Metabolism Photosynthesis Cellular Respiration
advertisement

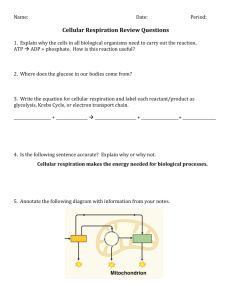
Cellular Respiration Chapters 8-10 Warm Up Exercise • Explain the difference between competitive and noncompetitive inhibitors • Describe the negative feedback demonstrated by ATP/ADP. Cellular Respiration Cellular Respiration • Cell respiration is a catabolic pathway. • Aerobic Cellular Respiration • Anaerobic Cellular Respiration (aka: Fermentation) Cellular Respiration • Organic compounds possess potential energy as a result of their arrangement of atoms • Fermentation – partial degradation of sugars that occur without Oxygen • Aerobic Respiration – most prevalent and efficient catabolic pathway (Oxygen is consumed) Cellular Respiration • Most eukaryotic and many prokaryotic can carry out aerobic respiration • Some prokaryotes can use substances other than oxygen as reactants – anaerobic respiration Cellular Respiration • Cellular Respiration uses both aerobic and anaerobic processes • However, usually refers to aerobic processes Cellular Respiration C6H12O6 +6O2 -> 6 CO2 + 6 H2O+ Energy (ATP +Heat) Cellular Respiration • Catabolic processes do not directly do the work; instead, catabolism is linked to work by chemical drive shaft – ATP • Require the regeneration of ATP… This is where Cellular Respiration comes into play Redox Reactions • How do the catabolic pathways that decompose glucose yield energy??? Redox Reactions • How do the catabolic pathways that decompose glucose yield energy??? • Due to the transfer of electrons during the chemical reaction. • Relocation of electrons releases energy stored in organic molecules and this energy is used to synthesize ATP Redox Reactions • Reduction vs. Oxidation • Why are carbs and fats the best molecules for energy? • Why must glucose be broken down in a series of steps rather than one quick reaction? Redox Reactions • Generally, there exists a transfer of one or more electrons from one reactant to another • This transfer leads to what is called an oxidation – reduction reaction (Redox) Redox Reactions • Oxidation – loss of electrons from one substance • Reduction – addition of electrons to another substance • Adding electrons • Negatively charged electrons added to atom reduce amount of positive charge of that atom Redox Reactions • Reducing Agent – Electron donor • Oxidizing agent – Electron acceptor • Transfer is ONLY between the Reactants in a chemical equation • Is this an ionic or covalent bond? REDOX Reaction Electron Transport • Dehydrogenase- removes electrons from glucose (or other substrate) transferring them to its coenzyme (NAD+) which is reduced to NADH. (NADH = potential energy) • NAD+ (nicotinamide adenine dinucleotide)- an electron carrier. • Cycles between NAD+ and NADH NAD to NADH NAD to NADH • NAD + is one of the most versatile electron acceptors in cellular respiration and functions in several redox steps during the breakdown of glucose • This is due to the fact that electrons lose very little of their potential energy when they are transferred from glucose to NAD+ • NADH represents stored energy that can be used to make ATP Electron Transport • As glucose is broken down (in many small reactions) electrons are shuttled (by NADH) down the Electron Transport Chain (ETC). • ETC is used to break the fall of electrons to Oxygen into several energy-releasing steps • ETC has many molecules (mostly proteins) built into the inner membrane of mitochondria of eukaryotic cells and the plasma membrane of aerobically respiring prokaryotes • Ultimately, oxygen is the final electron acceptor. Electron Transport • Electrons removed from glucose are shuttled by NADH to the “top”, higherenergy end of the chain. • At the “bottom”, lower-energy end of the chain O awaits • Ultimately, oxygen is the final electron acceptor. Electron Transport • Electron transfer from NADH to O is exergonic with free-energy change of -53 kcal / mol. • Instead of one with one big step and a greater loss of energy, electrons cascade down the chain from one carrier molecules to the next in series of redox reactions • Lose a small amount of energy with each step Electron Transport • Each downhill carrier is more electronegative • Is it oxidizing or reducing it’s “uphill” neighbor? Electron