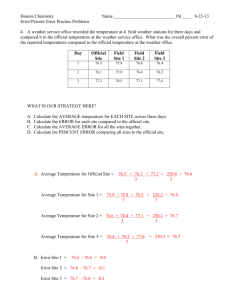
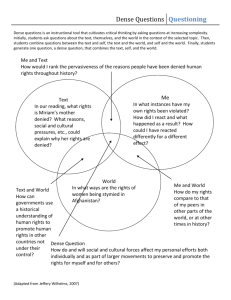
Unit 11: States of Matter
advertisement

Unit 1: Intro to Chemistry Phase Diagrams After today you will be able to… • Draw a phase diagram and label each state of matter, changes of state, and the triple point • Identify at what temperature or pressure a substance will change state • Identify which state is the most/least dense form and n.B.P and n.F.P. A phase diagram shows the conditions of temperature and pressure that are necessary for a substance to exist as a solid, liquid, or gas. Phase Diagram for Water Pressure (kPa) LIQUID 101.3 Triple Point GAS SOLID n.M.P.= 0°C n.B.P.=100°C Temperature (°C) Triple point: The conditions of temperature and pressure at which all three states of matter are in equilibrium with each other (S, L, G, exist simultaneously). The Triple Point of Water: Two Types of Phase Diagrams: 1. Liquid state is more dense than the solid state (Example: water – ice floats!) 2. Solid state is more dense than the liquid state (Example: most substances) • If the solid/liquid line slants to the left, the liquid state is more dense than the solid state (liquid region is larger). LIQUID SOLID GAS • If the solid/liquid line slants to the right, the solid state is denser than the liquid state (solid region is larger). LIQUID SOLID GAS