- The 1st Al Jahra Hospital International Conference in

Pneumonias
HAP/HCAP/VAP
Salim A Baharoon MD
Infectious Disease / Critical Care
King Saud Bin Abdulaziz University
Riyadh
DEFINITIONS
•
HAP
: Pneumonia that occurs
48 hours or more after admission and did not appear to be incubating at the time of admition.
•
Early and Late onset
•
VAP
: A type of HAP acquired at
48-72 hours after intubation
.
•
Early and Late onset
•
HCAP
: Non hospital patient with healthcare contact
•
IV therapy, wound care, chemotherapy within 30 days
•
Nursing home or long term care facility (Nursing Home Pneumonia)
•
Hospitalization >2 days ore more in past 90 days
•
Attendance at hospital or HD within 30 days
•
Family member with a MDR pathogen
ATS/IDSA Am J Respir Crit Care Med. 2005;171: 388-416
DIAGNOSIS
•
Progressive infiltrate on lung imaging and clinical characteristics such as:
•
Fever
•
Purulent sputum
•
Leukocytosis
•
Decline in oxygenation
•
Radiographic findings plus two of the clinical findings.
•
69% sensitivity and 75% specificity for pneumonia (autopsy as reference)
IMPERFECT DIAGNOSTIC TESTS
• Blood cultures, limited role, sensitivity is only 8% to 20%
• Sputum neither sensitive, nor specific
• Tracheo-bronchial aspirates- high sensitivity
• does not differentiate between pathogen and colonizer
• Quantitative cultures increase specificity of the diagnosis of HAP.
• BAL, PSB’s do not differ from less invasive tests in terms of sensitivity, specificity or, more importantly, morbidity and mortality.
• Negative lower respiratory tract cultures can be used to stop antibiotic therapy in a patient who has had cultures obtained in the absence of an antibiotic change in the past
72 hours.
• Role of rapid diagnostic test (PCR) (Multiplex PCR)
EPIDEMIOLOGY
•
HAP is the second most common nosocomial infection in the US
•
HAP increased hospital stay by an average of 7-9 days per patient
•
Estimated occurrence of 5-10 cases per 1,000 hospital admissions
•
0.88 per 1000 patients admission in Taif (1999-
2003)
•
0.5 per 1000 patient days of admission in Iran
•
HAP accounts for up to 25% of all
ICU infections and more than 50% of antibiotics prescribed
Study of 4543 pts. with Culture Positive
Pneumonia: Incidence (%)
Kolle MH, et al. Epidemiology and outcomes of healthcare associated pneumonia: results from a large US database of culture positive pneumonia. Chest 2005;128:3854 62
OUTCOME
•
HAP-associated mortality remains the leading cause of death among hospital-acquired infections
•
Crude mortality of HAP is 30-70%
•
Attributable mortality is 20-50%
•
Worse outcomes in patients with bacteremia, medical rather than surgical illness, ineffective and late antibiotic therapy.
P<.0001
P>.05
P<.0001
Kollef MH, et al. Chest. 2005;128:3854-62.
MORTALITY AND TIME OF PRESENTATION OF HAP
P<.001
50
40
30
20
10
*
0
*Upper 95% confidence interval
None
P<.001
*
P = .504
*
Early Onset Late Onset
Nosocomial Pneumonia
Ibrahim, et al. Chest.
2000;117:1434-1442.
MRSA INFECTION
Crit Care 2006:10(3):R97.
HAP: NON-VENT VS. VENTED PTS.
Pennsylvania study on nosocomial pneumonia, 2009-2011
Davis J. The breath of hospital-acquired pneumonia: nonventilated versus ventilated patients in
Pennsylvania. Focus on Infection Prevention. Pennsylvania Patient Safety Advisory. 2012;9:99-105.
ETIOLOGY
• Aerobic gram-negative bacteria:
• P. aeruginosa, Escherichia coli, Klebsiella pneumoniae , and Acinetobacter species
• Gram-positive cocci
• S. pneumonia.
• H. influenzae
• Staphylococcus aureus (50% in ICU due to MRSA)
• More common in patients with diabetes mellitus, head trauma and those hospitalized in the
ICU.
• Oropharyngeal commensals (viridans group streptococci, coag-negative Staph,
Neisseria species and
Corynebacterium
) may be relevant in mostly immunocompromised patients.
