Ethanol Combustion
advertisement
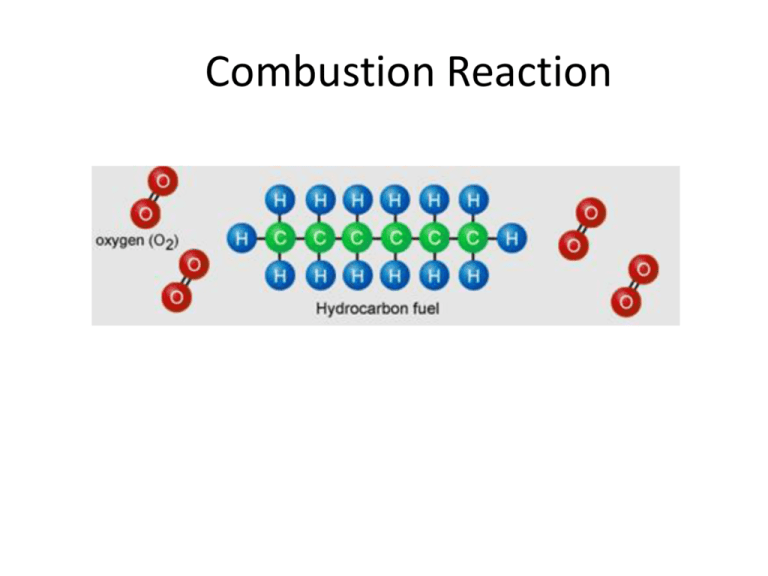
Combustion Reaction Heat of Reaction (q) C6H14(l) + O2 (g) Energy required to break bonds CO2 (g) + H2O (g) Energy released as bonds form Heat of Reaction (q) • Reactants use energy to break their bonds – C-H (413 kJ per bond in hexane) – O=O (498 kJ per bond in oxygen gas) • Products release energy as bonds reform – – C=O (803 kJ per bond in carbon dioxide) O-H (463 kJ per bond in water) • Once the accounting is done, there will be a net difference in energy (q). • Combustion reactions always produce MORE energy than they use up EXOTHERMIC Measuring Heat of Combustion (q) • Calorimetric experiments are performed to indirectly measure the amount of heat released by a fuel • A known quantity of water will be heated by the combustion of a fuel • It is assumed: Heat absorbed = heat released by by the water combustion of fuel q = mCΔT • q is the amount of heat energy (in Joules) absorbed by the water. • Therefore, all values (m, C, T) are related to water m=mass of water (grams) C=heat capacity of water (constant: 4.18 J/Kg) ΔT=temperature rise of water (Tf–TI) *q is always be (+) for combustion reactions Calorimetry • A known quantity of water must be measured and weighed • The exact amount of fuel used to heat the water must be determined. This is done by accurately weighing the mass of the spirit burner before and after the water is heated. Keep the cover on the burner to prevent any loss in mass by evaporation. • Measure the starting and final temperature of the water. • Calculate q (heat of reaction). Calculate q Eg. m = mass of water = 150.0 g C = heat capacity of water = 4.18 JK-1g-1 ΔT = temperature rise = 18.0o q = 150.0 x 4.18 x 18.0 = 11,286 Joules = 11.3 KJ (heat absorbed by water) ΔH (enthalpy) and q (heat) q = heat energy absorbed by water ΔH = heat energy released by the reaction So, ΔH = -q (opposite signs) If q = 11.3kJ, then ΔH = -11.3 kJ *ΔH will always be (-) for an exothermic reaction Calculate ΔH per gram q = 11.3 kJ Recall that the spirit burner used 1.5 grams of fuel Recall that ΔH= -q Therefore, ΔH = -11.3 kJ ΔH= -11.3 kJ = - 7.6 kJ of energy released per 1.5 grams gram of hexane burned Calculate ΔH per mole • Molar heat of combustion tells us how much the water was heated by for every mole of hexane used. • The spirit burner needs to be weighed before/after to determine the amount used (grams) • mass will need to be converted into moles ΔH=q/mol Calculate molar heat of combustion ΔH = -11.34 kJ mol = 1.5 g x Recall that hexane has a molar mass of 86 g/mol 1 mol = 0.0174 mol 86.0 g ΔH = q = -11.34 kJ = - 650 kJ/mol mol 0.0174 mol Comparing ΔH values • q values will vastly differ as it depends on the amount of water being heated and the temperature rise of the water; q values cannot be compared or published • Molar heat of combustion values (ΔH) are widely known and published for every type of fuel; ΔH can be compared








