Plant Growth Regulators
advertisement

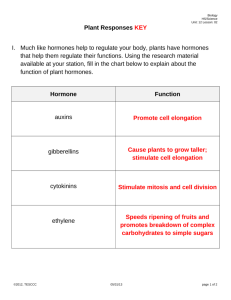
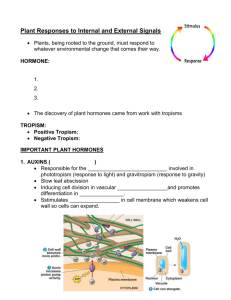
PLANT GROWTH REGULATORS Plant Growth Regulators AKA Plant Hormones Plant Growth Regulators - control growth, development and movement Plant growth hormones and regulators Hormones: Naturally occuring Regulators: Synthetic eg: 2,4-D (2,4Dichlorophenoxyacetic acid) and Kinetin Growth: It may be defined simply as an irreversible increase in mass due to the division and enlargement of cells, and may be applied to an organism as a whole or to any of its parts. Many plants, such as radishes and pumpkins, go through a sequence of growth stages. They grow rapidly at first, then for a while they show little, if any, increase in volume, and eventually, they stop growing completely. Finally, tissues break down, and the plant dies. Such growth is said to be determinate. Parts of other plants may exhibit indeterminate growth and continue to be active for several to many years. After cells have enlarged, differentiation occurs; that is, the cells develop different forms adapted to specific functions, such as conduction, support, or secretion of special substances The coordination of growth and differentiation of a single cell into multicellular tissues and organs is called development. What regulates development? What determines where and when growth and differentiation occur? The answers to these questions lie in the organism’s genes and its environment. In the 1950s, F. C. Steward and his colleagues at Cornell University illustrated this concept with a tissue culture experiment. They isolated cells from a carrot root and placed them in jars containing nutrients. The cells divided, grew, and developed into embryo-like structures. The embryos eventually grew into new carrot plants, demonstrating that a cell has all the genes necessary to form a new individual. Certain genes are programmed to synthesize specific enzymes continually in every living cell, while other genes produce enzymes only in specific tissues or at specific times during a plant’s life cycle. The environment determines which genes will be expressed. In an organ or an organism, the environment includes both external and internal factors. The internal environment includes nutrients, vitamins, and hormones. The external environment includes water, minerals, gases, light, and temperature, among many factors. Nutrients are substances that furnish the elements and energy for the organic molecules that are the building blocks by which an organism develops Vitamins play an important role in reactions catalyzed by enzymes: coenzymes or parts of coenzymes Genes also dictate the production of hormones, which influence many developmental phenomena. 1 produced mostly in actively growing regions of plants 2 They are organic substances that differ from enzymes in structure. 3 active in far smalleramounts than vitamins and enzymes. 4 Hormones ordinarily are transported from their point of origin to another part of the plant where they have specific effects, such as causing stems to bend, initiating flowering, or even inhibiting growth. 5 Because some of the effects of vitamins are similar to those of hormones, they are sometimes difficult to distinguish from one another. 6 The term growth regulator has been applied to both natural and synthetic compounds 7 hormones act by chemically binding to specific receptors. The hormone-receptor association triggers a series of biochemical events, including turning genes on and off. The biochemical events are called signal transduction PLANT GROWTH REGULATORS (PLANT HORMONES) Internal and external signals that regulate plant growth are mediated, at least in part, by plant growth-regulating substances, or hormones (from the Greek word hormaein, meaning "to excite"). Plant hormones differ from animal hormones in that: No evidence that the fundamental actions of plant and animal hormones are the same. Unlike animal hormones, plant hormones are not made in tissues specialized for hormone production. (e.g., sex hormones made in the gonads, human growth hormone pituitary gland) Unlike animal hormones, plant hormones do not have definite target areas (e.g., auxins can stimulate adventitious root development in a cut shoot, or shoot elongation or apical dominance, or differentiation of vascular tissue, etc.). PLANT GROWTH REGULATORS PLANT GROWTH REGULATORS ARE NECESSARY FOR, BUT DO NOT CONTROL, MANY ASPECTS OF PLANT GROWTH AND DEVELOPMENT. - BETTER NAME IS GROWTH REGULATOR. THE EFFECT ON PLANT PHYSIOLOGY IS DEPENDENT ON THE AMOUNT OF HORMONE PRESENT AND TISSUE SENSITIVITY TO THE PLANT GROWTH REGULATOR substances produced in small quantities by a plant, and then transported elsewhere for use have capacity to stimulate and/or inhibit physiological processes at least five major plant hormones or plant growth regulators: auxins, cytokinins, gibberellins, ethylene and abscisic acid General plant hormones Auxins (cell elongation) Gibberellins (cell elongation + cell