RESULTS, TIME OF INFECTION
• HAP:
• Early onset (0-4 days): S. pneumoniae, H. influenzae
• Late onset (5+ days): oxacillin resistant S. aureus, P. aeruginosa
• VAP:
• Early onset (0-4 days): oxacillin susceptible S. aureus, S. pneumoniae, Hemophilus sp.
• Late onset (5+ days): Acinetobacter sp. and S. maltophilia
PATHOGENS AMONG PNEUMONIA TYPES
Kollef MH,et al. Chest .2005;128:3854-62.
CAP HCAP HAP VAP
PATHOGENS ASSOCIATED WITH NAP
40
P = .003
35
30
25
20
15
P = .043
P = .408
P = .985
P = .144
10
5
0
PA MSSA MRSA
Pathogen
ES
Ibrahim, et al. Chest.
2000;117:1434-1442.
SM
Early-onset NP
Late-onset NP
PA = P aeruginosa
OSSA = Oxacillin-sensitive
S aureus
ORSA = Oxacillin-resistant
S aureus
ES =
SM =
Enterobacter species
S marcescens
ETIOLOGY
•
Fungal pathogens: most common is
Candida and
Aspergillus
•
Most commonly in organ transplant or immunocompromised, neutropenic patients.
•
Aspergillus
- contaminated air ducts or local construction.
•
Candida
- common airway colonizer and rarely requires treatment.
ETIOLOGY
•
Viral Pathogens: low incidence in immunocompetent hosts.
•
Influenza A is the most common viral cause of HAP and
HCAP in adults.
•
Risk for secondary bacterial infection “super-infection”
•
Streptococcus, H. influenza, Group A Streptococcus, S. aureus
MDR RISK FACTORS
•
Host risk factors for infection with MDR pathogens include:
•
Treatment with antibiotics within the preceding 90 days.
•
Current hospitalization of >4 days
•
High frequency of antibiotic resistance in the community or hospital unit
•
Immunosuppressive disease and/or therapy
•
Hospitalization for >/= 2 days within the last 90 days
•
Severe illness
•
Antibiotic therapy in the past 6 months
•
Poor functional status
Colonization Aspiration
HAP
PATHOGENESIS
•
Number and virulence of organisms entering the lower respiratory tract and response of the host.
• microaspiration of organisms which have colonized the upper respiratory/gastrointestinal tract
•
Hospitalized patients tend to become colonized with organisms in the hospital environment within 48 hours.
•
Common mechanisms include: mechanical ventilation, routine nursing care, lack of hand washing of all hospital personnel.
•
Disease state also plays a role: alteration in gastric pH due to illness, certain medications, malnutrition and supplemental feedings.
M
MECHANISMS THAT LEAD TO ORAL AND OROPHARYNGEAL GNR COLONIZATION
Lam OLT, et al. Effectiveness of oral hygiene interventions against oral and oropharyngeal reservoirs of aerobic and facultatively anaaerobic graminegative bacilli. AJIC 2012;40:175-82.
WHICH PATIENTS ARE AT RISK?
• Liver disease prior to and during transplantation
• End-stage renal disease undergoing hemodialysis
• Cardiovascular disease undergoing surgery
• Abdominal cancer, head and neck cancer
• Leukemia
• COPD
• Cerebral palsy
• Asthma, stroke, chronic bronchitis, pharyngitis, HIV infection, diabetes, alcoholism, Parkinson’s Disease
• Hospitalized, Institutionalized elderly individuals
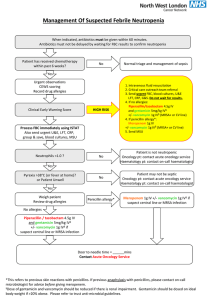
MANAGEMENT
HOSPITAL ADMISSION
• Decision to admit remain clinical
• Severity scores can help.
• CURB-65 criteria (>2, more-intensive treatment)
• Confusion
• Urea 7 mmol/L (20 mg/dL)
• Increased respiratory rate >30
• low blood pressure (SBP <90 or DBP <60)
• Pneumonia Severity Index (PSI)
• uses demographics, the coexistence of co-morbid illnesses findings on physical examination, vital signs and essential laboratory findings
PSI SCORE
INITIAL APPROPRIATE ANTIBIOTIC THERAPY
A Study by Kollef and Colleagues Evaluating the Impact of Inadequate Antimicrobial Therapy on Mortality
60
52 * *P<.001
50
40
42
*
30
20
10
24
0
All-Cause Mortality
Inadequate antimicrobial treatment
(n=169)
18
Infection-Related Mortality
Adequate antimicrobial treatment
(n=486)
ATS=American Thoracic Society; IDSA=Infectious Diseases Society of America.
