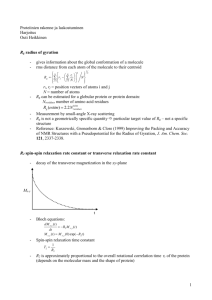
Chem781Part9
advertisement

Chem 781 Part 9
Relaxation and Dynamics, and
advanced methods
• NOE is a relaxation phenomenon
• Measure of dipolar interaction between two
spins
• NOEs can not be observed between two
equivalent nuclei.
• Direct measurement of relaxation time allows
for measurement of dipolar coupling between
equivalent spins
Measurement of longitudinal
relaxation time T1
180
90x
T
Acquisition
Inversion recovery
The experiment is repeated with values of T between 0 and
5 T1
z
z
z
180 x
T
y
y
y
90
90
z
z
y
y
x
Relaxation of longitudinal magnetization
By definition, longitudinal relaxation is the recovery of z magnetization
towards equilibrium.
For two spins IA and IB coupled by dipolar coupling it is
dIzA/dt = (2 W1A + W0 +W2) (I0A – IzA) + (W2-W0) (I0B – IzB)
9.1
If IA ≠ IB (for example 13C-1H) and we decouple IB (1H), then longitudinal
relaxation depends only on the first term and IzB = constant = 0 throughout the
experiment.
As already mentioned in part one of the lecture we get an mono exponential
recovery of z-magnetization:
The above equation assumes Mz(0) = 0 (90° pulse or saturation). The second term
in 9.1 only influences the equilibrium value M0 by accounting for the NOE.
T1 recovery curve
Obtain T1 by fitting recovery curve to exponential equation with Mz(0) = - Mz0 :
Mz(T) = Mz0 [1-2 exp(-T/T1)]
Longitudinal Relaxation by dipolar
coupling
Using the relations for W1, W0 and W2 used for NOE, one obtains for non-equivalent
spins from 9.1:
1/T1A = (2W1A + W0AB + W2) = 1/10D2AB[J(ωA-ωB) + 3J(ωA) + 6J(ωA+ωB)]
(heteronuclear)
For equivalent spins (IA = IB) one obtains from 9.1 with IzB = IzA
1/T1A = (2W1A+2W2AB) = 3/10 D2AB [J(ωA) + 4J(2ωA)]
(homonuclear)
The expressions depend both on the dipolar coupling and the motion of the molecule:
•
DAB = (γAγB ℏ2)/rAB3
• J ( )
c
1 2 c2
contains the distance information
Contains the motion of the molecule
Longitudinal relaxation (T1) and
distance
•
To measure distance using T1 we need to know motion
•
One could try to measure motion (τc) independently, but that is typically not easily
possible
•
2
/
5 /ω0
Relaxation will be most efficient when .τC =
•
This point can be often obtained by changing the temperature and finding the
minimum T1 (the correlation time will depend on temperature)
•
T1(min) only depends on the distance and the field:
1/T1min = 615/910
2
/
5 γA44/(ω0 r6AB)
Example: Dihydride vs. dihydrogen
complex
T1(min) of 0.23 s is only compatible with dihydride structure. However, the value
can not be used to calculate the exact bond distance, since the equation assumed an
isolated pair of protons. There are other protons around, and other relaxation
mechanisms possible.
