lecture seven
advertisement

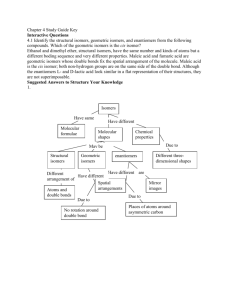
Chapter 9 INTRODUCTION TO STEREOCHEMISTRY Isomers are compounds with the same molecular formula but not identical structures Constitutional isomers are isomers which have the same molecular formula but differ in the way their atoms are connected Constitutional Isomers Constitutional isomers are isomers which have the same molecular formula but differ in the way their atoms are connected Drawing Constitutional or Structural Isomers of Alkanes Example 8.4 Write all the constitutional isomers having the molecular formula C6H14 start by connecting the carbons in a line C C C C CC CC C C CC C C C C C C C C determine the C skeleton of the other isomers C C C Example 8.4 Write all the constitutional isomers having the molecular formula C6H14 H fill in the H to give each C 4 bonds H H H H H C C C C C C H C C H H H H H C H C H C H C C H C C H H H H H H C C C C H C C H H H C H H C H H H H H H H C H H H H H H H H C H C H CC C C H C H C H H H H H H C C C H H H H H H H H C CC C C CC C C C H H H C H H C H H H H Example 8.4 Write all the constitutional isomers having the molecular formula C6H14 convert each to a carbon skeleton formula – each bend and the ends represent C atoms H H H H H H H C C C C C C H H H H H H H H H H H H C H H H C C C H H H H H H H H C C H H C C H H H H H H C C C C C H H H H C H H H H H H H C C C H C H H H H H H H H H H H C C C C H H H C H H C H H H H Stereoisomers have the same molecular formula, maintain the same connectivity, but differ in the way their atoms are arranged in space Conformational isomers (or conformers or rotational isomers or rotamers) are stereoisomers produced by rotation about single bonds, and are often rapidly interconverting at room temperature Conformations of Alkanes and Cycloalkanes Conformations of Ethane ethane Staggered conformation of ethane Newman Projection Sawhorse Representation Eclipsed conformation of ethane Newman Projection Sawhorse Representation ethane Staggered conformation of ethane Newman Projection Sawhorse Representation « Chem3D Embed » Eclipsed conformation of ethane Newman Projection Sawhorse Representation « Chem3D Embed » Conformations of Alkanes and Cycloalkanes Conformations of Butane butane Staggered conformation of butane Eclipsed conformation of butane Conformations of Alkanes and Cycloalkanes Staggered conformation of butane « Chem3D Embed » Eclipsed conformation of butane « Chem3D Embed » Configurational Isomers are stereoisomers that do not readily interconvert at room temperature and can (in principle at least) be separated. Geometric isomers are configurational isomers that differ in the spatial position around a bond with restricted rotation (e.g. a double bond): Geometric (Cis and Trans) Isomers Geometric (Cis and Trans) Isomers result from restriction rotation Compounds with double bonds cis isomer – have same substituents on the same side of the double bond (= Z with more complex molecules having high priority groups on the same side) trans isomer – have the same substituents on the opposite side of the double bond (= E with more complex molecules having high priority groups on opposite sides) Compounds with bonds in a ring: cis isomer – have the same substituents on the same side of the ring trans isomer - have the same substituents on the opposite side of the ring Cis-trans (Geometric) isomerism in Alkenes cis-1,2-dichloroethene trans-1,2-dichloroethene (Z)-1,2-dichloroethene (E)-1,2-dichloroethene « Chem3D Embed » « Chem3D Embed » Cis-trans (Geometric) isomerism in Alkenes *If one of the two carbon atoms of the double bond has two identical substituents, there are no cis-trans isomers for that molecule 1,1-dichloroethene Identifying cis and trans isomers of Alkenes Example 11.3 Two isomers of 2-butene are shown below. Which is the cis isomer and which is the trans isomer cis-2-butene trans-2-butene Naming cis and trans compounds Example 11.4 Name the following geometric isomers. trans-3,4-dichloro-3-heptene