division translated into growth) Cytokinins (cell division + inhibits senescence) Abscisic acid (abscission of leaves and fruits + dormancy induction of buds and seeds) Ethylene (promotes senescence, epinasty, and fruit ripening) EARLY EXPERIMENTS ON PHOTROPISM SHOWED THAT A STIMULUS (LIGHT) RELEASED CHEMICALS THAT INFLUENCED GROWTH Results on growth of coleoptiles of canary grass and oats suggested that the reception of light in the tip of the shoot stimulated a bending toward light source. Auxin Discovered as substance associated with phototropic response. Occurs in very low concentrations. Isolated from human urine, (40mg 33 gals-1) In coleoptiles (1g 20,000 tons-1) Differential response depending on dose. Auxins 1881 Charles Darwin and his son Francis development of Coleoptile: Metal foil blocked its growth towards light and the growth resumed after the removal of the foil. Later it was found to be a water soluble influence. 1926 Frits went: placed cut coleoptile tips on agar, cut the agar into blocks at that place and placed them onto cut tips of coleoptile. When placed in the center the coleoptile grew straight and it bent when placed off center Demonstration of transported chemical Auxin • • • Auxin increases the plasticity of plant cell walls and is involved in stem elongation. Arpad Paál (1919) - Asymmetrical placement of cut tips on coleoptiles resulted in a bending of the coleoptile away from the side onto which the tips were placed (response mimicked the response seen in phototropism). Frits Went (1926) determined auxin enhanced cell elongation. Auxins Auxins 1926 Frits went:His experiments showed that something moved out of the Coleoptile tip into the agar. He called these substances as auxins (Greek to increase) 1 First discovered hormones 2 At least three major groups apparently promote the growth of plants (auxins, gibberellins, and cytokinins). Depending on the concentration, some may also have an inhibitory effect. 3 Structure similar to tryptophan 4 Production of auxins occurs mainly in apical meristems,buds, young leaves, and other active young parts of plants. 5 It is sometimes difficult to predict how cells will respond to auxins because responses vary according to the concentration,location, and other factors. 6 At appropriate concentrations, auxins normally stimulate the enlargement of cells by increasing the plasticity (irreversible stretching) of cell walls. 7. Hormone concentrations can be measured by Gass chromatography Auxins Auxins also may have many other effects, including triggering the production of other hormones or growth regulators (especially ethylene), causing the dictyosomes to increase rates of secretion, playing a role in controlling some phases of respiration, and influencing many developmental aspects of growth. Auxins promote cell enlargement and stem growth, cell division in the cambium, initiation of roots, and differentiation of cell types. Auxins delay developmental processes such as fruit and leaf abscission, as well as fruit ripening, and inhibit lateral branching. Sensitivity to auxins is less in many monocots than it is in dicots, and shoots are less sensitive than roots. Auxin • Auxin promotes activity of the vascular cambium and vascular tissues. – plays key role in fruit development • Cell Elongation: Acid growth hypothesis – auxin works by causing responsive cells to actively transport hydrogen ions from the cytoplasm into the cell wall space Signal-transduction pathways in plants Auxin interacts with calcium ions which in turn calmodulin, a protein, which regulates many processes in plants, animals, and microbes. Loosening of cell wall Auxins Movement of auxins from the cells where they originate requires the expenditure of energy stored in ATP molecules. It is slow, polar i.e. from stem tip (source) toward the base even when the stem is inverted. Movement is from cell to cell of the parenchyma cells of the vascular bundles and not phloem sieve tubes Polar transport of Auxin Auxin • Synthetic auxins widely used in agriculture and horticulture prevent leaf abscission prevent fruit drop promote flowering and fruiting control weeds Agent Orange - 1:1 ratio of 2,4-D and 2,4,5T used to defoliate trees in Vietnam War. Dioxin usually contaminates 2,4,5-T, which is linked to miscarriages, birth defects,leukemia, and other types of cancer. Auxins indoleacetic acid (IAA) phenylacetic acid (PAA) 4-chloroindoleacetic acid (4-chloroIAA), is found in germinating seeds of legumes. indolebutyric acid (IBA), occurs in the leaves of corn and various dicots. Synthetic growth regulators presently in use include NAA (naphthalene acetic acid), 2,4-D (2,4dichlorophenoxyacetic acid), and MCPA (2-methyl-4-chlorophenoxyacetic acid). Additional responses to auxin abscission - loss of leaves flower initiation sex determination fruit development apical dominance Control of abscission by auxin Apical Dominance Lateral branch growth are inhibited near the shoot apex, but less so farther from the tip. Apical dominance is disrupted