Schematic Spin-Lattice vs. Spin-Spin
relaxation
s
s
Relaxation of x,y magnetization
(transverse relaxation)
•
Dephasing of x,y magnetization is caused by both random transitions between
levels (longitudinal relaxation) AND incomplete averaging of orientation dependent
shifts (dephasing by chemical exchange)
•
That results in an additional J(0) term
•
1/T2A = 1/10D2AB [4 J(0) + J(ωA-ωB) + 3J(ωA) + 6J(ωA+ωB)]
Spin-spin Spin-Lattice (Same as T1)
Fast tumbling
averages dipolar
coupling to zero
Measurement of T2
We need to separate the effects of inhomogeneity (reversible) from relaxation
of x,y magnetization (irreversible):
90
180
τ
τ
n
Acquisition τ : a few ms
Spin echo experiment
Take a series of experiments and vary the number of 180⁰ pulses with
refocusing delay (n). The intensity of the resulting spectrum will decay
with T2 :
Mx,y = exp(-2nτ/T2)
T1 and T2 vs. correlation time
• For short correlation times (small molecules), T1 = T2
• For long correlation times,(large molecules), T2 is getting shorter and shorter
=> Lines are getting broader as molecule gets larger
Relaxation Mechanisms
Relaxation can occur through many mechanisms:
•
While cross relaxation (and thus NOE) is solely determined by dipolar
coupling, other mechanisms can contribute additive to the overall longitudinal
relaxation rate
•
•
1/T1total = 1/T1dipol + 1/T1CSA + 1/T1Quad + 1/T1SR + 1/T1paramagnetic ...
CSA: Chemical shift anisotropy Quad: Quadrupol coupling SR: Spin rotation
•
For protons and C-H, N-H usually dipolar coupling is dominant, but for non
protonated carbons or quadrupolar nuclei other relaxation mechanisms become
important. In paramagnetic molecules, the electron-nucleus interaction is often
dominant.
Relaxation by chemical shift anisotropy:
•
Chemical shielding can depend on the orientation of the molecule with respect to
the field. While in solution only an average isotropic shift is observed, the nucleus
actually experiences an fluctuating local field.
•
For axial symmetry the magnitude of the field fluctuation is given by the difference
between the orientations with maximum and minimum shielding (σ∥ and σ⊥):
ΔB0CSA = 1/3 γ B0 (σ- σ)
1
T1CSA
c
2 2 2
B0 ( || ) 2
15
1 02 c2
Importance of CSA
• CSA is an important relaxation mechanism for tertiary C=O and C≡ groups
as there are no protons nearby and the anisotropy of the shielding is
particulary large.
• CSA is also important for heavy I = ½ nuclei (103Rh, 183W, ...) As they also
exhibit large chemical shift ranges.
• Note the dependence on B02 for 1/T1CSA. At higher fields this relaxation
mechanism becomes more important. That is good news for metal NMR as
repetition times are reduced, but may cause less than maximum NOE’s for
C-H groups at very high field.
• Measurement of T1 at different fields allows to separate T1CSA from T1DD
and other relaxation
Relaxation from Quadrupol
interaction
•
This mechanism occurs only for I > ½ as those nuclei are not spherical but shaped
like an ellipsoid
•
In a non symmetric environment different orientations of the nucleus relative to the
environment will have different energies
(interaction with electric field gradient).
•
As this energy can be quite large (several MHz) it
is the dominant relaxation mechanism for all
I > ½ nuclei and results in often extremely
short relaxation times for these nuclei.
Consequences of quadrupolar
relaxation
•
I > ½ nuclei usually have very broad lines except when in very symmetric
environments (octahedral or tetrahedral) and/or for nuclei with very small
quadrupolar coupling constant (2D, 11B, 7Li)
•
A very small repetition delay d1 (0.1s) and acquisition time (td = 4k) can be used in
many cases
•
almost never is NOE observed for any of those nuclei (7Li NMR is one exception)
•
For neighboring nuclei the fast relaxation acts like decoupling (if 1/T1 > J) and
couplings are often not observed: that is why we don’t see 14N-H coupling (14N >
99%). Only very large couplings or couplings to low QC nuclei is observed, or in
case of a very symmetrical molecule like NH4+. In some cases, neighboring
coupling partners will appear broadened: one other reason why N-H protons might
appear broadened
Other relaxation mechanisms
• Interaction with unpaired electrons (spin) will be discussed
with NMR of paramagnetic compounds. Note that O2 is
paramagnetic and best NOE results of small molecules require
degassing of sample.