cis-3,4-dimethyl-3-octene Identifying Geometric Isomers Example 11.5 Determine whether each of the following molecules can exist as cis-trans isomers: (1) 1-pentene (2) 3-ethyl-3-hexene (3) 3-methyl-2-pentene 1-pentene cis-3-methyl-2-pentene 3-ethyl-3-hexene trans-3-methyl-2-pentene Cis and Trans Fatty Acids cis-9-octadecenoic acid trans-9-octadecenoic acid Cis and Trans Fatty Acids « Chem3D Embed » « Chem3D Embed » Cis-trans (Geometric) isomerism in Cycloalkanes Cis-trans isomers are molecules having the same arrangement of atoms but differ in the spatial orientation of their substituents. cis-1,2-dichlorocyclohexane trans-1,2-dichlorocyclohexane Naming cis-trans Isomers of Substituted Cycloalkanes Example 10.6 Determine whether the following susbstituted cycloalkanes are cis or trans isomers. trans-1,2-dimethylcyclopentane cis-1,2-dimethylcyclopentane Conformations of Alkanes and Cycloalkanes Conformations of Cyclohexane cyclohexane Chair conformation of cyclohexane Boat conformation of cyclohexane Chair conformation of cyclohexane « Chem3D Embed » Boat conformation of cyclohexane « Chem3D Embed » Chair-Chair Interconversion « Chem3D Embed » « Chem3D Embed » Optical isomers are configurational isomers that differ in the 3D relationship of the substituents about one or more atoms. Diastereomers are optical isomers (stereoisomers) that are not enantiomers. Enantiomers are optical isomers that are non-superimposable mirror images. Chirality Chiral objects are objects with left-handed and right-handed forms Achiral objects - objects that have superimposable mirror images Nonsuperimposable mirror images - a mirror image that is not the same as the image itself - chiral objects have nonsuperimposable mirror images Assymetric Center Chirality is not reserved just for objects - molecules can be chiral Chiral molecules - generally molecules containing an asymmetric center Asymmetric (chiral) center - tetrahedral atom bonded to four different groups indicated with an asterisk (*) Chiral Molecules with One Asymmetric Center Molecules with one chiral center would have 2 enantiomers *Chiral molecules would have 2n enantiomers (where n is the number of chiral centers) Chiral vs Achiral « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » How to Represent Enantiomers: Perspective Formulas Perspective Formulas - shows two bonds of the asymmetric center as lines in the plane of the paper, another bond as a solid wedge protruding forward out of the paper, and the fourth bond as a hatched wedge extending behind the paper « Chem3D Embed » « Chem3D Embed » Fischer Projections Fisher Projection - representation of an asymmetric center as the point of intersection of two perpendicular lines Horizontal lines represent bonds that project out of the plane of the paper Vertical lines represent bonds that extend back from the plane of the paper away from the viewer Fischer Projections « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » « Chem3D Embed » Naming Enantiomers -R,S System STEP 1. Rank the groups/atoms bonded to the asymmetric center in order of priority. - rank in terms of atomic mass. Higher atomic mass, higher priority. - let’s have 1-chloro-1-ethanol as an example. 1. 2. 3. 4. Chlorine = 36 amu Oxygen = 16 amu Carbon = 12 amu Hydrogen = 1 amu STEP 2. Rotate the molecule so that the lowest priority group is pointing away from the reader. Naming Enantiomers -R,S System Step 3. Trace your finger around the three highest priority groups in order of the priority. - If the circle is moving counterclockwise the CIP designation is “S”. If the circle is moving clockwise the CIP designation is “R”. « Chem3D Embed » Step 4. Name the enantiomer in terms of its R or S configuration. - the name for this isomer is (S)-1-chloro-1-ethanol Naming Enantiomers -R,S System Assigning Priorities to Groups 1. 2. 3. 4. Oxygen (from CH2OH) = 16 Carbon (from CH2CH3) = attached to carbon Carbon (from CH3) = attached to hydrogen Hydrogen = 1 Naming Enantiomers -R,S System « Chem3D Embed » « Chem3D Embed »