Adapted from Kollef MH et al. Chest.
1999;115:462-474.
ATS/IDSA. Am J Respir Crit Care Med . 2005;171:388-416.
EFFECT OF TIMING ON SURVIVAL
Time from hypotension onset (hours)
Crit Care Med 2006;34:1589-96
JAMA 2010 The outcome of patients with sepsis and septic shock presenting to emergency departments in Australia and New Zealand.
Crit Care Resusc.
2007 Mar;9(1):8-18.
Antipseudomonal cephalosporin
OR
Antipseudomonal carbepenem
OR
β -Lactam/ β -lactamase inhibitor
Plus
Antipseudomonal fluoroquinolone
OR
Aminoglycoside
Plus
Sever pneumonia, necrotizing or cavitary
Anti MRSA infiltrates, empyema
Anti Legionella pneumophila and anti
Viral
INITIAL EMPIRIC THERAPY IN PATIENTS
WITHOUT RISK FACTORS FOR MDR PATHOGENS
Potential Pathogens
Streptococcus pneumoniae
H influenzae
Methicillin-sensitive S aureus (MSSA)
Antibiotic-sensitive, enteric, gram-negative bacilli
E coli
K pneumoniae (ESBL-)
Enterobacter spp
Proteus spp
Serratia marcescens
Recommended Antibiotic
Ceftriaxone/Azithromycin or
Levofloxacin, moxifloxacin, or ciprofloxacin
† or
Ampicillin/sulbactam/Azithromycin or
Ertapenem/Azithromycin
Adapted from ATS/IDSA. Am J Respir Crit Care Med.
2005;171:401. Table 3.
LINAZOLID VS VANCOMYCIN IN PNEUMONIA
• Retrospective study suggest survival benefit than vancomycin in MRSA pneumonia (Chest
2003;124;1789-1797.)
• Meta-analysis in MRSA pneumonia: non-inferior than glycopeptide (2010)
• Latest randomized, double blinded trial suggest better
(non-inferior) clinical success than vancomycin (2010 idsa abstract)
KPC
• Combination therapy
• Synergistic testing
• Suggested regimens include colistine plus tigecycline plus carbapenem/rifampin
• Other drugs include fosfomycin, aztreonam
PREVENTION
Davis J. The breath of hospital-acquired pneumonia: nonventilated versus ventilated patients in
Pennsylvania. Focus on Infection Prevention. Pennsylvania Patient Safety Advisory. 2012;9:99-105.
ASPIRATION PRECAUTION BUNDLE (APB)
• Ensure bedside swallow screen completed (if failed, physician order for speech consult/NPO status
• HOB elevated 30 degrees or greater
• Oral care every 4 hours (brush teeth every 12 hours)
• No straws
• Ambulate/up in chair TID and prn
• Sit upright 90 degrees for meals/snacks
• Observe patient during meals (check temperature
60 minutes after meal for fever spike)
• Incentive spirometry (IS), Acapella (preferred) or
PEP therapy
• Suction set-up in patient room
• Order Aspiration Precaution on SBAR
Amulti-disciplinary group comprised of nursing, speech pathology, respiratory therapy and infection prevention developed an Aspiration Precaution
Bundle (APB). respiratory therapy and speech therapy participation such as oral care every four hours,
Acapella or PEP therapy and bedside swallow screening.
In addition, a laminated sign was created to place in the patient room
SUMMARY
• HAP is a leading infection among all hospital acquired infections
• HAP is associated with high mortality, long hospital stay, high economic burden
• HAP is still diagnosed with relatively non specific methods
• HAP etiology vary between geographical locations and each region should have real-time data
• Treatment of MDR organism is posing a very significant problem
• Prevention of HAP through established protocols
Thank you
Questions?
MODIFIABLE RISK FACTORS: INTUBATION AND
MECHANICAL VENTILATION
• Intubation and mechanical ventilation increase the risk of HAP 6-21 fold.