• Spin Rotation: currents induced by rotation of molecule, only
important for very small molecules and in gas phase,
sometimes methyl groups
Problem of measuring ultra large
molecules
T2 relaxation time becomes shorter as
Molecule becomes bigger
Increasing line width not only causes
more overlap, but also at some point
makes magnetization transfer
impossible
Typically, different relaxation
mechanisms are additive.
However, as the motions leading to
different relaxation mechanisms are the
same, the effects can sometimes cancel
or subtract
Motions modulating the different
mechanisms are not independent
1/T1total = 1/T1dipol + 1/T1CSA + 1/T1Quad + 1/T1SR + 1/T1paramagnetic ...
• The different relaxation rates will only add up if the motions are independent
for the different mechanisms
• Since often different interactions are modulated by the same motion, the
different relaxation mechanisms are often not independent
• Cumulative effects of different relaxation mechanisms are not straightforward
Example: Dipolar- and CSA relaxation of N-15
Consequences of correlated relaxation
• Nitrogen N-Ha line will relax faster than the N-Hb line
• For large molecules, T2 for the N-Ha line will be shorter and the line will be
broader, and the N-Hb line will be sharper
• At high enough magnetic field magnitude of CSA will equal dipolar coupling,
and the two interactions will cancel for N-Hb line
Hb
Hb
n0N
•
For a peptide N-H nitrogen, CSA and dipolar relaxation cancel for one doublet line,
and add for the other
•
In a regular N-15 decoupled HSQC the
broader line would still dominate the
linewidth of the spectrum
•
Instead it is better to take an experiment
that observes only the sharp line and
discards the other.
•
=> Transverse Relaxation Optimized Spectroscopy (TROSY)
Small molecule example of TROSY:
Use viscous solvent and low temperature to achieve short T2
Na2HPO3 in Ethylene Glycol / MeOD
P- 31 NMR, no proton decoupling
-
O
H-P O
O
273 K
233 K
223 K
5.5
5.0
4.5
4.0
3.5
3.0
2.5
2.0
1.5
1.0
0.5
ppm
2D TROSY selects
Na2HPO3 P- 31 T=298K
ppm
1
1
31
H{ P} HSQC
no decoupling in f1 and f2
31
H{ P} TROSY
0.5
1.0
1.5
2.0
2.5
3.0
3.5
4.0
4.5
5.0
5.5
6.0
7.6
7.4
7.2
7.0
6.8
6.6
6.4
ppm
7.6
7.4
7.2
7.0
6.8
6.6
6.4
6.2 ppm
Large Molecule application of TROSY
2D 1H-15N HSQC spectrum of the same 35
kDa tumor suppressor protein
2D 1H-15N TROSY spectrum of a 35 kDa tumor
suppressor protein at 900 MHz.
Rubin group, UC Santa Cruz http://qb3.berkeley.edu/qb3/nmr/5.cfm
NMR and dynamics
Relaxation and molecular dynamics
•
If the magnitude of the interaction (i.e. distance) is known, the relaxation times can
be used to probe for molecular motion
•
Example 13C-H bonds: Due to its low natural abundance, and the r-6 dependence on
the distance, relaxation of the 13C nucleus of a C-H group usually solely depends on
dipolar relaxation to the directly attached hydrogen.
As there are many data on C-H bond lengths, 13C relaxation measurements can be
used to probe for molecular motion
•
•
For small molecules the relaxation time is proportional to the correlation time::
C2 H2 2 1
1 C2 H2 2
C
6
6
T1
6Ddiff
rCH
rCH
Example aliphatic chains
•
The above equation is valid for a rigid spherical molecule. In real molecules, fast
internal rotation about single bonds will contribute to the diffusion coefficient,
resulting in different correlation times for rigid and flexible parts of the molecule.
•
Relaxation measurements thus can reveal internal rotations in the MHz range.
2.0 0.96 0.72 0.53
CH3
3.3
1.4
1.0
CH3
0.8 0.58 0.45
Anisotropic and internal rotation
•
Often the overall tumbling of the molecule is anisotropic, i.e. tumbling about
different axes takes place at different rates.