Transport • Each downhill carrier is more electronegative • Is it oxidizing or reducing it’s “uphill” neighbor? • Oxidizing it’s uphill neighbor • Overall process: • Glucose-> NADH -> ETC -> Oxygen Warm Up Exercise • What is the function of NAD+? • Explain the terms oxidation and reduction. • What is the difference between aerobic and anaerobic respiration? What the Stages of Respiration? • On a piece of paper, list the stages of cellular respiration and list where each stage occurs. When you are finished turn it into the tray. Stages of Respiration • Glycolysis (in cytoplasm)- can occur with or without oxygen. • Pyruvate Oxidation (in mitochondria) • Citric Acid Cycle (in mitochondria) • Oxidative Phosphorylation: Electron Transport Chain and Chemiosmosis (in the outer membrane of the mitochondria) Stages of Respiration • asdklf Stages of Respiration • Glycolysis (in cytoplasm)- catabolic • Citric Acid Cycle (catabolic) • Oxidative Phosphorylation: Electron Transport Chain and Chemiosmosis (in the outer membrane of the mitochondria) Stages of Respiration • Glycolysis (in cytoplasm)- begins process by breaking down glucose into two molecules of pyruvate • Citric Acid Cycle in mitochondrial matrix – completes breakdown of glucose by oxidizing a derivative of pyruvate to carbon dioxide • Oxidative Phosphorylation: Electron Transport Chain and Chemiosmosis powered by the redox reactions of the ETC ADP to ATP • Oxidative Phosphorylation- inorganic phosphate is added to ADP to produce ATP. • Occurs in ETC and chemiosmosis. • Accounts for 90% of the ATP generated by respiration • Substrate-Level Phosphorylation- an enzyme transfers a phosphate group from a substrate molecule to ADP to form ATP. • Occurs in glycolysis and citric acid cycle. • Substrate = an organic molecule generated as an intermediate in glycolysis. Glycolysis • Sugar –splitting • 6C sugar is split into two 3C sugars • Can be divided into 2 phases: energy investment and energy payoff • Energy investment – cell actually spends payoff • Payoff – substrate- level phosphorylation produces ATP and reduction of NAD • Net Yield: • 2 ATP • 2 NADH • kjh Warm Up Exercise • Without using your notes, name the four major processes of cellular respiration and where in the cell they occur. • Explain the difference between oxidative and substratelevel phosphorylation. Oxidative Phosphorylation • Pyruvate enters mitochondria (via active transport) and is converted to Acetyl CoA Citric Acid/ Kreb’s Cycle Citric Acid/Kreb’s Cycle • Acetyl CoA (from oxidative phosphorylation) enters the Citric Acid cycle and combines with oxaloacetate to form citrate, the ionized form of citric acid. Warm Up Exercise • Walk through the Kreb’s cycle, stating the reactants and the products and where they came from, or go to ETC • Cytochromeselectron carriers in ETC. They are proteins with a Heme group attached. • Represent a series of redox reactions. Chemiosmosis • Chemiosmosis- energy coupling mechanism that uses H+ gradient to drive cellular work. • ATP Synthase- enzyme that makes ATP from ADP in the inner membrane of mitochondria. Chemiosmosis • Proton Motive Force- the H+ gradient that results from the pumping of H+ ions from the matrix of the mitochondria to the intermembrane space. • kjh Energy Totals • Kh; Warm Up -Cell Resp Challenge• Why does NADH have more energy than FADH2? • Explain the idea of energy coupling that occurs in chemiosmosis. • What element (atom) helps to pull electrons down the ETC? • How many total ATPs are produced per molecule of glucose in aerobic respiration? Alternatives to Aerobic Respiration • Anaerobic Respiration- uses ETC with a different final electron receptor (besides oxygen) • Fermentation- no ETC. Glycolysis followed by a fermentation process. • Two main types: Alcoholic and Lactic Acid Fermentation • Alcoholic Fermentation- pyruvate is converted to acetaldehyde then to ethanol (ethyl alcohol). CO2 byproduct. Fermentation • Lactic Acid Fermentation- pyruvate is reduced by NADH to form lactate, with no release of CO2. Aerobic vs. Anaerobic • Obligate Anaerobesorganisms that cannot survive in the presence of oxygen. • Carry out only fermentation or anaerobic respiration. • Facultative Anaerobesorganisms that can survive using fermentation or respiration.