Nuclear Chemistry
advertisement

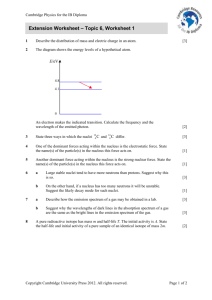
Nuclear Chemistry Nine Mile Oswego, NY Radioisotope – an isotope that is radioactive Example: Carbon-14 Radioactive isotopes can be naturally occurring, or they can be produced by bombarding stable isotopes with high speed particles Stability All nuclei with atomic numbers greater than 83 are unstable They are all radioactive Stability is also dependant upon the ratio of protons to neutrons The closer an isotope is to a 1:1 ratio the more stable it is Transmutation Any change in the nucleus, which causes the element to change into a new element (change of atomic number) Can occur naturally or artifically Natural Transmutation Occurs naturally Single nucleus undergoes decay Example: 3719K → 3718Ar + 0+1e Artificial Transmutation If the change is brought about by bombarding the nuclei by high energy particles Two reactants – a fast moving particle and the target material Example: 3216S + 10n→ 3215P + 11H Equations Mass must be conserved Atomic mass and atomic number must be the same on both sides of the equation Remember 42He 4 = superscript = mass number (atomic mass) = protons + neutrons 2 = subscript = atomic number = protons Equation Examples 1. What is X? 6 Li 3 2. + 10n → 42He + X What is X? 14 C 6 → X + 0-1e Types of Radiation – Table O Alpha particles – helium nucleus, 2 protons, 2 neutrons Beta particles – an electron, negative charge, no mass Positron – electron with a positive charge, no mass Gamma radiation (γ) – similar, but more energy than X-rays, no mass, no charge Particle Mass Charge (amu) Symbol Penetrating Power Shielding Alpha 4 +2 α, 42He Low Paper, clothing Beta 0 -1 β-, 0-1e Moderate Metal foil Positron 0 +1 β+, 0+1e Moderate Metal foil Gamma 0 0 γ High Lead, concrete Charges of Decay Particles •Negative particles will be attracted to positive charges •Positive charges will be attracted to negative charges •Non charged particles are not affected by charges Alpha Decay – unstable nucleus emits an alpha particle Example: 22688Ra → 22286Rn + 42He Beta Decay – unstable nucleus emits a beta particle Example: 21482Pb → 21483Bi + 0-1e Positron Emission – unstable nucleus emits a positron Example: 3719K → 3718Ar + 0+1e Conversion of Mass to Energy E = mc2 – E = energy (J) – m = mass (kg) – c = velocity (speed) of light = 3.0x108m/s Example: How many joules of energy are released if 1.0g is converted to energy? Mass Defect The actual atomic mass of an atom is less than what we would predict based upon the mass of individual protons and neutrons The difference is because energy is released when the protons and neutrons combine The larger the mass defect, the more energy is released upon formation, and the more stable the particle is Examples 1. Calculate the predicted mass of He-4. 1 proton = 1.00728, 1 neutron = 1.00867 • The actual mass of He-4 = 4.00150 • The mass defect is = 2. Convert the mass defect to energy (using E = mc2) Fission Splitting of a heavy nucleus to produce lighter nuclei Nuclear Power Plants Neutron joins with a nucleus of a heavy element Intermediate product is very unstable Splits apart producing Two mid weight nuclei At least one neutron A great amount of energy Fission • Example: 235 U 92 + 1 n 0 9236Kr + 141 Ba 56 + 3 1 n 0 + energy • The three neutrons given off can be reabsorbed by other U-235 nuclei to continue fission as a chain reaction • A tiny bit of mass is lost (mass defect) and converted into a huge amount of energy • See Fission Chain Reaction The neutrons that are emitted can become reactants causing more nuclei to undergo fission and release more energy The reaction can be controlled by limiting the number of interactions between neutrons and nuclei Nuclear Power Plants Main Components Fuel – Uranium or Plutonium Control Rods - absorb neutrons to control the rate of the reaction Containment Structure – building that houses the reactor Main Components Coolant – Water, cools the reaction Cooling Tower – cools the discharge water, releases water vapor Nuclear Power Advantages: Cleaner than conventional fossil fuels – no greenhouse gases or acid rain More efficient, cleaner Disadvantages: Many of the bi-products of the reactions are radioactive (unstable) and have long half-lives, making the storage and disposal of these wastes dangerous Fusion Combining of light nuclei to produce a heavier nucleus The Sun, Hydrogen Bomb Example: 21H + 21H → 42He + energy See Fusion Fusion Advantages Products are not highly radioactive Produces a lot of energy Disadvantages Requires extremely high temperatures and pressures, therefore not yet available to produce energy on Earth Half-Life Time it takes for half of the atoms in a given sample of an isotope to decay Each isotope has its own half-life (Table N) The shorter the half-life of an isotope, the less stable it is Half-life is a constant factor, it is not affected by temperature or pressure Geiger counter can be used to record the decay of an isotope •Use Table N for HalfLife and Decay Modes 1. Calculate the mass of I-131 that remains after 32.28 days, if the mass of the original sample was 100.0g. 2. If 50.0g of a radioactive isotope decays to 6.25g in 60.0 days, what is the isotope’s half-life? 3. 4. 5. What fraction of a phosphorus-32 sample will remain after 28.6 days? 50.0g of cobalt-60 decays for 21 years. How many grams remain after this time? After 14.4 seconds, 3.00g of nitrogen-16 remains. What was the mass of the original nitrogen-16 sample? Dating Each radioactive substance is presently decaying at the same rate as when the substance By comparing the amount of 14C that remains to the amount of 12C that is present, the amount of 14C (and the age) can be calculated Chemical Tracers A radioisotope is used to follow the path of a chemical process Example: C-14 is used to follow the path of carbon in organic reactions Medical Applications • Some radioisotopes have the ability to kill living tissue • Any radioisotope used in medicine must have a short half-life, making it quickly eliminated by the body Medical Examples • Cancer: Cobalt-60 emits large amounts of gamma radiation which can be used to kill tumor cells • Thyroid: Iodine-131 is used in the detection and treatment of thyroid conditions • Gamma Radiation: meats are irradiated to kill bacteria, producing a longer shelf life • Anthrax: Cobalt-60 and Cesium-137 are two sources of gamma radiation that can be used to destroy anthrax Radiation Risks Can damage normal cells High doses can cause illness, death Can cause mutations that can be passed onto offspring