Beer's Law Lab
advertisement

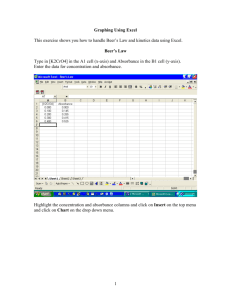
Beer’s Law Lab Using the absorbance of light to measure concentration of a solution Calorimeter Absorbance and Concentration • This lab is based on the principle that the “darker” a solution, the more concentrated it is •“darker” solution contains more solutes more solutes absorb more light less light will be transmitted through it •Concentration of solution can be measured QUANTITATIVELY by measuring how much light is absorbed Creating a calibration curve • First, calibrate your colorimeter using solutions with KNOWN concentrations (standard solutions) • You will calculate the concentrations of your standard solutions beforehand (using the dilution equation) and record their absorbances Finding the concentration of your unknown • Once you have the absorbances of your standard solutions, you can measure the absorbance of your unknown • Using the graph/equation of the trendline, you can obtain the concentration of your unknown Beer’s law graph -Chart title goes hereCalori M. and R. Cuvettes 0.6 0.5 Absorbance y = 1.2109x 0.4 0.3 0.2 0.1 0 0 0.05 0.1 0.15 0.2 0.25 Concentration (mol/L) 0.3 0.35 0.4 0.45 Example • If your unknown is measured to have an absorbance of 0.250, what is its concentration? • Use the graph • Use the equation: y = 1.2109 x 0.250 = 1.2109 x (0.250) / (1.2109) = x 0.206 mol/L = x (linear relationship) Expectations • You will be evaluated on the following: 1. A pre-lab quiz testing your knowledge of the theory & procedure (only 8 MC) 2. Your dilution calculations for your standard solutions 3. Your graph 4. The accuracy of your unknown concentration