•
Bistolane and the shown cobalt cluster complex are cigar shaped and the relaxation
of the phenyl carbons depends on three diffusion coefficients: tumbling
perpendicular to the main axis (D⊥), tumbling parallel to the main axis (D∥) and
internal rotation RPh. Quantitative analysis of such data can become very tedious:
where A = (3 cos2Θ-1)/4, B = 3 sin2Θ cos2Θ and C = (3 sin4Θ)/4 with Θ the angle
between the C-H bond and the rotational axis.
For Cpara the C-H bond is part of the rotation axis for both internal rotation and D∥.
Dipolar coupling for this for this group is thus only modulated by D⊥ which is
considerably smaller and thus relaxation times for these carbons are much shorter.
Backbone dynamics of proteins
2.5
2.0
R1 1.5
•
Relaxation data reveal
increased flexibility in the
linker region.
•
Also, the difference in
correlation time of the two
domains can be related to
the slightly different sizes
of the domains.
/s-1
1.0
15
R2
/s
10
-1
5
148 residues
-1
MW: 16000gmol
c / ns
Calmodulin:
2+
two globular
domains joined
by a linker
S2
binds 4 Ca
9
8
7
6
0.8
0.6
0.4
0.2
sec. structure
a
a
a
a
a
a
a
0 10 20 30 40 50 60 70 80 90 100 110 120 130 140 150
Residue number
Dynamics from Lineshape
• Relaxation is sensitive for dynamics in the scale of MHz
• Dynamics has an effect on the the line shape of a signal when
interchange between two groups of different chemical shift becomes
of the same order of magnitude as the chemical shift separation
measured in Hz
• Typically 1 s-1 – 10-3 s-1
• Much faster interchange gives one average signal
• Much slower exchange gives two separate signals
Example N,N-dimethylformamide
O
•
•
•
In the intermediate temperature
range the two peaks will broaden
with rising temperature, merge into
one and then gradually sharpen to
give one sharp signal.
The temperature where the signal is
at its broadest is called the
coalescence point
CH3
N
H
CH3
The rate at the coalescence point is
given by
kcoal = π/
2 (νA-νB) = π/
2 B0 ΔδAB
From: “Measuring Rates by NMR”, Hans Reich,
http://www.chem.wisc.edu/areas/reich/nmr/08-tech-03dnmr.htm
Approximations to measure rates
•
•
Finding the exact coalescence temperature is not always trivial or practical
At temperatures below the coalescence point (slow exchange limit) the line width
can be used to approximately determine kAB.
kAB = π Δνex
(Slow exchange limit)
The exchange broadening Δνex is obtained from the line width by
subtracting the natural line width Δνex = Δνobs - Δν0
•
at temperatures above the coalescence point:
kAB = π(νA-νB)2/(2Δνex)
(Fast exchange limit)
Note the need to determine or at least estimate the chemical shift difference
Line shape analysis
•
•
•
exchange can involve more than two species
Intermolecular exchange can involve species of different populations (equilibrium
constant KC)
=> the broadening pattern can become very complex and the above approximations
will not be applicable.
