Chemistry January Agenda: Mon, Jan 11 RR: How many molecules
advertisement

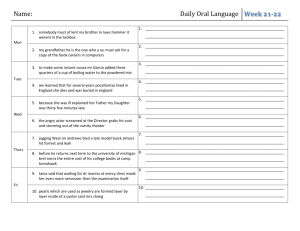
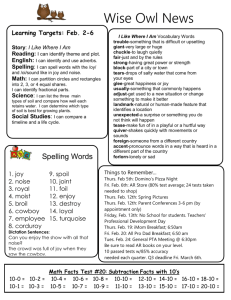
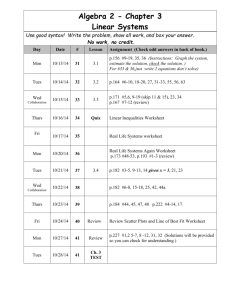
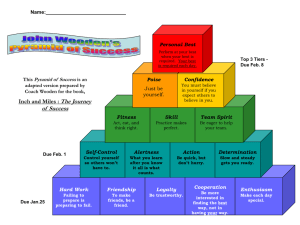
Chemistry January Agenda: Mon, Jan 11 RR: How many molecules of KBr are in 5 moles of KBr? Conversion Factor Station Activity Molar Mass Notes Tues, Jan 12 RR: Study Quiz: Avogadro’s number and the mole Molar Mass Notes Molar Mass WS (due Fri) Wed, Jan 13 RR: What unit do we use to measure atomic mass? Notes Avogadro’s Number WS (due Fri) Molar Mass WS (due Fri, Thurs for Pre-AP Chem) Thurs, Jan 14 RR: What is the difference between a mole and a molecule? Avogadro’s Number WS (Part 1 due Fri) Fri, Jan 15 RR: Pick a problem from Avogadro’s WS Part 2 to review in class. Lab: Molar Mass HW due: Avogadro’s Worksheet (Part 1) Tues, Jan 19 RR: Write generic equations to convert the following: mass moles, particles moles, and mass particles. Finish lab Molar Mass Grams & Particles WS (due Wed) HW due: Avogadro’s WS (Part 1 and 2) Wed, Jan 20 RR: How does the percentage of an ingredient affect a total product? (Think of things that have ingredients.) Finish lab, work on bonus review HW due: Grams & Particles WS Thurs, Jan 21 RR: Study Test: The Mole – Avogadro’s Number and Mass Chemistry Fri, Jan 22 RR: Why is it important to consider % composition in daily life? %Composition Notes %Composition Lab—Bubble Gum Mon, Jan 25 RR: Calculate the % composition when 13.3g Fe combines completely with 5.7g O (example in notes). % Composition Notes % Composition Lab – Bubble Gum finish CW: % composition WS (part 1) Tues, Jan 26 RR: What is the % composition of Ag2O? CW: % composition WS (part 1) Wed, Jan 27 RR: Pick a problem from % composition WS to review. CW: %composition WS (part 2) Empirical Formula Notes Thurs, Jan 28 RR: Take out notes and a clean sheet of paper. Quiz (open notes): % composition Ratio Card Activity CW: Test Corrections Fri, Jan 29 RR: What is an empirical formula? Notes: Empirical Formula Steps CW: Empirical formula practice WS Mon, Feb 1 RR: What is the first step in determining empirical formula? Lab: MgO Tues, Feb 2 RR: Determine the empirical formula of a compound consisting of 38.7% carbon, 9.75% hydrogen, and the rest oxygen. Notes: Molecular Formula Lab: MgO Wed, Feb 3 RR: What is the only conversion factor used in determining empirical and molecular formula? CW: Molecular Formula Practice WS Chemistry Review Problem from sheet Assign review sheet Mixed Practice Thurs, Feb 4 RR: Study Test: %Composition, Empirical and Molecular Formula Fri, Feb 5 RR: How do you know a chemical reaction is taking place? Notes: Reactions CW: Word equations Mon, Feb 8 RR: What kind of smart phone would you want and why? CISM – Reading, vocab, and coding Tues, Feb 9 RR: What is CISM? CISM – Note-taking guide and discussion Wed, Feb 10 RR: What is CISM CISM – extended writing (ad campaign) Thurs, Feb 11 RR: What are the physical states written in chemical reactions? Lab: Chemical Change Fri, Feb 12 RR: How do molecules and compounds form? Reactions Modeling and Intro to Lab Tues, Feb 16 RR: What are the 5 types of chemical reactions? Lab: Classification of Reactions Wed, Feb 17 RR: Write the generic reaction for chemical decomposition. Lab: Classification of Reactions Thurs, Feb 18 RR: Write the generic reaction for chemical synthesis. Lab: Reactions WS Chemistry Fri, Feb 19 RR: Write the generic reaction for double replacement. Lab: Reactions WS Assessment Reactions Notes: Catalysts and Balancing Chemical Equations Mon, Feb 22 RR: Write the general equation for a combustion reaction. Reactions Notes: Types of Reactions & Balancing Equations Classification of Rxns WS (due Friday) Tues, Feb 23 RR: Study Quiz: Classification of Reactions CW: Rxns Packet (Balancing Equations Sheet #1) Wed, Feb 24 RR: What does it mean to be balanced? CW: Rxns Packet (Balancing Equations Sheet #1) Thurs, Feb 25 RR: What does it mean to synthesize? Reactions Notes: Types of Reactions cont. (synthesis) CW: Rxns Packet (Synthesis Predicting) Fri, Feb 26 RR: Write the word equation for 2KClO3 2KCl + 3O2. Reactions Notes: Types of Reactions cont. (decomposition) CW: Rxns Packet (Decomposition Predicting) Mon, Feb 29 RR: Write the skeleton equation, balance it and classify the following word equation: Silver Nitrate (aq) + Aluminum (s) Aluminum Nitrate (aq) + Silver (s) Reactions Notes (single replacement/ activity series) CW: Rxn Packet (single replacement predicting) Tues, Mar 1 RR: What happens to methane gas as it is burned in oxygen? Summarize in a balanced equation. Demo Lab: Combustion Reactions Notes (double replacement/combustion) CW: Rxn Packet (double replacement and identifying reactions) Wed, Mar 2 RR: Write a Skeleton Equation from the word equation: Lithium Hydroxide (aq) + Hydrochloric Acid (aq) Lithium Chloride (aq) + Water Chemistry CW: Review Sheet Types of Reactions Thurs, Mar 3 RR: Write word equation, skeleton equation, and balanced equation for the following and classify: When calcium hydroxide reacts with hydrochloric acid in water, dissolved calcium chloride and water form. This reaction gives off heat. CW: Review Sheet – Types of Reactions Fri, Mar 4 RR: Study Test: Chemical Reactions & Balancing Equations