So far*.
advertisement

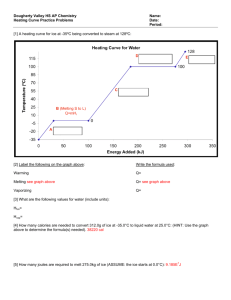
Which one feels hotter/colder? Based on feeling the 2 objects, it’s relatively easy to tell which one is colder and which one is hotter • That is, we can easily tell that there is a difference in temperature • Or is there??? Do they have the same temperature? can humans really feel temperature? So far…. • Transfer of energy qualitatively Energy is transferred into or out of a system via different “accounts” (Eth or Eph) • Now, we need to be able to distinguish how much energy is transferred when changes take place in a system • First, we need to start thinking about how different substances take in or release energy Have you ever wondered… • Why, on a hot summer day, the pool is cooler than the hot cement? • Why the ocean is cooler than the hot sand? • EVEN THOUGH both systems are being supplied the same amount of energy from the same energy source (the sun) • This phenomenon is just one of many examples that demonstrate certain substances absorb thermal energy at different rates What factors would determine the rate at which energy is taken in or released? • Heat flow (q) proportional to mass of sample (m) • Heat flow (q) proportional to temperature change (∆T) 𝑞∝𝑚 𝑞 ∝ ∆𝑇 𝑞 ∝ 𝑚 ∝ ∆𝑇 𝑡ℎ𝑒𝑟𝑒𝑓𝑜𝑟𝑒 𝑞 = 𝑚∆𝑇 • But what would this indicate? Any materials with same mass which is supplied w/ same amount of energy will absorb and release that energy at the same rate Our own experience tells us this just isn’t true (pool & cement example) Therefore, something must be missing from the equation Specific Heat (c) • • • • 𝑞 = 𝑚𝑐∆𝑇 Units of specific heat (c)? 𝑞 𝐽𝑜𝑢𝑙𝑒𝑠 (𝐽) 𝐽 𝑐= = = 𝑚∆𝑇 𝑔𝑟𝑎𝑚 𝑔 ∗ ℃ 𝑔 ∗ ℃ Specific heat is the amount of energy required to change the temperature of 1g of a substance by 1 0C It’s a measure of the relative ease w/ which a substance may be heated or cooled Physical property of each pure substance Liquid water has one of the highest specific heat values of any pure substance • What change would require the most amount of energy? • Solid to liquid • Liquid to gas Remember as time increases, energy increases Specific Heat • So what does it mean for a substance to have a high specific heat or low specific heat? Substance with high specific heat means it takes more energy to raise its temperature (water, vegetable oil) Substance with low specific heat means it takes less energy to raise its temperature (iron and many other metals) How do we calculate amount energy gained/released during phase change? • Can we still use our original equation? 𝑞 = 𝑚𝑐∆𝑇 • It wouldn’t make sense to use this equation during the plateau range on the heat curve because change in temp = 0 This would indicate that there is no energy being added/released for a system to change states of matter We know this isn’t true because even though the temp. doesn’t change, the energy is still being used for something in the system • So we must introduce similar equation with subtle differences if we are going to calculate the amount of energy gained/lost during phase change Heat of vaporization and heat of fusion • Heat of vaporization (Hvap): amount of energy required to convert a liquid into vapor without a change in temperature (can use for condensing as well) • Heat of fusion (Hfus): amount of energy required to melt one gram of that substance (can use for freezing as well) 𝑞 = 𝑚𝐻𝑣𝑎𝑝 𝑞 = 𝑚𝐻𝑓𝑢𝑠 Remember, earlier we said it would require more energy to change liquid water into gas than it would to change ice into liquid water. Do the following values for back that claim up? Hfus = 334 J/g Hvap = 2260 J/g Make sense when you consider pulling apart particles of liquid water until they’re widely separated in a gas is more difficult than simply giving solid water enough energy to allow the particles to move freely past one another Practice • How much energy is required to raise the temperature of 475 g of water for a pot of tea from 14 degrees C to 95 degrees C? Answer in both J and kJ Practice • A student observes a loss of 803 J as 141 g of aluminum cools from 31.7oC to 25.00C. Calculate the specific heat of aluminum from these data. .85 J/goC Practice • Calculate the heat flow when 155 g of sodium freezes. Express the answer in both J and kJ. Hfus for sodium = 113 J/g 17,515 J 17.5 kJ Practice • To what temperature will 545 g of cobalt be raised if, beginning at 25.0 oC, it gains 3140 J of energy? Specific heat of cobalt is .46 J/goC 37.5oC Practice • Calculate the energy released as 227 g of sodium vapor condense Hvap for sodium = 4.27 kJ/g 969 kJ Practice • If you are going to heat some water to boiling to prepare tea, will it take more time or less time if you start with hot water than if you start with cold, or will the times be the same?