Thomson, Plum Pudding Model, & Rutherford notes
advertisement

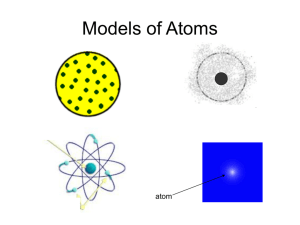
Thomson, Plum Pudding Model, & Rutherford notes Thomson’s Experiment J.J. Thomson used a cathode ray tube to show that the atoms of any element can be made to emit tiny negative particles. He knew the particles were negative because they were repelled by the negative part of an electric field. He concluded that all types of atoms must contain these negative particles. These particles are now called ELECTRONS. Rutherford’s Ernest Rutherford, a student of J.J. Thomson, designed the gold foil experiment. The gold foil experiment design directed alpha particles at a piece of gold foil. The gold foil was surrounded by a screen that made tiny flashes when it was hit by an alpha particle. Most of the alpha particles passed through the gold foil, but some of the alpha particles were deflected at an angle. The Plum Pudding Model J.J. Thomson and Lord Kelvin proposed that the atom might be something like a plum pudding (a pudding with raisins randomly distributed throughout). They reasoned that the atom was a positively charged with negative electrons scattered throughout to counterbalance the positive charge. Experiment His conclusion was that the Plum Pudding Model could not be correct. That the large deflections of alpha particles could only be caused by a center of concentrated positive charge that would repel the positively charged alpha particles. Most of the particles passed through the foil directly because the atom is mostly open space. We now call this concentrated center of positive charge the NUCLEUS and the electrons travel around the nucleus in mostly empty space.