• NIPPV, data shows use to avoid reintubation may be associated with more incidence of HAP.
• Sedation protocols to accelerate ventilator weaning.
• Reintubation increases the risk of VAP
• Oral gastric and tracheal tubes rather than nasal may reduce incidence of sinusitis and subsequent lower respiratory tract infection (HAP).
• Limiting use of sedative and paralytic agents that depress cough.
• Keep endotracheal cuff to >20 cm H2O
MODIFIABLE RISK FACTORS
• Strict infection control
• Alcohol-based hand disinfection
• Microbiologic surveillance with timely data on local MDR pathogens
• Removal of invasive devices
• Programs to reduce or alter antibiotic-prescribing practices
MODIFIABLE RISK FACTORS: MODULATION OF
COLONIZATION: ORAL ANTISEPTICS AND
ANTIBIOTICS
• Oropharyngeal colonization is an independent risk factor for ICU-acquired HAP by enteric gram-negative bacteria and
P. aeruginosa
• Oral antiseptic chlorhexidine significantly reduced rates of nosocomial infection in post-operative patients and is routinely used in the ICU as part of “oral care”.
• Selective decontamination fo the digestive tract (SDD): using non-absorbable antibiotics either orally or through GT has shown benefit in reducing HAP/VAP.
However not widely used in the US due to risk for drug resistance.
MDR: STRESS BLEEDING PROPHYLAXIS,
TRANSFUSION, AND GLUCOSE CONTROL
• H2 blockers have shown an increased risk for VAP, risk vs. benefit for stress bleeding should be considered
• Multiple studies have identified allogeneic blood products as a risk factor for post-operative pneumonia, and the time length of blood storage as another risk factor. Blood transfusion is usually limited to Hb <7 in the patient who has no active bleeding.
• Hyperglycemia is an additional risk for blood stream infection, increased duration of mechanical ventilation increasing risk for HAP/VAP.
FOUR MAJOR PRINCIPLES UNDERLIE THE MANAGEMENT
• Avoid untreated or inadequately treated HAP, VAP or HCAP, failure to do so is a consistent factor associated with increased mortality.
• Recognize the variability of bacteriology from one hospital to another, one department from another and one time period to another.
• Avoid the overuse of antibiotics by focusing on accurate diagnosis, tailoring therapy and limit duration of therapy to the minimal effective period.
• Apply prevention strategies aimed at modifiable risk factors.
VAP VS. HAP FLORA
• Study of VAP and HAP pathogens for purposes of optimizing therapy
• University of North Carolina Hospitals study conducted system wide,
2000-2003
• Used definitions as described by ATS
• Did not include CAP or HCAP
• Specimens obtained via bronchoscopy, expectorated sputum, or tracheal aspirates
Weber DJ, et al. Microbiology of ventilator associated pneumonia compared with that of hospital acquired pneumonia. Infect Control Hosp
Epidemiol 2007;28:825 31
RESULTS, EPIDEMIOLOGY
588 LOWER RESPIRATORY THERAPY TRACT INFECTIONS IN 556 PATIENTS
INCIDENCE OF PNEUMONIA: 0.37%
Assessment of Non-Responders
Wrong Organism
Drug-resistant Pathogens:
(Bacteria, Mycobacteria, Virus, Fungus)
Inadequate Antimicrobial Therapy
Wrong Diagnosis
Atelectasis
Pulmonary Embolism
ARDS
Pulmonary Hemorrhage
Underlying Decease
Neoplasm
Complications
Empyema or Lung Abscess
Clostridium Difficile Colitis
Occult Infection
Drug Fever
RESULTS, PATHOGENS
PATHOGENS ISOLATED FROM 92.4% OF PATIENTS WITH VAP AND 76.6% FROM HAP
PATIENTS
RESULTS, TIME OF INFECTION
• Pathogens statistically associated with VAP:
• Early onset (0-4 days): oxacillin susceptible S. aureus, S. pneumoniae, Hemophilus sp.
• Late onset (5+ days): Acinetobacter sp. and S. maltophilia
• HAP:
• Early onset (0-4 days): only S. pneumoniae.
