Ionic Compounds: Naming

Topic: Naming Ionic Compounds
Do Now: Write formulas for the following
1. Na and Cl Na +1 Cl -1 = NaCl
2. Mg and I Mg +2 I -1 = MgI
2
3. Al and S Al +3 S -2 = Al
2
S
3
Naming Binary Ionic Compounds there is a flow chart for you to use add to your reference table packet
1. Always name metal (the cation/+ ion) first
2. Leave a space
3. Write stem of nonmetal (the anion/ - ion) and Add ending “ide” to nonmetal
Binary = only 2 types of elements
(two capital letters)
Stems of nonmetals
Nitr
Phosph
Ox
Sulf
Arsen
Hydr is the stem for H
Selen
Tellur
Fluor
Chlor
Brom
Iod
Metals with one oxidation state
• CaO
• BaS
• AlN
Calcium Oxide
Barium Sulfide
Aluminum Nitride
• LiCl Lithium Chloride
• Al
2
Se
3
• Na
2
O
• K
3
N
• MgF
2
Aluminum Selenide
Sodium Oxide
Potassium Nitride
Magnesium Fluoride
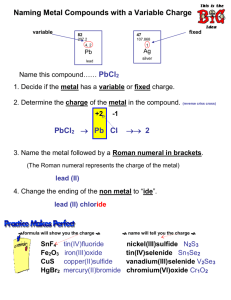
The First Step in Naming
• Find metal on PT
• If metal has only one oxidation state it’s easy
• If metal has more than one oxidation state, there’s an extra step
Metals with more than 1 oxidation state
• Use formula to figure out which oxidation state metal ion has
• Ex: Fe can be Fe +2 or Fe +3
» FeO vs Fe
2
O
3
– two different compounds so cannot both be called iron oxide
– so we have to use Roman Numeral to Tell which Fe (Fe +2 or Fe +3 ) we are using
– Iron (II) Oxide –uses Fe +2
– Irons (III) Oxide – use Fe +3
When deterring Oxidation State of Metal being used, assigned the nonmetal it’s oxidation number first (will be the top oxidation number from the PT)
FeO
• Compounds are electrically neutral
• Oxygen is -2
FeO
1 O which is -2 so Fe must be +2
Name: Iron (II) oxide but means Iron (+2) Oxide
(roman numeral II = charge on Fe is +2)
FeO and Fe
2
O
3
Each Fe is +3 Each O is -2
Fe
2
O
3
Total positive charge must be +6
There are 3 O’s
3 X (-2) = -6
Total negative charge
Name: Iron (III) oxide but means Iron (+3) Oxide
(roman numeral III = charge on Fe is +3)
Name the following
• TiCl
3
• MnO
2
• Co
2
O
3
• PdBr
2
Titanium (III) chloride
Manganese (IV) oxide
Cobalt (III) oxide
Palladium (II) bromide
• AuCl
3
• MoN
Gold (III) chloride
Molybdenum (III) nitride
• MnO
• TiO
Manganese (II) oxide
Titanium (II) oxide
Summary for Binary Ionic
Compounds
Compounds are electrically neutral
• Formula: positive first
• If metal has more than 1 oxidation state, name has roman numeral (MOST ALL
TRANSITION METALS need this)
• Name = metal + stem of nonmetal + ide
Some ionic compounds have a polyatomic ion – see table E
EXAMPLE
KOH - K = metal (positive ion) and OH = polyatomic ion (negative ion)
MgSO
4
– Mg = metal (positive ion) and SO
4
= polyatomic ion (negative ion)
Naming compounds with polyatomics
• polyatomic ions have names (Table E)
• naming is parallel to binary naming
• positive always written first
• if (+)’ve ion is a metal, check to see how many oxidation states it has
– if > 1 then name must have roman numeral
• if (–)’ve is polyatomic - 2 nd part of name is name of polyatomic (don’t modify ending)
Name the following
• NaOH Sodium hydroxide
• KHCO
3
• LiNO
3
Potassium hydrogen carbonate
Lithium nitrate
• CaSO
4
• Al(NO
3
)
3
• Fe(OH)
2
• CuSO
4
• CuSCN
Calcium sulfate
Aluminum nitrate
Iron (II) hydroxide
Copper (II) sulfate
Copper (I) thiocyanate