a chemical reaction. 2KI = the 2 represents a coefficient. So, 2 KI units are involved in this reaction. Or KI and KI Examples: 2H2O 2NaCl 2CO2 How do YOU balance this equation? Use the chart below Kinds of Atoms Ag H S Ag + H2S Number of Atoms Ag2S + H2 Unbalanced Equation Kinds of Atoms Mg O Mg + Number of Atoms O2 MgO Unbalance Equation Kinds of Atoms Cl Na Br Number of Atoms Cl2 + NaBr NaCl + Br Unbalanced Equation Kinds of Atoms Cu Cl H S CuCl2 Number of Atoms + H2S CuS + HCl Catalyst Lab