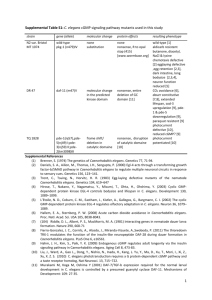
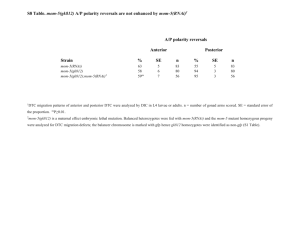
C. elegans
advertisement

C. elegans as a Model System • C. elegans: introduction (anatomy, life cycle, genome and etc.) • Essentially invariant lineage of C. elegans. • Cell death as a cell fate. • Genes that affect lineage specification and timing: link to miRNAs. • RNAi discovery. • miRNAs and RNAi. C. elegans timeline. • • • • • • • • • Developed by Sydney Brenner (1963) Mutants published by Brenner (1974) 1976 postembryonic cell lineages determined (Sulston and Horwitz) 1982 "Programmed cell death" (Horwitz et al.) (Nobel Prize Brenner, Sulston, Horwitz 2002) 1983 complete embryonic cell lineages determined (Deppe et al., Sulston et al.) 1986 Complete connectivity of nervous system established (White et al.) "The mind of a worm” 1991-98 RNAi and miRNA discovered in worms. (Nobel Prize: Fire, Mello 2006) 1994 - First use of GFP in animals (Novel Prize: Chalfie, 2008) 1998 First animal genome sequenced (97Mb, now 100.3Mb) This is about 1/30 the size of the human genome (3 Gb). C. elegans have about 20,000 coding sequence genes, more than half that of humans (30,000-40,000 genes). First animal to be sequenced! Knowing the sequence allows genes of interest to be easily cloned. Also it opened opportunity for reverse genetic approach. C. (Caenorhabditus) elegans 1st introduced and used by Sydney Brenner (1963) to study development and neurology. • Adults are ~1mm long (small). • They can be grown on agar plates with lawn of bacteria. • They have a short generation time- 3 days from egg-laying to adulthood, brood size > 300. • They are transparent, so internal anatomy can be easily observed. Goldstein lab http://www.bio.unc.edu/faculty/goldstein/lab/crawl.mov Caenorhabditis elegans 959 somatic cells XX karyotype XO karyotype 1031 cells http://www.wormatlas.org • 5 autosomes I-V and X (6 total) • XX animals become hermaphrodites • XO animals become males (chromosome non-disjunction) • With one less chromosome, males develop 79 more neurons and 25 more muscle cells than hermaphrodites, used for mating. • Hermaphrodite produces many progeny--300 worms! • Adult hermaphrodites have 959 somatic cells while males have 1031. C. elegans morphology Organs and tissues Muscles http://www.wormatlas.org Epidermal system Digestive system Reproductive system Muscular system Excretory system Nervous system Nervous system Hermaphrodite 302 neurons (118 types) 5000 chemical synapses 2000 neuromuscular junctions Male 381 neurons By comparison 105 neurons in flys, ~300 in worms Interneurons and motorneurons Major neurotransmitters: Ach -exitatory GABA- inhibitory Serotonin (HSN neuron- egg laying) Dopamine FMRF amide peptides Wood W.B. 1988. The Nematode Caenorhabditis elegans. 1-667. Life cycle of Caenorhabditis elegans Proliferation generation of embryonic founder cells bulk of cell divisions and gastrulation. a spheroid of cells: ectoderm - hypodermis and neurons mesoderm - pharynx and muscle, endoderm - germline and intestine. Organogenesis terminal differentiation 3 days at 22C http://www.wormatlas.org C. elegans cell lineage • One amazing advantage of worms is that every worm has the exact same number of somatic cells! Hatching larva=558 cells Males (XO)=1031 somatic cells+ ~1000 sperm Hermaphrodite (XX)= 959 somatic cells+ ~2000 eggs and sperm. • Somatic cells arise by an INVARIANT cell lineage. The divisions are invariant in pattern, timing, and orientation of each division. Here it is: C. elegans cell lineage EMS Know how to read a lineage diagram! 1) Each branch indicates a cell division 2) When a cell differentiates and no longer divides, line ends 3) Line ending in X= programmed cell death 4) Moving from top to bottom is moving ahead in time P1 P2 P 3 Postembryonic cell lineage was completed first. In the presence of food, cell divisions resume and postembryonic developmental program begins 3 hours after hatching. a first stage larva has approximately 671 cells, 113 undergo programmed death in the course of development. About 10% of the remaining 558 cells in a newly hatched larva (51 in hermaphrodites, 55 in the male), are blast cells that divide further. Divisions Migrations Differentiation Death http://www.wormatlas.org Direct observations are possible Cell divisions. Sequential photographs of an L1 hermaphrodite, lateral view; Nomarski optics vcn, ventral cord neurons. 