• Late onset (5+ days): oxacillin resistant S. aureus and
P.aeruginosa
VAP etiology
Other
Staph areus
Pseudomonas
PATHOGENS CAUSING NOSOCOMIAL PNEUMONIA (TRENDS OVER TIME)
NATIONAL NOSOCOMIAL INFECTIONS SURVEILLANCE SYSTEM
20
15
10
30
25
5
0
19751 1992 –19982 20031
1. Gaynes R, et al. Clin Infect Dis .2005; 41:848-54. NNIS system report. Am J Infect Control. 2000; 28(6):429-48.
2. Richards MJ, et al. Infect Control Hosp Epidemiol. 2000; 21:510-5.
S aureus
P aeruginosa
Enterobacter spp.
E coli
K pneumoniae
Serratia marcescens
Acinetobacter spp
Lung Penetration Concentration vs MIC
90 of Linezolid
Against Gram-Positive Organisms
• 5 doses of linezolid
600 mg q12h were administered orally to
25 healthy volunteers
• Plasma and pulmonary epithelial lining fluid
(ELF) linezolid concentrations exceeded MIC
90 for staphylococci and streptococci through the dosing interval
MIC
90
=minimum concentration needed to inhibit 90% of organisms.
Adapted from Conte JE Jr et al. Antimicrob Agents Chemother . 2002;46:1475-1480.
Time After Last Dose (h)
Epithelial lining fluid
Plasma
MIC
9 0
S aureus
MIC
9 0
Enterococcus spp
MIC
90
S pneumoniae
PHARMACOKINETIC CHARACTERISTICS OF
LINEZOLID IN ADULTS
Parameter
Oral bioavailability
Ingestion of food
Effect
100%
No dose adjustment
Volume of distribution Total body water, 40 L to 50 L
Dosage formulations
Distribution
Protein binding
IV, tablets, oral suspension (PO)
Readily distributes into well-perfused tissues
31%, independent of drug concentration
LINEZOLID PHARMACOKINETICS IN VAP
16 critical-care patients with late-onset VAP ( ≥5 days on the ventilator)
Pharmacokinetic profile was evaluated after 2 days of linezolid (600 mg q12h IV) therapy. ELF samples were collected by mini-BAL brush
Steady State Concentrations in 16 VAP Patients
Plasma (mg/L)
ELF (mg/L)
Peak
17.7
±4
14.4
±5.6
Trough
2.4
±1.2
2.6
±1.7
Boselli E et al. Crit Care Med . 2005;33:1520-1533.
FIRST PROSPECTIVE COMPARISON OF LINEZOLID VS
VANCOMYCIN FOR EMPIRIC TREATMENT OF NOSOCOMIAL
PNEUMONIA (NP)
A randomized, double-blind, multicenter, multinational, comparator-controlled trial to compare the safety and efficacy of linezolid versus vancomycin for NP
70
60
50
40
30
20
10
0
53 52
55
46
58
50
86/161 74/142 31/56 19/41 18/31 10/20
Intent-to-treat (ITT)
Linezolid 600 mg q12h IV
S aureus NP MRSA NP
Vancomycin 1 g q12h IV
Safety and efficacy of linezolid versus vancomycin were compared in 402 patients with NP, including VAP;
398 patients received at least 1 dose of study medication. Patients were treated for 7 to 21 days, with optional aztreonam 1 g to 2 g q8h. Clinical cure rates were assessed 12 to
28 days after end of therapy.
Rubinstein E et al. Clin Infect Dis . 2001;32:402-412.
Data on file. Pfizer Inc.
SECOND PROSPECTIVE COMPARISON OF
LINEZOLID VS VANCOMYCIN FOR EMPIRIC
TREATMENT OF NP
A randomized, double-blind, multicenter, multinational, comparator-controlled trial to compare the safety and efficacy of linezolid versus vancomycin for NP.
70
60
50
40
30
20
10
0
53 52
49
42
135/256 128/245
ITT
40/81 40/95
S aureus NP
Linezolid 600 mg q12h IV
60
18/30
29
12/41
MRSA NP
Vancomycin 1 g q12h IV
The safety and efficacy of linezolid IV versus vancomycin IV were compared in 623 patients with NP, including VAP. Patients were treated for 7 to 21 days, with optional aztreonam 1 g to 2 g q8h. Clinical cure rates were assessed 15 to 21 days after end of therapy.
Wunderink RG et al. Clin Ther . 2003;25:980-992.
Data on file. Pfizer Inc.