in some plants by removing the shoot tip, causing the plant to become bushy. Apical dominance is the suppression of the growth of the lateral buds (also called axillary buds), each of which can form a branch with its own terminal bud. Apical dominance is believed to be brought about by an auxinlike inhibitor in a terminal bud. It is strong in trees with conical shapes and little branching toward the top (e.g., many pines, spruces, firs) (Fig. 11.6) and weak in trees that branch more often (e.g., elms, ashes, willows). Senescence The breakdown of cell components and membranes that eventually leads to the death of the cell is called senescence.The leaves of deciduous trees and shrubs senesce and drop through a process of abscission every year. both ABA and ethylene promote senescence. On the other hand, auxins, gibberellins, and cytokinins delay senescence in a number of plants that have been studied. Other factors, such as nitrogen deficiency and drought, also hasten senescence. Orchardists spray fruit trees with auxins to promote uniform flowering and fruit set, and they later spray the fruit to prevent the formation of abscission layers and subsequent premature fruit drop. If auxins are applied to flowers before pollination occurs, seedless fruit can be formed and developed. Some orchardists use auxins for controlling the number of fruits that will mature in order not to have too many small ones, and some even control the shapes of plants for sales appeal. synthetic auxins, including 2,4-D, 2,4,5-T, 2,4,5-TP, NAA, and MCPA, when sprayed at low concentrations, kill some weeds. Gibberellin Gibberellins • Gibberellins are named after the fungus Gibberella fujikuroi which causes rice plants to grow abnormally tall. – synthesized in apical portions of stems and roots – important effects on stem elongation – in some cases, hastens seed germination Gibberellins 1926, Eiichi Kurosawa of Japan reported the discoveryof a new substance that was causing what was referred to as bakanae or “foolish seedling,” a serious disease of rice. Rice grains twice in size but weakened stems. Substances extracted and crystallized from this fungus (Gibberella fujikuroi) was , called as gibberellin. There are now more than 110 known gibberellins, customarily abbreviated to GA. Each form of GA is identified by a subscript (e.g., GA6). They have been isolated from immature seeds (especially those of dicots), root and shoot tips, young leaves, and fungi. Discovered in association with In 1930's, bakanae or foolish seedling disease of rice (Gibberella fujikuroi) • • • In 1930's, Ewiti Kurosawa and colleagues were studying plants suffering from bakanae, or "foolish seedling" disease in rice. Disease caused by fungus called, Gibberella fujikuroi, which was stimulating cell elongation and division. Compound secreted by fungus could cause bakanae disease in uninfected plants. Kurosawa named this compound gibberellin. – – – Gibberella fujikuroi also causes stalk rot in corn, sorghum and other plants. Secondary metabolites produced by the fungus include mycotoxins, like fumonisin, which when ingested by horses can cause equine leukoencephalomalacia - necrotic brain or crazy horse or hole in the head disease. Fumonisin is considered to be a carcinogen. Gibberellins No single plant species has thus far been found to have more than 15 kinds of GA, which probably also occur in algae, mosses, and ferns. Acetyl coenzyme A, which is vital to the process of respiration, functions as a precursor in the synthesis of GA. Interest in the hormonal properties of GA is high, for their ability to increase growth in plants is far more impressive than that of auxin alone, although traces of auxin apparently need to be present for GA to produce its maximum effects. e.g. monocots and dicots Gibberellins Gibberellins not only dramatically increase stem growth, but they are also involved in nearly all of the same regulatory processes in plant development as auxins. Flowering, the dormancy of buds and seeds can be broken, lower the threshold of growth; that is, plants may start growing at lower temperatures In some varieties of lettuce and cereals, the seeds normally germinate only after being subjected to a period of cold temperatures. An application of GA can eliminate the cold requirement for germination. It is common for seedlings to produce juvenile foliage at first and then different adult foliage as the plant matures. In such plants, GA treatment of buds that would normally develop into adult branches causes the foliage to develop in juvenile form. Effects of Gibberellins • Cell elongation. • GA induces cellular division and cellular elongation; auxin induces cellular elongation alone. • GA-stimulated elongation does not involve the cell wall acidification characteristic of auxin-induced elongation • Breaking of dormancy in buds and seeds. • Seed Germination - Especially in cereal grasses, like barley. Not necessarily as critical in dicot seeds. • Promotion of flowering. • Transport is