CH105.2 Part 1: Problem Solving in Organic Chemistry
advertisement

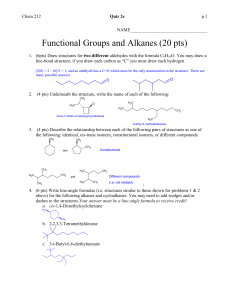
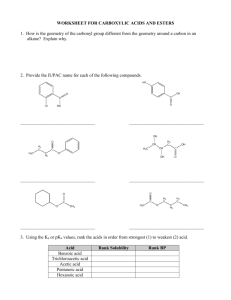
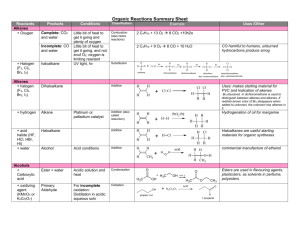
CH105.2 Part 1: Problem Solving in Organic Chemistry Carbon Intermediate Types Before anything else… • Remember that organic reactions, no matter how difficult they may seem, simply involve the reaction between positive/partial positive and negative/partial negative sites. • After determining these sites, threedimensional steric considerations come into play. Ok? One way of looking at reactions… What type of intermediate is formed? • • • • Carbocation Carbanion Radical Special Intermediate: Benzyne CARBOCATIONS in reactions… • Combine with nucleophiles (positive + negative or partial negative) • Lose proton (elimination) • Rearrangement (more stable carbocation) • General observation: mainly (but NOT exclusively) involves alkenes Carbocations in reactions… SN1 and E1 • Key step in both: formation of carbocation neutral to slightly acidic conditions CH3 H3C CH3 H3C CH3 Br H3C CH3 X CH3 + C CH3 CH2 basic conditions • SN1: combine with nucleophile • E1: lose proton H3C CH3 Carbocations in reactions… Addition to Alkenes • Straightforward carbocation formation: hydrogen halide addition H3C H-X CH2 H3C + X- CH H X H3C H Carbocations in reactions… Addition to Alkenes • Variations of carbocation formation: cationic 3-membered ring intermediate H3C A CH2 H3C + A CH A H3C • The more substituted carbon can then be attacked by a nucleophile to form the addition product – X2 addition; halohydrin formation; oxymercuration + Carbocations in reactions… Addition to Alkenes • Variations of carbocation formation: 4membered ring transition state (Henson, pakidrawing nalang, hindi ko alam paano sa chemsketch eh. Haha) • HYDROBORATION Carbocations in reactions… • Acid catalyzed dehydration of alcohols OH H3C + H+ OH2 -H2O H3C CH3 H3C + CH CH3 CH3 H2C CH3 • Electrophilic aromatic substitution E+ C + H E elimination E Carbocations in reactions… • Hydrolysis of tert-butyl esters (vs. usual mechanism) O H C 3 H3C O CH3 + H O H C 3 + H H3C CH3 O O CH3 H3C CH3 CH3 OH + H3C + C CH3 • Diazotization HONO H3C NH2 H3C -N2 + N N H3C – Carbocation may undergo each of the 3 processes + CH2 Carbocations in reactions… • Pinacol rearrangement H3C CH3 H3C CH3 HO H3C H+ H3C OH pinacol CH3 H2O H3C CH3 + CH3 + C OH H3C CH3 OH H3C H3C CH3 H3C H3C -H2O O H3C H3C H3C CH3 H3C + OH pinacolone • Acid-catalyzed polymerization of alkenes H3C + C OH CH3 CARBANIONS in reactions… • Acts as base (abstracts proton) • Acts as nucleophile (reacts with electrophiles) • General observation: involves – Substitution with carbon nucleophiles – Organometallic reagents – Carbonyl chemistry Carbanions in reactions… • SN2 with CN- as nucleophile • Nucleophilic aromatic substitution – Carbanion intermediate formed: Meisenheimer complex • Grignard reagent – + H2O = alkane – + aldehyde, ketone, ester = alcohol – + RCΞN = ketone Carbanions in reactions… • Gilman reagent (diorganocopper lithium) R2CuLi + R’X → R-R’ • 1,2 and 1,4 addition to α,β-unsaturated carbonyls: governed by HARD-HARD/SOFTSOFT chemistry – Carbonyl, C=O: hard (direct 1,2-addition by hard :Nu) e.g. Grignard reagent – Alkene, C=C: soft (conjugate 1,4-addition by soft :Nu) e.g. Gilman reagent Carbanions in reactions… • Carbonyl α-substitution reactions – Usually by aldehydes, ketones, and esters, especially β-DIKETONES and β-KETO ESTERS – Common reactions: • Halogenation of α-position – Monohalogenation in acidic medium – Multiple halogenation in basic medium (excess halogen: Haloform reaction occurs) – Hell-Volhard-Zelinski Reaction (Br2, PBr3) » If 2nd step is H2O = α-brominated carboxylic acid » If 2nd step is ROH = α-brominated ester » If 2nd step is excess NH3 = α-amino acid Carbanions in reactions… • Carbonyl α-substitution reactions – Common reactions: • Alkylation of α-position – Direct alkylation (SN2 with CH3I) – Malonic and acetoacetic ester synthesis RADICALS in reactions… • Combine with another free radical Br• + Br• → Br–Br • Abstract an atom from a molecule RO• + H–Br → ROH + Br• • Add to an unsaturated molecule Br + H2C CH2 CH2 • Decompose into 2 fragments R–COO• → R• + CO2 Br Radicals in reactions… • Indicators of a radical reaction: – Radical initiators: benzoyl peroxide or AIBN – Heat or light may also initiate radical formation