•
Full simulation of line shape possible with a computer to determine rate constants
(line shape analysis) becomes necessary
•
This allows the rate constant to be determined for multiple temperatures
CH3
X
N
(Ph3P)2Cu
S
3
4
1
H
N2
H
5
CH3
CH3
H
X
1
1
N
(Ph3P) 2Cu
S
1
N2
3
4
H
5
CH3
Cp*
Cp*
*
Cp
*
Ir
Cp
Ir
H
k AA'
H
Ir
k
H
Ir
AA'
Co
H
Co
Co
Co
Cp
Co
Cp
Cp
Co
Co
Cp
Co
Cp
Cp
Cp
Cp
A
CH 3
CH
CH 3
A
CH
3
3
k AB
k AB
k
AB
k
AB
CH
Cp*
CH3
Cp*
*
*
Cp
Cp
3
Ir
CH
H
Co
3
Ir
H
Cp
Co
Co
Co
Co
Cp
Ir
H
H
Co
Ir
CH3
Cp
Co
Cp
Co
Cp
B
Cp
k
Cp
Cp
B
k BC
BC
k
BC
k
BC
Cp*
CH3
Cp*
*
Cp
*
CH
CH3
3
Ir
k CC'
Cp
Ir
k
CC'
Ir
CH
3
Ir
Co
Co
Cp
H
Co
Co
Co
Cp
Co
Cp
H
Cp
Cp
H
Co
Cp
Co
H
Cp
C
C
Cp
*
Cp
Ir
Co
H
Co
Cp
Cp
-1
k = 750 s
AB
CH3
5 -1
k = 3x10 s
BC
T = 280 K
k = 34 s
-1
AB
4 -1
k BC= 1.8x10 s
T = 250 K
-1
k = 3.0 s
AB
-1
k = 1500 s
T = 230 K
BC
k = 0.23 s
-1
AB
k = 400 s
T = 210 K
-1
BC
-3 -1
k = 5x10 s
AB
T = 181 K
k = 6.0 s
BC
Experimental
simulated
-1
Exchange and magnetization
transfer
Chemical exchange which is slow on the chemical shift time scale can
have a similar effect on NOE spectra as relaxation via W0 . In order to be
effective two conditions have to be fulfilled:
• one has to observe two separate signals, which means kAB << ΔδAB
• the rate of exchange needs to be larger or at least not much slower than
longitudinal relaxation rate
• The condition for magnetization transfer via exchange is
(νA-νB) > kAB > 1/T1
• Exchange too slow to give broadening still can still be probed by NMR
using magnetization transfer. Usually that is the case for rate constants
of the order 10-2 s-1 - 10 s-1
1D Magnetization transfer
14.9 ppm
-25.4 ppm
Numerical best parameter fit
2D NOESY techniques:
Fitting NOE cross peak intensities
Rate constants and activation
barriers
•
Measuring the temperature dependence of rate constants yields activation barrier
•
Eyring equation:
k AB
•
G
kT RT
e
h
Typically a logarithmic plot is obtained:
kAB : rate constant of exchange
k : Boltzmann constant
k AB h
G H 1 S
ln
kT
R
R T
R
slope
•
The accuracy in particular of ΔS≠
depends strongly on the temperature
range sampled.
int ercept
Sensitivity of NMR Experiments
Signal/
Noise ∼ N∙ 𝑛𝑠 ∙ Polarization ∙ μobserved ∙
γB0/kT
induction ∙ T2*/T1 ∙QProbe ∙ Efficiency
γ 𝐼(𝐼 + 1) ∼ ω0 = γB0
N: num ber of spins
S
concentration
S
isotope abundance
S
tube diam eter
S
length of coil
ns: num ber of accum ulations
T22** : determ ines line width
T11 : determ ines repetition
time
Q : Q uality factor of probe
- coil geom etry
- f ill factor (M icroprobe)
- C ryogenic P robe
P olar ization: Excess of
exc ited spins in lower level
γB 0/kT
μ observed depends on nature of
observed isotope
induction: actual voltage
induced in coil
Ef ficie ncy: how m uch of the
total m agnetitization can be
transferred, loss due to T 2
• Higher magnetic field (20 T currently max.) $$$$
• Reduce noise of electronics: cool detection circuit and preamplifier with cold
heluim gas (cryo probe) or nitrorogen gas (cryo probe Prodigy) $$
• Concentrate sample and scale down dimension of probe ( micro probe)
Improving sensitivity: manipulate
Boltzmann distribution
In some cases, coupling to higher energy levels (rotational,
optical, electron spin) can be used to obtain highly improved
population differences
• Dynamic Nuclear Polarization (DNP)
• Chemically induced nuclear polarization (CIDNP)
• Para hydrogen induced Nuclear Polarization
All methods are currently commercialized for more general use
Dihydrogen Gas is a mixture of
two Spin Isomers
Singlet Para-hydrogen
Triplet Ortho-hydrogen
ms +1
T (triplet)
Itot=1
S (singlet)
Itot=0, ms =0
0
-1
aa abba bb
J
abba
Singlet-Triplet conversion forbidden, and each isomer is stable in pure hydrogen gas
At room temperature: 75% orthohydrogen (expected from Boltzmann distribution)
At 77 K (Liq. N2):