non-polar, bidirectional producing general responses. Gibberellins Several growth retardants available commercially inhibit or block the synthesis of GA. Applications of growth retardants result in stunted plants because stem elongation is inhibited. When applied to certain commercially grown crops such as chrysanthemums, flowers with thicker, stronger stalks are produced. GA has been used experimentally to increase yields of sugar cane and hops In seedless grapes: by increasing the size of the fruit and lengthening fruit internodes, which results in slightly wider spaces between grapes in the bunches. Better air circulation between grapes reduces their susceptibility to fungal diseases,and the need for hand thinning is eliminated. Gibberellins: GA has been used in navel orange orchards to delay the aging of the fruit’s skin and in celery to increase the length and crispness of petioles (the parts that are most in demand as a raw food). GA is now used to increase seed production in conifers and to increase the rate of malting in breweries by enhancing starch digestion. Gibberellins and Fruit Size • Fruit Formation - "Thompson Seedless" grapes grown in California are treated with GA to increase size and decrease packing. Wild Radish – Rosette & Bolt A FLOWERING ANNUAL YEAR ONE YEAR ONE Common Mullen – Rosette & Bolt A FLOWERING BIENNIAL YEAR ONE YEAR TWO Mobilization of reserves Cytokinins Cytokinins: 1913, Gottlieb Haberlandt of Germany discovered an unidentified chemical in the phloem of various plants. This chemical stimulated cell division and initiated the production of cork cambium. In 1941, Johannes van Overbeek discovered that coconut “milk” (a liquid endosperm) contained something that increased thegrowth rate of tissues and embryos. In 1955, a substance named kinetin (isolated from heat killed sperms) was found, in the presence of auxin, to stimulate the proliferation of parenchyma cells in tobacco pith. It demonstrated that a simple chemical could promote cell Division. In 1964, the identity of a substance very similar to kinetin was also determined in kernels of corn. These various stimulants to cell division came to be known as cytokinins. Cytokinins: They are similar to adenine. Have been found only in RNA. Cytokinins are synthesized in root tips and germinating seeds. If auxin is present during the cell cycle, cytokinins promote cell division by speeding up the progression from the G phase to the mitosis phase, but no such effect takes place in the absence of auxin. Cytokinins also play a role in the enlarging of cells, the differentiation of tissues, the development of chloroplasts, the stimulation of cotyledon growth, the delay of aging in leaves, and in many of the growth phenomena also brought about by auxins and gibberellins. Cytokinins move throughout plants via the xylem, phloem, and parenchyma cells. 2 Cytokinins: Experiments have shown that cytokinins prolong the life of vegetables in storage. Related synthetic compounds have been used extensively to regulate the height of ornamental shrubs and to keep harvested lettuce and mushrooms fresh. They also have been used to shorten the straw length in wheat, so as to minimize the chances of the plants blowing over in the wind, and to lengthen the life of cut flowers. Many have not yet been approved for general agricultural use. Discovery of cytokinins • Gottlieb Haberlandt in 1913 reported an unknown compound that stimulated cellular division. • In the 1940s, Johannes van Overbeek, noted that plant embryos grew faster when they were supplied with coconut milk (liquid endosperm), which is rich in nucleic acids. • In the 1950s, Folke Skoog and Carlos Miller studying the influence of auxin on the growth of tobacco in tissue culture. When auxin was added to artificial medium, the cells enlarged but did not divide. Miller took herring-sperm DNA. Miller knew of Overbeek's work, and decided to add this to the culture medium, the tobacco cells started dividing. He repeated this experiment with fresh herring-sperm DNA, but the results were not repeated. Only old DNA seemed to work. Miller later discovered that adding the purine base of DNA (adenine) would cause the cells to divide. • Adenine or adenine-like compounds induce cell division in plant tissue culture. Miller, Skoog and their coworkers isolated the growth facto responsible for cellular division from a DNA preparation calling it kinetin which belongs to a class of compounds called cytokinins. • In 1964, the first naturally occurring cytokinin was isolated from corn called zeatin. Zeatin and zeatin riboside are found in coconut milk. All cytokinins (artificial or natural) are chemically similar to adenine. • • Cytokinins move nonpolarly in xylem, phloem, and parenchyma cells. Cytokinins are found in angiosperms, gymnosperms, mosses, and ferns. In angiosperms, cytokinins are produced in the roots, seeds, fruits, and young leaves Function of cytokinins Promotes cell division. Morphogenesis. Lateral bud development. Delay of senescence. Cytokinins • Cytokinins, in combination with auxin, stimulate cell division and differentiation. – most cytokinin produced in root apical meristems and transported throughout plant • inhibit formation of lateral roots – auxins promote their formation Cytokinins Interaction of cytokinin and auxin in tobacco callus (undifferentiated plant cells) tissue Organogenesis: Cytokinins and auxin affect organogenesis High cytokinin/auxin ratios favor the formation of shoots Low cytokinin/auxin ratios favor the formation of roots. Abscisic acid In 1940s, scientists started searching for hormones that would inhibit growth and development, what Hemberg called dormins. In the early 1960s, Philip Wareing confirmed that application of a dormin to a bud would induce dormancy. F.T. Addicott discovered that this substance stimulated abscission of cotton fruit. he named this substance abscisin. (Subsequent research showed that ethylene and not abscisin controls abscission). Abscisin is made from carotenoids and moves nonpolarly through plant tissue. Functions of abscisic acid General growth inhibitor. Causes stomatal closure. Produced in response to stress. Abscisic Acid • Abscisic acid is produced chiefly in mature green leaves and in fruits. – suppresses bud growth and promotes leaf senescence – also plays important role in controlling stomatal opening and closing Abscisic Acid: Torsten Hemberg, a Swedish botanist, showed that substances produced in dormant buds (i.e., buds whose development is temporarily arrested) blocked the effects of auxins. He called these growth inhibitors dormins. In 1963, three groups of investigators working independently in the United States, Great Britain, and New Zealand discovered a growth-inhibiting hormone, which was in 1967 officially called abscisic acid (ABA). Later, it was shown that ABA and dormins were the same compound. Abscisic Acid: ABA is synthesized in plastids, apparently from carotenoid pigments. Found in fleshy fruits. Inhibits/prevents seeds from germinating other hormones are inhibited by ABA ABA was originally believed to promote the formation of abscission layers in leaves and fruits. However, the evidence suggesting that ethylene is far more important than ABA in abscission Abscisic Acid: ABA apparently helps leaves respond to excessive waterloss. When the leaves wilt, ABA is produced in amounts several times greater than usual. This interferes with the transportor retention of potassium ions in the guard cells, causing the stomata to close. When the uptake of water again becomes sufficient for the leaf’s needs, the ABA breaks down, and the stomata reopen. ABA produced in times of drought is transported from the shoot to the root and causes an increase in both root growth and water uptake. Discovery of ethylene In the 1800s, it was recognized that street lights that burned gas, could cause neighboring plants to develop short, thick stems and cause the leaves to fall off. In 1901, Dimitry Neljubow identified that a byproduct of gas combustion was ethylene gas and that this gas could affect plant growth. In R. Gane showed that this same gas was naturally produced by plants and that it caused faster ripening of many fruits. Synthesis of ethylene is inhibited by carbon dioxide and requires oxygen. Ethylene H H \ / C = C / \ H H Ethylene: In 1901, Dimitry Neljubow, a Russian student at the St. Petersburg Botanical Institute, discovered that pea seedlings grown in the dark in a laboratory showed reduced stem elongation, increased swelling of stems, and abnormal horizontal growth. When the seedlings were placed outside in fresh air, however, they resumed normal growth due to gas ethylene, produced by gas lamps in the laboratory. In 1934, R. Gane discovered that ethylene was produced naturally by fruits, although it had been known for some time that the ripening of green fruits could be accelerated artificially by placing them in ethylene. It is now known that ethylene is produced not only by fruits, flowers, seeds, leaves, and even roots. The production of ethylene by plant tissues varies considerably under different conditions Ethylene: In 1901, Dimitry Neljubow, a Russian student at the St. Petersburg Botanical Institute, discovered that pea seedlings grown in the dark in a laboratory showed reduced stem elongation, increased swelling of stems, and abnormal horizontal growth. When the seedlings were placed outside in fresh air, however, they resumed normal growth due to gas ethylene, produced by gas lamps in the laboratory. In 1934, R. Gane discovered that ethylene was produced naturally by fruits, although it had been known for some time that the ripening of green fruits could be accelerated artificially by placing them in ethylene. It is now known that ethylene is produced not only by fruits, flowers, seeds, leaves, and even roots. The production of ethylene by plant tissues varies considerably under different conditions Ethylene: When a storm blows across a green field or animals pass across it, plants in the field respond to these