Enzyme Lab - WordPress.com
advertisement

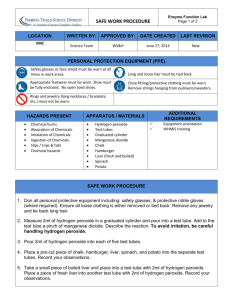
Enzymes: A Study of Biochemical Reactions A chemical reaction occurs when chemicals come together (the reactants) and their molecules interact by the breaking or formation of bonds. The results are new chemicals (the products). Sometimes chemical reactions happen by themselves. These reactions are usually very fast and spontaneous and give off energy. Other chemical reactions need energy to happen, and without energy proceed v e r y s l o w l y or not at all. These types of chemical reactions can be helped to occur more quickly by using enzymes. Enzymes are made out of protein (amino acid chains!) and they speed up the rate of a chemical reaction by acting as a catalyst. A catalyst provides the necessary environment for the chemical reaction to occur, which speeds up the reaction. Certain catalysts work for certain kinds of reactions. In other words, each enzyme has a particular type of reaction that it can activate. Enzymes, as we just said, are proteins, which are molecules that are very large and dynamic. They can be very fussy, and sometimes need to be in certain environments or conditions to work, and the ideal conditions are usually reflective of where the enzyme must normally function in the body. In other words, different tissues are home to different enzymes, and an enzyme should be able to function in the conditions that surround it in the tissue it's supposed to function in. Some enzymes can be damaged under certain conditions, such as heat. A damaged enzyme will no longer work to catalyze a chemical reaction. (NOTE! This is a huge hint for later in the lab!) One source of enzymes is the liver, which needs to break down many substances in the body. Catalase is one enzyme from liver that breaks down harmful hydrogen peroxide into oxygen gas and water. When this chemical reaction occurs, you can see the oxygen gas bubbles escaping and causing the reaction to foam. In our cells there are many reactions occurring. One by-product from these reactions is hydrogen peroxide. Hydrogen peroxide can be toxic to our bodies if a sufficient amount is built up, so our cells contain an enzyme, catalase, which helps break down hydrogen peroxide into nonharmful substances. The reaction that takes place is shown below: 2H2O2 (Hydrogen Peroxide) ---> 2H2O (water) + O2 (Oxygen) 2 hydrogen peroxide molecules react to form 2 water molecules and 1 oxygen molecule. This reaction is V E R Y S L O W without an enzyme. Since enzymes remain unchanged in a reaction, catalase is able to catalyze a new reaction over and over until all of the hydrogen peroxide is broken down. The cells we are extracting the enzymes from are chicken liver cells because they contain large amounts of catalase, since the function of the liver is to detoxify and purify substances in our bodies. Purpose: To compare the action of catalase to a non-protein catalyst under different conditions. Materials: manganese dioxide stirring rod potato test tubes 250 mL beaker scalpel fresh or frozen liver hydrogen peroxide sand hot water bath ice scale graduated cylinder Procedure with Analysis Questions (record answers to questions in your logbook) A. Catalytic reactions Add 2 mL of hydrogen peroxide to two test tubes. Place 0.1 g of sand in one test tube and add 0.1 g of manganese dioxide to the second tube. Observe and record the rates of reaction. The manganese dioxide and hydrogen peroxide reaction is a decomposition reaction - the bonds within the H2O2 are broken. As you already know, the MnO2 is a catalyst, so it is not included in the balanced equation: 2H2O2 → 2H2O+O2 A1) What gas do you think evolves? A2) What role do you think the sand and H2O2 test tube played in this procedure? B. The effect of an enzyme Add 2 mL of hydrogen peroxide to each of two clean test tubes. In one place a small piece of liver and in the other a small piece of potato. Record the rates of reaction and compare these results to those with manganese dioxide. Do not discard these materials. C. Re-using an enzyme Divide the liquid portion of the previous tube in B containing the liver into two test tubes. Cut the liver from procedure B into two equal portions and add these to the two test tubes. To the first add a fresh piece of liver and to the second add 1 mL of hydrogen peroxide. Record your observations. C1) What could explain the reaction in the test tube containing the fresh liver? C2) What do you think would happen if additional hydrogen peroxide were added to the second tube? D. Effect of particle size Place a small piece of liver in one test tube and a small piece of potato in a second. Add a pinch of sand to each tube and crush these materials with separate stirring rods. Add 2 mL of hydrogen peroxide to each tube, then observe and record the rates of these reactions. D1) How do these results compare with those of the uncrushed liver and potato in part B? E. Effect of temperature Place a small piece of liver into each of two test tubes. Place one test tube in a 37'C water bath for 5 minutes and the second test tube in an ice-water bath for the same length of time. Remove both test tubes from the water baths and add 2 mL of hydrogen peroxide to each tube. Record the rates of the reactions. Observations: Record your results in a chart similar to the one shown below. Rates of reaction can be designated as follows: 0 = no reaction, + = slow, ++ = moderate, +++ = fast. 0 = no reaction, + = slow, ++ = moderate, +++ = fast. Mixture Part A H2O2 + Sand H2O2 +MnO2 Part B H2O2 + Liver H2O2 + Potato Part C Re-use + Fresh Liver Re-use + H2O2 Part D H2O2 + Crushed Liver H2O2 + Crushed Potato Part E H2O2 + Liver + Hot Water Bath H2O2 + Liver + Cold Bath Questions: answer in complete sentences in your logbook 1. 2. 3. 4. 5. What is catalase and what does it help our bodies do? Why do you think we chose liver to extract the catalase from? What did the Hydrogen Peroxide represent in this lab? Were there any results that surprised you? With the various temperatures (cold, room temp, body temp, hot), which one had the greatest activity? Propose a reason why this would be. 6. For two situations where there was no reaction (or very little), propose reasons for these results. Think about how an enzyme’s shape plays a role in its ability to function. 7. Using your results, describe the effect of temperature and particle size on the rate of enzyme action.