0 min, interphase; 16 and 21 min, P10 prophase; 24 min, P10 metaphase; 26 min 10 anaphase; 27 mm, P10 telophase; 29 min, P9 prophase; 33 and 34 min, P9 metaphase. Sulston & Horvitz. Developmental Biology 56:110-156 (1977) Founder cell derivation by asymmetric cell division and inductions http://www.wormclassroom.org/db/sampleLineage.html Founder cells: AB, MS, E, C, D and P4. Distinctive properties of founder cells defined by: division rate nature of their progeny Germline formation in C. elegans 6 Founder Cells +98 die +14 die Small posterior cell goes germline +1 dies How are invariant lineages established and maintained? • Partitioning of prexisting maternal components • Cell-autonomous programs • Cell-cell interactions Asymmetric cell division • Sperm entry defines posterior pole. Sperm aster microtubules organize microfilaments needed to establish asymmetries. • Several genes identified that are required for this partitioning of material and are called par genes in worms. Differential partitioning of maternally expressed proteins to anterior and posterior of egg occur: PAR-1, -2 associate with posterior surface; PAR-3, -6, PKC-3 associate with anterior surface. They coordinate the polarization of cytoskeleton affecting distribution of other cell components. • P granules(ribonucleoprotein particals) segregate to posterior pole until 16-cell stage and P4, just one cell, will give a rise to germline. A-P partitioning occurs in all germline precursors. Early inductions P2-EMS • 1st transcription occurs at 4-cell stage in EMS. • Can block transcription get to 100 cell stage • Lineage commitment can occur through multiple mechanisms • P2 induces ABp but not ABa by contact • P2 induces EMS asymm. division. Where do specific tissues come from? Lineages do not strictly produce single tissue types. AB MS E C D P4 While intestine & germ-line come from single founder cells, the muscle, nervous system & skin cells each arise from multiple lineages. This means, for example, that muscles do not have one “founder” cell early in development. Cell death can be a genetically programmed cell fate. Indicate death event The Nematode Caenorhabditis Elegans. 1988. Editor William B. Wood , Cold Spring Harbor Laboratory. Specific cells with diverse developmental origins undergo programmed cell death at specific times during development. Programmed cell death is characterized by a series of specific morphological changes. There must be genes that control both the decision to express that fate and the execution. Genetics of Programmed Cell Death: CED-3, CED-4 and CED-9 have human counterparts. A ced-1 has defect in the engulfment of dying cells. Programmed cell death does not occur in a ced-3 mutant. ced-3 (caspase) is a killer. egl-1 (dead HSN neuron) Search for supression of egl (HSN restored) ced-4 (Apaf-1-like-protein) is a killer. The sister of NSM neuron in the pharynx survives instead of dying ced-9 (gf) ced-9 has similarities to the proto-oncogene Bcl-2 (B cell lymphoma) http://nobelprize.org/nobel_prizes/medicine/laureates/2002/horvitz-lecture.html The genetic pathway for programmed cell death in C. elegans http://nobelprize.org/nobel_prizes/medicine/laureates/2002/horvitz-lecture.html The principle of biological universality “One point that emerges from the studies of programmed cell death in C. elegans and other organisms is the striking similarity of genes and gene pathways among organisms that are as superficially distinct as worms and humans... I like to refer to this theme as “The principle of biological universality,” and it underlies my strong conviction that the rigorous, detailed and analytic study of the biology of any organism is likely to lead to findings of importance in the understanding of other organisms.” http://nobelprize.org/nobel_prizes/medicine/laureates/2002/horvitz-lecture.html miRNAs • miRNAs were discovered in C. elegans. • Additionally, much of what we know about miRNAs and their targets has come from studies in C. elegans. • C. elegans has over 200 miRNAs. Humans have over 1000. • Most biological processes are touched in some way by miRNAs. • The excitement generated by