and other mechan- ical stresses with an increase in the production of fibers and collenchyma tissue. Cell elongation is also inhibited, resulting in shorter, sturdier plants. The responses, called thigmomorphogenesis, are under the control of genes that are activated by touch. Several substances, including enzymes, ethylene, and a protein called calmodulin, are involved. Ethylene Today, the commercial use of ethylene is extensive. It has been used for many years to ripen harvested green fruits, such as bananas, mangoes, and honeydew melons, and to cause citrus fruits to color up before marketing. Since ethylene pro- duction almost ceases in the absence of oxygen, apple and other fruit growers have found that if they place unripe fruit in sealed warehouses after harvest, reduce temperatures to just above freezing, and replace the air with inert nitrogen gas or carbon dioxide, the fruit will remain metabolically inactive for long periods. The growers can then remove the fruit in batches throughout the year, add as little as one part per million of eth- ylene, and have ripe fruit at any time there is a demand. This is why apples are always available in supermarkets, even though harvesting is usually confined to a few weeks in the fall. A number of compounds called oligosaccharins, which are released from cell walls by enzymes, influence cell d ifferentiation, reproduction, and growth in plants and there- fore must be considered hormones. However, oligosaccharins are 1000 times less effective than other growth regulators Functions of ethylene Gaseous in form and rapidly diffusing. Gas produced by one plant will affect nearby plants. Fruit ripening. Epinasty – downward curvature of leaves. Encourages senescence and abscission. Initiation of stem elongation and bud development. Flowering - Ethylene inhibits flowering in most species, but promotes it in a few plants such as pineapple, bromeliads, and mango. Sex Expression - Cucumber buds treated with ethylene become carpellate (female) flowers, whereas those treated with gibberellins become staminate (male) flowers. HOW PLANTS RESPOND TO ENVIRONMENTAL STIMULI • Tropisms - plant growth toward or away from a stimulus such as light or gravity. • Nastic Movements - response to environmental stimuli that are independent of the direction of the stimulus. Pre-determined response. Tropic responses Directional movements by growth in response to a directional stimulus Phototropism Growth movement Phototropisms • Phototropic responses involve bending of growing stems toward light sources. – Individual leaves may also display phototrophic responses. • auxin most likely involved Plants Respond to Gravity • Gravitropism is the response of a plant to the earth’s gravitational field. – present at germination • auxins play primary role – Four steps • • • • gravity perceived by cell signal formed that perceives gravity signal transduced intra- and intercellularly differential cell elongation Gravitropism • Increased auxin concentration on the lower side in stems causes those cells to grow more than cells on the upper side. – stem bends up against the force of gravity • negative gravitropism • Upper side of roots oriented horizontally grow more rapidly than the lower side – roots ultimately grow downward • positive gravitropism Gravitropism = Geotropism Statoliths Plants Respond to Touch • Thigmotropism is directional growth response to contact with an object. – tendrils Thigmotropism SEISMONASTY - a nastic response resulting from contact or mechanical shaking Mimosa pudica L. (sensitive plant) Pulvinus of Mimosa pudica Plants Response to Light • • Photomorphogenesis – nondirectional, light-mediated changes in plant growth and development • red light changes the shape of phytochrome and can trigger photomorphogenesis • Stems go from etiolated (in dark or Pfr) to unetiolated (in light with Pr). Photoperiodism – Regulates when seeds of lettue and some weeds. Presence of Pr inhibits germination, while its conversion to Pfr in red light induces germination Red light ===> germination Far-red light ===> no germination Red ===> far-red ===> red ===> germination Red ===> far-red ===> red ===> far-red ===> no germination Those seeds not buried deep in the ground get exposed to red light, and this signals germination. – Regulates when plants flower; either in the Spring or later in the Summer and Fall. How Phytochrome Works NYCTINASTY • sleep movements • prayer plant - lower leaves during the day and raises leaves at night • shamrock (Oxalis) • legumes Credit:(http://employees.csbsju.edu/ssa upe/biol327/Lab/movie/movies.htm) Circadian Clocks • Circadian clocks are endogenous timekeepers that keep plant responses synchronized with the environment. – circadian rhythm characteristics • must continue to run in absence of external inputs • must be about 24 hours in duration • can be reset or entrained (to determine or modify the phase or period of <circadian rhythms entrained by a light cycle>) • can compensate for temperature differences