Section 7.3
advertisement

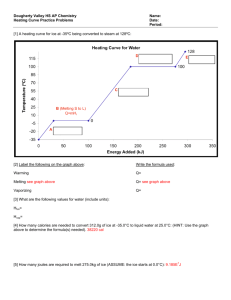
Section 7.3—Changes in State What’s happening when a frozen ice pack melts? Change in State To melt or boil, intermolecular forces must be broken Breaking intermolecular forces requires energy The energy being put into the system is used for breaking IMF’s, not increasing motion (temperature) A sample with solid & liquid will not rise above the melting point until all the solid is gone. The same is true for a sample of liquid & gas Melting When going from a solid to a liquid, some of the intermolecular forces are broken The Enthalpy of Fusion (Hfus) is the amount of energy needed to melt 1 gram of a substance The enthalpy of fusion of water is 80.87 cal/g or 334 J/g All samples of a substance melt at the same temperature, but the more you have the longer it takes to melt (requires more energy). H m H fus Energy needed to melt Mass of the sample Energy needed to melt 1 g Example Example: Find the enthalpy of fusion of a substance if it takes 5175 J to melt 10.5 g of the substance. Example Example: Find the enthalpy of fusion of a substance if it takes 5175 J to melt 10.5 g of the substance. H m H fus H = enthalpy (energy) m = mass of sample Hfus = enthalpy of fusion 5175 J (10.5 g ) H fus 5175 J H fus 10.5 g Hfus = 493 J/g Vaporization When going from a liquid to a gas, all of the rest of the intermolecular forces are broken The Enthalpy of Vaporization (Hvap) is the amount of energy needed to boil 1 gram of a substance The Hvap of water is 547.2 cal/g or 2287 J/g All samples of a substance boil at the same temperature, but the more you have the longer it takes to boil (requires more energy). H m H vap Energy needed to boil Mass of the sample Energy needed to boil 1 g Example Example: If the enthalpy of vaporization of water is 547.2 cal/g, how many calories are needed to boil 25.0 g of water? Example Example: If the enthalpy of vaporization of water is 547.2 cal/g, how many calories are needed to boil 25.0 g of water? H (25.0 g ) 547.2 cal H m H vap H = enthalpy (energy) m = mass of sample Hfus = enthalpy of fusion g H = 1.37×104 cal Increasing molecular motion (temperature) Changes in State go in Both Directions Vaporizing or Evaporating Liquid Melting Freezing Solid Gas Condensing Going the other way The energy needed to melt 1 gram (Hfus) is the same as the energy released when 1 gram freezes. If it takes 547 J to melt a sample, then 547 J would be released when the sample freezes. H will = -547 J The energy needed to boil 1 gram (Hvap) is the same as the energy released when 1 gram is condensed. If it takes 2798 J to boil a sample, then 2798 J will be released when a sample is condensed. H will = -2798 J Example Example: How much energy is released with 157.5 g of water is condensed? Hvap water = 547.2 cal/g Example Example: How much energy is released with 157.5 g of water is condensed? Hvap water = 547.2 cal/g H m H vap H = enthalpy (energy) m = mass of sample Hfus = enthalpy of fusion Since we’re condensing, we need to “release” energy…H will be negative! H (157.5g ) 547.2 cal g H = - 8.6×104 cal Heating Curves Heating curves show how the temperature changes as energy is added to the sample Melting & Freezing Point Temperature Boiling & Condensing Point Heating curve of w ater 150 100 50 0 -50 Energy input Going Up & Down Moving up the curve requires energy, while moving down releases energy Temperature Heating curve of w ater 150 100 50 0 -50 Energy input States of Matter on the Curve Liquid & gas Energy added breaks remaining IMF’s Liquid Only Energy added increases temp Temperature Heating curve of w ater 150 100 50 0 -50 Energy input Solid Only Energy added increases temp Solid & Liquid Energy added breaks IMF’s Gas Only Energy added increases temp Different Heat Capacities Liquid Only Cp = 1.00 cal/g°C The solid, liquid and gas states absorb water differently—use the correct Cp! Temperature Heating curve of w ater 150 100 50 0 -50 Energy input Gas Only Cp = 0.48 cal/g°C Solid Only Cp = 0.51 cal/g°C Changing States Liquid & gas Hvap = 547.2 cal/g Temperature Heating curve of w ater 150 100 50 0 -50 Energy input Solid & Liquid Hfus = 80.87 cal/g Adding steps together Temperature Heating curve of w ater 150 100 50 0 -50 Energy input If you want to heat ice at -25°C to water at 75°C, you’d have to first warm the ice to zero before it could melt. Then you’d melt the ice Then you’d warm that water from 0°C to your final 75° You can calculate the enthalpy needed for each step and then add them together Example Example: How many calories are needed to change 15.0 g of ice at -12.0°C to steam at 137.0°C? Useful information: Cp ice = 0.51 cal/g°C H m Cp T Cp water = 1.00 cal/g°C H m H fus Cp steam = 0.48 cal/g°C Hfus = 80.87 cal/g H m H vap Hvap = 547.2 cal/g Example Example: How many calories are needed to change 15.0 g of ice at -12.0°C to steam at 137.0°C? Warm ice from -12.0°C to 0°C Melt ice Warm water from 0°C to 100°C Boil water Warm steam from 100°C to 137°C Useful information: Cp ice = 0.51 cal/g°C H m Cp T Cp water = 1.00 cal/g°C H m H fus Cp steam = 0.48 cal/g°C Hfus = 80.87 cal/g H m H vap Hvap = 547.2 cal/g H 15.0 g 0.51cal 0 C 12 C g H 15.0 g 80.87 cal 1213 cal H 15.0 g 0.48 cal 91.8 cal g H 15.0 g 1.00 cal 100 C 0 C g H 15.0 g 547.2 cal 1500 cal 8208 cal g gC 137.0 C 100 C 266 cal Total energy = 11279 cal Let’s Practice Example: How many needed to change 40.5 g of water at 25°C to steam at 142°C? Useful information: Cp ice = 0.51 cal/g°C H m Cp T Cp water = 1.00 cal/g°C H m H fus Cp steam = 0.48 cal/g°C Hfus = 80.87 cal/g H m H vap Hvap = 547.2 cal/g Let’s Practice Example: How many needed to change 40.5 g of water at 25°C to steam at 142°C? Warm water from 25°C to 100°C Boil water Warm steam from 100°C to 142°C Useful information: Cp ice = 0.51 cal/g°C H m Cp T Cp water = 1.00 cal/g°C H m H fus Cp steam = 0.48 cal/g°C Hfus = 80.87 cal/g H m H vap Hvap = 547.2 cal/g H 40.5g 1.00 cal 100 C 25 C g H 40.5g 547.2 cal H 40.5 g 0.48 cal 22162 cal g gC 3038 cal 142.0 C 100 C 816 cal Total energy = 26016 cal