the discovery of miRNAs, and their similarity to siRNAs, has made a huge impact on our thinking about genetic control. Genes that affect timing and Discovery of microRNAs (miRNAs) The definition of heterochrony is a change in the relative timing of developmental events. Each scale of development, the cycle of cell divisions, the growth of tissues, the formation of organs, requires proper timing. Two general phenotypes are seen in heterochronic mutants — ‘precocious,’ in which developmental events are skipped, ‘retarded,’ in which they are repeated. A heterochronic mutation may affect different tissues: intestine, epidermis, muscle, and neurons, and different kinds of developmental events: a pattern of cell division, a cell cycle lengths, and differentiation. An example: lin-4 and lin-14 mutants Hypodermal cell lineage Moss E. 2007. Current Biology, R425. Lin-4 regulates transition from L1 to L2 stage. Lin-14 gene is a regulator of transcription. Lin-4 is the first microRNA gene. lin-4 encodes a small RNA molecule, a rare 61 nt pre-lin-4 precursor that is processed into a 22nt miRNA. These hairpin precursor is a characteristic feature of the miRNA class of regulatory RNAs. imperfectly base-paired to complementary One of lin-4’s target genes, lin-14, sequences on target messenger encodes a novel nuclear protein and is a putative transcription factor. The lin4 microRNA regulates lin-14 through specific sequences in the 3’UTR of the lin-14 mRNA. Upon lin-4 expression, lin-14 protein levels are reduced. Although transcription from the lin-14 gene still occurs, it is of no consequence. (Posttranscriptional control). Ambros V. 2004. Nature 431, 350-355 The conservation of let-7 across animal phylogeny showed that microRNAs are not exclusive to C. elegans. let-7 controls the L4-to-adult transition in C. elegans. lin-41 is one of the targets. let-7 mutants fail to execute the L4to-adult transition (the seam cells fail to terminally differentiate and continue to divide), and die by bursting through the vulva (Reinhart et al., 2000). Moss E. 2007. Current Biology, R425. Temporal regulation of the miRNAs lin-4 and let-7 and their target genes. Vella, M.C. and Slack, F.J. C. elegans microRNAs (September 21, 2005), WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/ wormbook.1.26.1, http://www.wormbook.org. MicroRNAs increase in abundance at each stage and repress specific targets that encode developmental regulators. The change in regulators at each stage leads to a succession of developmental events. MicroRNA functions in animals and disease. E. Wienholds, R.H.A. Plasterk / FEBS Letters 579 (2005) 5911–5922 miRNA-mediated repression of translation Nelson T. 2007. TRENDS in Genetics, 23: 243 Target mRNAs are recognized by miRNAs in the form of ribonucleoprotein complexes (miRNPs), through sequence complementarity. This interaction can have direct and indirect effects on translation: 1) Direct effects through inhibition of initiation of translation, which results in prevention of ribosome association with the target mRNA, or through inhibition of translation post-initiation. In the case of post-initiation repression – which includes premature ribosome drop off, slowed or stalled elongation, and cotranslational protein degradation – the repressed mRNA seems to be present in polysomes. 2) miRNPs can have other effects on targeted mRNAs, including promoting deadenylation, which might result in degradation (increased turnover). It might take place in P bodies (denoted by P), which are enriched for factors involved in mRNA degradation. 3) targeted mRNAs could be sequestered from the translational machinery and degraded or stored for subsequent use. RNAi (RNA interference) RNAi is an evolutionally conserved process of post-transcriptional gene silencing (PTGS) by which double stranded RNA (dsRNA), when introduced into a cell, causes sequence-specific degradation of homologous mRNA sequences. • 1991 Fire et al. showed that antisense RNA could inhibit gene expression when injected into worms. • Didn't always work!!!!! • 1991-1997 several papers showed that either antisense or sense RNA could interfere (Guo and Kemphues Cell:81,1995) • 1998 Fire et al. and the Mello lab showed that combination of sense and antisense worked all the time