LINEZOLID DEMONSTRATES EXCELLENT EFFICACY IN A
RETROSPECTIVE ANALYSIS OF TWO PROSPECTIVE CLINICAL TRIALS
A retrospective analysis of the combined results from the 2 prospective, identical design trials in 1019 patients with NP including ventilator-associated pneumonia (VAP)
70
59
60
53
52 52
50 43
40
36
30
20
10
0
221/417 202/387 70/136 59/136 36/61 22/62
ITT S aureus NP
Linezolid 600 mg q12h IV
MRSA NP
Vancomycin 1 g q12h IV
Linezolid was equally effective in the ITT and S aureus NP populations ( P =NS).
The outcome difference in the MRSA NP subgroup is provided as a descriptive measure only.
No further inference should be drawn due to the retrospective nature of the analysis ( P <.01).
Wunderink RG et al. Chest . 2003;124:1789-1797.
Data on file. Pfizer Inc.
LINEZOLID DEMONSTRATES EXCELLENT EFFICACY IN A
RETROSPECTIVE ANALYSIS OF TWO PROSPECTIVE CLINICAL TRIALS
A retrospective analysis of 544 patients with VAP from the two prospective, identical design trials in 1019 patients with NP.
80
70
62
60
50
45
49
37
35
40
30
20
21
10
0
103/227 76/207 43/88 32/91 23/37 7/33
ITT S aureus NP
Linezolid 600 mg q12h IV
MRSA NP
Vancomycin 1 g q12h IV
Linezolid was equally effective in the ITT and S aureus NP populations ( P =NS).
The outcome difference in the MRSA NP subgroup is provided as a descriptive measure only.
No further inference should be drawn due to the retrospective nature of the analysis ( P <.01).
Kollef MH et al. Intens Care Med.
2004;30:388-394.
Wunderink RG et al. Chest . 2003;124:1789-1797.
Data on file. Pfizer Inc.
100%
80%
60%
40%
20%
0%
VANCOMYCIN FAILURE DESPITE
ADEQUATE MIC IN MRSA BACTEREMIA
* P = .01
9
21
56
MIC ≤ 0.5
Sakoulas G, et al. J Clin Microbiol 2004;42:2398 – 402
10
MIC 1-2
Failure
Success
HIGHER VANCOMYCIN MICS ASSOCIATED WITH HIGHER
MORTALITY RATES
• Relationship of vancomycin MIC to mortality in patients with
MRSA HAP, VAP and HCAP . Chest 2010 June 17.
• An increase of 1 Vancomycin MIC leads to odds ratio of death as 2.97 folds .
Inadequate Antimicrobial Therapy
Vancomycin for MRSA NP/VAP
• 40% failure rate for MRSA NP with vancomycin at standard dosing (1 g q12h)
• Despite appropriate therapy with glycopeptides, mortality in VAP with MRSA > VAP without MRSA
• In VAP / severe sepsis underdosing
• Enhance renal blood flow
• Increase volume of distribution (hyperdynamic)
• Suggest a higher dose (trough 15-20 mg/L) than traditional dosage (5-15 mg/L)
ATS/IDSA. Am J Respir Crit Care Med . 2005;171:388-416 .
Craven DE et al. Infect Dis Clin N Am.
2004;18:939-962.
Rello J, et al. Crit Care Med 2005; 33:1983 –7.
HETERORESISTANCE
• Etest Macromethod: using a higher inoculum to detect the presence of a less susceptible subpopulation
• J Clin Microbiol 2007;45:329-32.
The annals of pharmacotherapy 2010;44:844-850
.
POTENTIAL BENEFIT OF LINEZOLID
SUMMARY OF CLINICAL TRIALS FOR NOSOCOMIAL
PNEUMONIA DUE TO MRSA
LINEZOLID 600 MG IV Q12H VS VANCOMYCIN 1 G IV Q12H
Trials
Rubinstein,
CID 2001
Prospective
RCT, N=396
Wunderink,
Clin Ther 2003
Prospective
RCT, N=623
Clinical response (ITT)
53% vs 52%
(p = 0.79)
52% vs 52%
(p = NS)
Wunderink,
Chest 2003
Kollef,
ICM 2004
Retrospective
N=1019
Retrospective
N=544
MRSA
59% vs 35%
(p < 0.01)
MRSA
62% vs 21%
(p = 0.001)
-
Microbiological eradication
67% vs 71%
(p = 0.69)
-
Survival
61% vs 53%
(p = NS)
MRSA
60% vs 22%
(p = 0.001)
-
MRSA
80% vs 63%
(p = 0.03)
MRSA:
84% vs 61%
(p = 0.02)
LINEZOLID VS VANCOMYCIN IN
NOSOCOMIAL PNEUMONIA
(EMPIRICAL)
Linezolid vs Vancomycin Chest 2003;124;1789-1797.
