Naming Hydrocarbons Powerpoint
advertisement

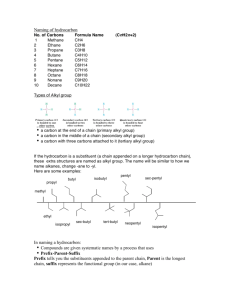
HYDROCARBONS ALKANES, ALKENES AND ALKYNES ORGANIC COMPOUNDS • Carbon based compounds • Can be bonded to other elements (H, O, N, S and halogens) • Carbon makes 4 bonds, which can be single, double or triple • Isomers: same molecular formula (# of each atom) but different arrangement TYPES OF HYDROCARBONS • Saturated: Contain the maximum number of hydrogens, single bonds between all carbons • Unsaturated: Contain 1+ double or triple bonds TYPES OF HYDROCARBONS • Aliphathic Carbons are arranged in chains • Cyclic: Carbons are arranged in rings • Aromatic: Contain a benzene ring TYPES OF HYDROCARBONS Name Alkane Alkene Alkyne Definition Hydrocarbon with only single bonds between carbon atoms. Hydrocarbon with at least one double bond. Hydrocarbon with at least one triple bond. General Formula C2H2n+2 C2H2n C2H2n-2 STRUCTURAL SHORTHAND Explicit hydrogens (those required to complete carbon’s valence) are usually left off of drawings of hydrocarbons H CH3 H H H H3C H C1 C2 C3 C4 H H H H H C1 C2 C3 C4 Line intersections represent carbon atoms HYDROCARBON ROOT NAMES # of Carbons 1 2 3 4 5 6 7 8 9 10 Root Name methethpropbutpenthexheptoctnondec- NAMING ALKANES Based off the number of C atoms in the longest chain 1. Count the number of C’s in the longest chain 2. Determine the appropriate root 3. Add the suffix “ane” 1. 3 carbons 2. 3 Carbons = Prop 3. Propane NAMING BRANCHED ALKANES Based off the number of C atoms in the longest chain 1. Count the number of C’s in the longest chain 2. Determine the appropriate root 3. Use the numbered C’s to give the branches a position number add “yl” suffix 4. Add the suffix “ane” RULES FOR NAMING ALKANES (NOMENCLATURE) For a branched hydrocarbon, the longest continuous chain of carbon atoms gives the root name for the hydrocarbon H3C1 2 3 4 H3C CH3 4 carbon chain = butane NAMING BRANCHED ALKANES Important Rules: 1. Start numbering from the end that will give you the lowest number of branches 2. If there is more than one type of branch, name the branches in alphabetical order 3. If there is more than two of the same type of branch, give the branch a position number and prefixes “di”, “tri” “tetra” etc. 4. Put commas between numbers and hyphens between numbers and letters RULES FOR NAMING ALKANES (NOMENCLATURE) When alkane groups appear as substituents, they are named by dropping the -ane and adding -yl. —CH3 Methyl —CH2CH3 Ethyl —CH2CH2CH3 Propyl —CH2CH2CH2CH3 Butyl H3C H3C Methyl CH3 RULES FOR NAMING ALKANES (NOMENCLATURE) The positions of substituent groups are specified by numbering the longest chain of carbon atoms sequentially, starting at the end closest to the branching. H3C1 2 3 4 H3C Methyl CH3 RULES FOR NAMING ALKANES (NOMENCLATURE) The location and name of each substituent are followed by the root alkane name. The substituents are listed in alphabetical order (irrespective of any prefix), and the prefixes di-, tri-, etc. are used to indicate multiple identical substituents. H3C1 2 3 4 H3C Methyl CH3 Name: 2-methylbutane NOMENCLATURE PRACTICE Name this compound CH3 H3C1 2 Cl 3 4 5 CH3 6 7 9 carbons = nonane 8 H3C9 Step #1: For a branched hydrocarbon, the longest continuous chain of carbon atoms gives the root name for the hydrocarbon NOMENCLATURE PRACTICE Name this compound CH3 H3C1 2 Cl 3 4 5 CH3 6 7 8 9 carbons = nonane CH3 = methyl chlorine = chloro H3C9 Step #2: When alkane groups appear as substituents, they are named by dropping the -ane and adding -yl. NOMENCLATURE PRACTICE Name this compound CH3 H3C1 2 Cl 3 4 5 6 7 9 carbons = nonane CH3 CH3 = methyl chlorine = chloro 8 H3C9 1 9 NOT 9 1 Step #3: The positions of substituent groups are specified by numbering the longest chain of carbon atoms sequentially, starting at the end closest to the branching. NOMENCLATURE PRACTICE Name this compound CH3 H3C1 2 Cl 3 4 5 CH3 6 7 9 carbons = nonane CH3 = methyl 8 chlorine = chloro H3C9 2-chloro-3,6-dimethylnonane Step #4: The location and name of each substituent are followed by the root alkane name. The substituents are listed in alphabetical order (irrespective of any prefix), and the prefixes di-, tri-, etc. are used to indicate multiple identical substituents. NAMING CYCLIC ALKANES ADD PREFIX “CYCLO” Cyclopropane, C3H6 Cyclobutane, C4H8 Cyclopentane, C5H10 Cyclohexane, C6H12 Cycloheptane, C7H14 Remember, explicit hydrogens are left out NAMING ALKENES & ALKYNES 1. Count the number of C’s in the longest chain containing the double/triple bond. • • This is the parent chain, determine the root Number the parent chain so that the double/triple bond has the lowest possible position number 2. Identify the position numbers of branches • Same rules as before • Add the prefix “cyclo” if its cyclic 3. Write the branches in alphabetical order 4. Write the root, including a prefix that identifies the position of the double/triple bond 5. Add the suffix “ene” if double bond or “yne” if triple bond NAMING AROMATICS Benzene: Cyclic 6 carbon ring of alternating single and double bonds (which gives it special properties) 1. Same rules 2. If benzene is the parent chain “benzene” suffix 3. If benzene is a branch group “phenyl” CIS/TRANS ISOMERS Cis Trans NAMING HALOALKANES/ALKENES/ALKYNES • Halogen: group 17, 7 valence shell electrons, form 1 single bond NAMING HALOALKANES/ALKENES/ALKYNES • Same rules as a side change • Side chain name: First part of halogen with –o: • Fluoro, Chloro, Bromo, Iodo • Often Represented as R-X • R = any hydrocarbon chain • X = halogen • Ex: 1. Longest chain is 5 carbons = pentane 2. Only side chain is bromine = bromo 3. Bromine at carbon 1 = 1-bromopentane