20 K
50 % parahydrogen
> 99% parahydrogen
NOT expected by a simple splitting
of levels by J = 240 Hz
Origin of the high energy difference
ΨDihydrogen = ψtrans • ψvibr • ψrot • ψspin
The whole nuclear wave function needs to be considered
General Pauli Principle:
The total wave function of spin ½ particles is always anti symmetric with respect
to exchange of two particles
Ψ(1,2) = - Ψ(2,1)
• Translation only depends on center of gravity
} Always Symmetrical
• Vibration only depends on absolute distance
• Rotation levels can be symmetrical for even quantum numbers 0,2,4,… (s,d,…) or
anti symmetrical for odd quantum numbers 1,3,5,… (p,f, …)
• Singlet spin function is anti symmetric, triplet spin function is symmetric
• symmetric rotational function can only combine with anti symmetric spin
function and vice versa
Population difference of spin states
determined by Rotational states
Rotational
States
J
2 (sym)
ms +1
T (triplet)
Itot=1
0
-1
aa abba bb
S (singlet)
Itot=0, ms=0
J
abba
Erot
1(anti sym)
m
0
-1
T (triplet) s +1
Itot=1
aa abba bb
S (singlet)
Itot=0, ms=0
J
0 (sym)
S (singlet)
Itot=0, ms=0
abba
• Population difference will be given
by rotational energy, several orders
of magnitude larger than magnetic
interaction
• In absence of a catalyst, there will be
no inter-conversion of triplet to
singlet state
abba
ms +1 0
-1
T (triplet)
Itot=1
aa abba bb
Only combination of symmetrical
rotational state and anti-symmetrical
spin function OR anti symmetrical
rotational state with symmetric spin
function are allowed
J
• Also, no transitions are allowed
between S and T
Analogy to Hund’s rule:Two electrons in two
degeneratre orbitals ϕ1 and ϕ2:
Two possible wave functions , one symmetric and one anti symmetric:
ΨS(1,2) = ϕ1(1) ϕ 2(2) + ϕ1(2) ϕ2(1)
ΨA(1,2) = ϕ1(1) ϕ 2(2) - ϕ1(2) ϕ2(1)
•
•
•
symmetric
anti-symmetric
Energies of ΨA and ΨS will be different, with ΨA lower in energy due to electron
repulsion
As total wave function needs to be anti-symmetric, ΨA will only go with symmetric
spin function, and Ψ S only with anti symmetric spin function
Also note that if ϕ1= ϕ2 (two electrons in the same orbital), ΨA will be zero and
only Ψ S will exist.
ΨS(1,2)
ΨA(1,2)
∆E determined by difference in electron
wave function (coulomb e-- e- repulsion)
How does that help with NMR ?
To be useful for NMR, two conditions need to
be met:
• conversion ortho – para needs to be fast to enrich para-H2 in
reasonable time
• After enrichment, symmetry needs to be broken fast enough to
observe transitions between former T to S states while
maintaining polarization
Presence of metal catalysts to speed
up para to ortho equilibrium
Metal
catalyst
H
H
H
M
H
• Ortho and Para hydrogen usually do not interconvert
• Temporary breaking of H-H bond will allow equilibrium to be achieved
• Any metal that weakly binds hydrogen will do
• Frozen solutions of hydrogenation catalysts stored under hydrogen gas at liquid
nitrogen temperature will do, for example inside NMR tube
Hydrogenation reaction breaks
symmetry of dihydrogen molecule
bb
abba
abba bb
abba
abba
aa
B=0
J>0
reaction outside magnet,
slow break of symmetry
B >0
J >0
A = B
reaction inside magnet
fast break of symmetry
Kirill V. Kovtunov a, Vladimir V. Zhivonitko a, Lioubov Kiwi-Minsker b and Igor V. Koptyug
*aChem. Commun., 2010, 46, 5764-5766
Paramagnetic Molecules
•
•
Molecules with unpaired electrons have net electron spin
Electron paramagnetic resonance same principle as NMR
•
Negative γ from negative charge results in -1/2 (anti parallel) state lower in
energy
E
1
•
ms = + / 2
Similar parameters: g- value and hyperfine
coupling (electron-nucleus scalar coupling)
ms = -1/2
B0
EPR spectra typically displayed in
dispersion mode
H2C(OCH3)
radical
NMR and EPR complimentary
• EPR is possible if electron relaxation
is slow enough.