LINEZOLID VS VANCOMYCIN IN
NOSOCOMIAL PNEUMONIA
(EMPIRICAL)
Linezolid vs Vancomycin. Chest 2003;124:1789-1797.
LINEZOLID VS VANCOMYCIN IN
NOSOCOMIAL PNEUMONIA
(EMPIRICAL)
• Reason for improved survival: poor penetration of vancomycin into the lungs
• Mean concentration of vancomycin in lung tissue VS serum
• 1h: 9.6 mg/kg vs 40.6mg/L
• 12h: 2.8 mg/kg vs 6.7mg/L
• Mean concentration of linezolid in ELF vs plasma
• 4h: 64.3 ug/ml vs 7.3 ug/ml
• 23h: 24.3 ug/ml vs 7.6 ug/ml
Linezolid vs Vancomycin. Chest 2003;124:1789-1797.
THE RECOMMENDATION FOR USING LINEZOLID
IN MRSA PNEUMONIA
• Linezolid is an alternative to vancomycin, and unconfirmed, preliminary data suggest it may have an advantage for proven
VAP due to MRSA.
Am J Respir Crit Care Med 2005. 171(4): 388-416.
LIMITATION OF THE RETROSPECTIVE STUDY
• Post hoc analysis
• Subgroup analysis is not randomized.
Chest 2004:126(1):314-316.
• Chest 2004:125(6):2370-2371.
• Chest 2005:127(6):2298-2301.
LIMITATION OF THE PHARMACOKINETICS
STUDY
• Vancomycin level study: study in patients without pneumonia.
Cruciani M. J antimicrob chemother 1996:38(5): 865-869.
• Other study in pneumonia patients did not show sub-therapeutic lung concentration.
Lamer C. Antimicrob agents chemother 1993. 37(2): 281-286.
LATEST EVIDENCE FOR LINEZOLID
USE
META-ANALYSIS
META-ANALYSIS
Walkey AJ, et al.
Chest 2010
Kalil AC, et al.
Crit Care Med 2010
Beibei L, et al.
International journal of antimicrobial agents 2010
Target population Comparator Primary end-point
Suspected MRSA nosocomial pneumonia
Linezolid vs glycopeptide
Clinical success
Clinical cure Nosocomial pneumonia
Gram-positive bacterial infections
Linezolid vs glycopeptide
Linezolid vs
Vancomycin
Treatment success
LINEZOLID VS GLYCOPEPTIDE FOR THE
TREATMENT OF SUSPECTED MRSA
NOSOCOMIAL PNEUMONIA
Walkey AJ, et al. Chest. E-publish Sep 23, 2010
LINEZOLID VS GLYCOPEPTIDE FOR THE
TREATMENT OF
SUSPECTED MRSA
NOSOCOMIAL PNEUMONIA
Walkey AJ, et al. Chest. E-publish Sep 23, 2010
LINEZOLID VS GLYCOPEPTIDE FOR THE
TREATMENT OF
SUSPECTED MRSA
NOSOCOMIAL PNEUMONIA
Walkey AJ, et al. Chest. E-publish Sep 23, 2010
LINEZOLID VS GLYCOPEPTIDE FOR THE
TREATMENT OF
SUSPECTED MRSA
NOSOCOMIAL PNEUMONIA
Risk of thrombocytopenia : no significantly 2.97 times higher in linezolid group (95% CI: 0.81-10.94. P=0.10)
Risk of renal impairment: no significantly difference (RR:
1.09, 95% CI: 0.35-3.38, P=0.89)
Walkey AJ, et al. Chest. E-publish Sep 23, 2010
II. LINEZOLID VS VANCOMYCIN OR
TEICOPLANIN FOR NOSOCOMIAL
PNEUMONIA
Kalil AC, et al. Crit care Med 2010;38(9); 1802-1808.
