Chemical Formulas and Chemical Compounds
advertisement

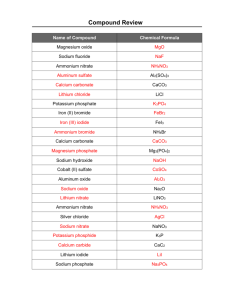
Chemical Formulas and Chemical Compounds Chemical Names and Formulas What’s in a name? What is the formula for: Epsom’s salt Magnesium Sulfate What’s in a name? Name the following compounds: 1. CCl4 2. MgCl2 Molecular and Ionic Compounds Molecular The formula gives the # of atoms of each element in a molecule. Ionic The formula is the simplest whole # ratio of ions Write formulas for the following compounds: Potassium sulfide Aluminum chloride Zinc bromide Copper (II) oxide Iron (III) sulfide Tin (IV) sulfide Copper (II) nitrate Sodium hydroxide Magnesium iodide Mercury (II) oxide Aluminum hydroxide Ammonium acetate Potassium permanganate Copper (II) chloride Strontium oxide Ammonium phosphate Calcium carbonate Calcium nitride Tin (II) chloride Sodium sulfate Naming Ionic Compounds Name = Cation name + Anion Name Monatomic cations Monatomic anions Mg2+ magnesium Cl- chloride Transition metal cations Fe3+ iron (III) Polyatomic cations NH4+ ammonium polyatomic anions SO42- sulfate Examples: 1. NaI Sodium iodide 2. CaO Calcium oxide 3. Al2O3 Aluminum oxide 4. Na3PO4 Sodium phosphate 5. CaSO4 Calcium sulfate Examples with transition metals 1. CuCl Copper (I) chloride 2. CuCl2 Copper (II) chloride 3. FeO Iron (II) oxide 4. Fe2O3 Iron (III) oxide Examples with polyatomic cations 1. NH4Cl Ammonium chloride 2. NH4OH Ammonium hydroxide 3. (NH4)2SO4 Ammonium sulfate Write the names of the following compounds: 1. Ag2S 2. NaMnO4 3. Ba(OH)2 4. NH4NO3 5. Fe(ClO)2 6. Ca(NO3)2 7. K2SO3 8. NaC2H3O2 silver sulfide sodium permanganate barium hydroxide ammonium nitrate iron (II) hypochlorite calcium nitrate potassium sulfite sodium acetate Write the names of the following compounds: 1. Rb2O 2. Ca(OH)2 3. CoCl3 4. SrI2 5. K2S 6. SnO2 7. NH4NO3 8. Cu(NO3)2 9. KClO3 10.Mo(SO4)3 Naming Binary Molecular (Covalent) Compounds • Binary – between 2 elements • Molecular compounds form between nonmetals only How is a molecular compound named? Numerical prefix + name of the first element + Numerical prefix + name of the second element with “ide” ending Example: N2O3 dinitrogen trioxide What are the numerical prefixes? 1 2 3 4 5 6 7 8 9 10 MonoDiTriTetraPentaHexaHepta Octa NonaDeca- Examples: N2O – dinitrogen monoxide P4O10- tetraphosphorus decoxide As2O5- diarsenic pentoxide Some Exceptions • “mono” is left off of the first element name if there is only one of those atoms CO2 CO Carbon dioxide _________________ Carbon monoxide _________________ Some Exceptions • Prefixes are sometimes shortened by dropping the “o” or “a” if the element name starts with a vowel N 2O Dinitrogen monoxide _____________ N 2O 4 Dinitrogen tetroxide _____________ Some Exceptions • Common names are used for some compounds H2O NH3 Water ______________ Ammonia ______________ Name the following compounds: SO2 Sulfur dioxide PCl3 Phosphorus trichloride CCl4 Carbon tetrachloride CS2 Carbon disulfide P4O6 Tetraphosphorus hexoxide PF5 Phosphorus pentafluoride XeF4 Xenon tetrafluoride SO3 Sulfur trioxide Write formulas for the following compounds. Nitrogen monoxide NO Oxygen difluoride OF2 Carbon tetraiodide CI4 Dinitrogen pentoxide N2O5 Dinitrogen tetroxide N2O4 Sulfur hexafluoride SF6 Naming Acids • Acids were first identified as substances that break up into hydrogen ions and anions when dissolved in water Example: HCl H2O H+ + ClBinary Acids – acids containing monatomic anions Name = Hydro + “element name” + “ic” acid HF Hydrofluoric acid HCl Hydrochloric acid Hydrobromic acid HBr Hydroiodic acid HI Acids with oxy-anions • Acids with anions containing oxygen and ending in “ate” Name = “anion name” + “ic” acid HNO3 Nitric acid H2SO4 Sulfuric acid H2CO3 Carbonic acid H3PO4 Phosphoric acid Acids with Oxy-anions • Acids with anions containing oxygen and ending in “ite” Acid name = anion name + “ous” + “acid” H2SO3 Sulfurous acid HNO2 Nitrous acid Covalent Network Compounds