• NMR is typically not possible in
those molecules
• If electron relaxation is fast, EPR
becomes difficult, but NMR spectra
are observable
• Typically in bi-radicals or metal
complexes
Example transition metal complexes
Lines typically broad and extremely shifted
Shift can be positive or negative
Cp2Co 19 electrons δ(H) = - 50.5 ppm
O
C
CoCp
46 electrons δ(H) = - 30 ppm
CpCo
CoCp
C
O
Origin of extreme chemical shift
•
Shift in paramagnetic complexes
arises from scalar coupling to
electron (contact shift)
•
Coupling is extremely large (MHz),
so the two lines will not be equally
populated
•
Slow electron relaxation will
effectively wipe out signal (but EPR
is possible)
•
Fast electron relaxation will give
average signal that is shifted
towards the higher populated line
Depending on sign of coupling
constant shift is positive or negative
•
hyperfine coupling
a: several MHz
neglecting electron relaxation:
----assuming equal population
of electron a and b states
excess population in lower
electron spin state
slow electron relaxation
-7
(10 s): signal too broad
to be observed
fast electron relaxation
(10-9 -10-10 s):
----assuming equal population
of electron a and b states
excess population in lower
electron spin state
Application: Shift Reagent
Metallo- Protein and Metal DNA
complexes
100
203Tl
129Xe
207Pb 195Pt
171Yb
199Hg
111Cd
113Cd
117Sn
119Sn
125Te
77Se
10
115Sn
1
13C
123Te
29Si
183W
205Tl
107Ag
109Ag
200
300
400
frequency (11 T) / MHz
1H
19F
31P
169Tm
103Rh
89Y
100
15N
187Os
57Fe
% Abundance
Multinuclear NMR
500
0
0.01
40
0.1
1
10
3H
203Tl
205Tl
80
3He
125Te
115Sn
117Sn
119Sn
171Yb
199Hg
77Se
29Si
111Cd 207Pb 195Pt
113Cd
13C
123Te
129Xe
109Ag
107Ag
60
15N
20
183W
19F
1H
31P
169Tm
89Y
103Rh
100
57Fe
187Os
% abundance
I = /2
1
100
D
0
33S
10
6Li
50
100
frequency (11T)/ MHz
150
127I
209Bi
75As
181Ta
133Cs
9Be
139La
35Cl
14N
39K
7Li
11B
141Pr
115In
187Re
121Sn59Co
45Sc
93Nb
55Mn
27Al
51V
23Na
63Cu
71Ga
87Rb
10B
37Cl
105Pd
47Ti
53Cr
25Mg95Mo
67Zn
193Ir 197Au
100
17O
43Ca
1
235U
83Kr
% abundance
I > /2 nuclei
1
200
53Cr
20
0
105Pd
relative sensitivity
17O
87Rb
1
10B
0.1
6Li
40
71Ga
60
10
187Re
121Sn
63Cu
11B
35Cl
80
D
43Ca
33S 47Ti
83Kr
37Cl
67Zn25Mg
95Mo
0.01
209Bi
55Mn
27Al
181Ta
133Cs
139La
23Na
127I
75As
9Be
14N
59Co
141Pr
45Sc
115In
51V
93Nb
7Li
39K
197Au
100
235U
193Ir
% abundance
I > /2 nuclei
1
100