II. LINEZOLID VS VANCOMYCIN OR
TEICOPLANIN FOR NOSOCOMIAL
PNEUMONIA
Clinical cure: RR 1.01 (0.93-1.10), P=0.83
Kalil AC, et al. Crit care Med 2010;38(9); 1802-1808.
II. LINEZOLID VS VANCOMYCIN OR
TEICOPLANIN FOR NOSOCOMIAL
PNEUMONIA
For MRSA pneumonia:
RR: 1.10 (0.83-1.38), P=0.44
Kalil AC, et al. Crit care Med 2010;38(9); 1802-1808.
II. LINEZOLID VS VANCOMYCIN OR
TEICOPLANIN FOR NOSOCOMIAL
PNEUMONIA
GI events Thrombocytopenia
Kalil AC, et al. Crit care Med 2010;38(9); 1802-1808.
Renal failure
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
International journal of antimicrobial agents. 2010;35: 3-12.
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
International journal of antimicrobial agents. 2010;35: 3-12.
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
Beibei L, et al.
International journal of antimicrobial agents.
2010;35: 3-12.
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
International journal of antimicrobial agents. 2010;35: 3-12.
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
International journal of antimicrobial agents. 2010;35: 3-12.
III.
LINEZOLID VS VANCOMYCIN FOR THE
TREATMENT OF GRAM-POSITIVE BACTERIAL
INFECTIONS
International journal of antimicrobial agents. 2010;35: 3-12.
META-ANALYSIS
Walkey AJ, et al.
Chest 2010
Kalil AC, et al.
Crit Care Med 2010
Beibei L, et al.
International journal of antimicrobial agents 2010
Target population Comparator Primary end-point
Suspected MRSA nosocomial pneumonia
Linezolid vs glycopeptide
Clinical success at the test of cure
(TOC) among clinically evaluable subjects
Clinical cure Nosocomial pneumonia
Gram-positive bacterial infections
Linezolid vs glycopeptide
Linezolid vs
Vancomycin
Treatment success
RANDOMIZED CONTROLLED STUDY
RANDOMIZED, DOUBLE BLINDED
TRIAL
• phase 4 study: nosocomial pneumonia due to proven MRSA
• compared the efficacy and safety of Zyvox with vancomycin
• Zyvox IV 600 mg every 12 hours or
• Vancomycin 15 mg/kg every 12 hours over the course of 7 to 14 days;
• vancomycin doses could be titrated at the investigator’s discretion based on creatinine clearance and vancomycin trough levels
48th Annual Meeting of the Infectious Diseases Society of America
RANDOMIZED, DOUBLE BLINDED
TRIAL
• 156 centers worldwide in 2004-2010
• randomized 1,225 patients
• 448 patients had proven MRSA nosocomial pneumonia (modified intent-to-treat group)
• 339 patients also met key protocol criteria at the end of study (per-protocol group) (> 5 days treatment)
RANDOMIZED, DOUBLE BLINDED
TRIAL
• Clinical success rates at the end of study (study 7-30 days post end of treatment) , perprotocol analysis
• 57.6 % (95/165) in Zyvox group
• 46.6 % (81/174) in Vancomycin group
• 95 % CI for the difference in response rates: 0.5%-21.6%
• p=0.042
RANDOMIZED, DOUBLE BLINDED TRIAL: SAFETY
• Intent-to-treat analysis: 1184 patients
Linezolid % diarrhea rash
3.7
2.7
constipation 1.0
nausea 1.0
vancomyci n diarrhea nausea rash anemia
%
Acute renal 1.4
failure
• No statistical significance in the risk of thrombocytopenia
4.3
1.9
1.7
1.4
RANDOMIZED CONTROLLED STUDY
SUMMARY
LINEZOLID IN MRSA INFECTION
• MRSA has high prevalence in nosocomial infection
• Lead to catastropic results in patients
• Fair treatment response to tranditional antimicrobials
• Increasing MIC of vancomycin and hetero-resistance
• Potential side effect while increasing trough
LINEZOLID IN MRSA INFECTION
• Retrospective study suggest survival benefit than vancomycin in MRSA pneumonia
2003;124;1789-1797.)
(Chest
• Meta-analysis in MRSA pneumonia: non-inferior than glycopeptide (2010)
• Latest randomized, double blinded trial suggest better (non-inferior) clinical success than vancomycin (2010 idsa abstract)