on certain genes. Probably got hairpins with sense or antisense alone. • 1998 Effect is post-transcriptional, enhanced degradation of target transcript. • 1999 First genes identified for RNAi • Essential for transposon silencing. • 2000 Dicer enzyme cleaves dsRNA precursors to 21-23bp duplex oligonuc. Important characteristics of RNAi in C. elegans systemic amplified heritable Ways to introduce dsRNA into worms: Injection Soaking feeding Plasmids for feeding Timmins L. & Fire A. 1998. Nature, 395:854. Segments were cloned between flanking copies of bacteriophage T7 promoter into a bacterial plasmid vector. A bacterial strain BL21/DE3 expressing T7 polymerase Gene from an inducible Lac promoter was used as a host. Large-scale screens for RNAi deficient mutants. Production of genome wide collections of RNAi feeding constructs RNAi (RNA interference) RNAi-induced silencing complex RNAi (RNA interference) and miRNAs 1. 2001 Three labs published papers showing miRNA were generated by dicer. 2. Explains generation of small temporal RNAs (stRNAs) such as lin-4 found in 1993. Screening for RNAi-deficient mutants The first mutants in the RNAi pathway identified by Tabara and Mello were called RNAi deficient (rde). These original screens were aimed at identifying of viable mutants, resistant to RNAi targeting pos-1, a gene important for viability. rde-1 non rde-2 Ste/him/mutator rde-3 Ste/him/mutator rde-4 non mut-2 Ste/him/mutator mut-7 Ste/him/mutator Tabara H. 1999. Cell, Vol. 99, 123–132, Existence of related silencing pathways with distinct triggering mechanisms Existence of related silencing pathways with distinct triggering mechanisms http://nobelprize.org/nobel_prizes/medicine/laureates/2006/mello-lecture.html Genetic links between RNAi and miRNA pathways. Rde-1 homologs. Grishok et al. 2001. Cell, 106: 23-34. Rde-1 homologs function: C. elegans - RNAi Drosophila - Development, epigenetic silencing Arabidopsis - development, gene silencing Phylogenetic Tree Grouping the RDE-1/AGO1/PIWI Protein Family members Grishok et al. 2001. Cell, 106: 23-34. alg-1/alg-2 (lf) show defects that are similar to those observed in heterochronic mutants Nothern blot Pre-miRNAs processing is impaired Function in miRNAs pathway Link between silencing pathways http://nobelprize.org/nobel_prizes/medicine/laureates/2006/mello-lecture.html RNAi pathway -post-transcriptional gene silencing On entering the cell, long dsRNAs act as a trigger of RNAi process. It is first processed by the RNAse III enzyme Dicer in an ATP-dependent reaction. Dicer processes dsRNAs into 21-23 nt short interfering RNA (siRNA) with 2-nt 3' overhangs. The siRNAs are incorporated into the RNA-inducing silencing complex (RISC) which consists of an Argonaute (Ago) protein as one of its main components. Ago cleaves and discards the passenger (sense) strand of the siRNA duplex leading to activation of the RISC. RNA-dependant-RNA-polymerase The remaining guide (antisense) strand of the siRNA guides RISC to its homologous mRNA, resulting in the endonucleolytic cleavage of the target mRNA http://nobelprize.org/nobel_prizes/medicine/laureates/2006/mello-lecture.html MicroRNAs - translational silencing. Novina C. & Sharp P. 2004. Nature, 430: 161 MicroRNAs – How many and what do they potentially regulate? Resources Alberts et al. Molecular Biology of the Cell. 4th Edition 21. Development of Multicellular Organisms. Caenorhabditis Elegans: Development from the Perspective of the Individual Cell. http://www.ncbi.nlm.nih.gov/books/bv.fcgi?highlight=Perspective,Individual,Development,Cell, ATLAS OF C. elegans ANATOMY. Chapter 1. INTRODUCTION TO C. elegans ANATOMY. http://www.wormatlas.org/ miRNAs Vella, M.C. and Slack, F.J. C. elegans microRNAs (September 21, 2005), WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/ wormbook.1.26.1, http://www.wormbook.org. Ambros V. The functions of animal microRNAs. Nature. 2004 Sep 16;431(7006):350-5. Review. Ambros V. The regulation of genes and genomes by small RNAs. Development. 2007;134(9):1635-41. RNAi Fire, A. et al (1998). Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391, 806–811. Timmons, L., and Fire, A. (1998). Specific interference by ingested dsRNA. Nature 395, 854. Grishok. RNAi mechanisms in Caenorhabditis elegans. FEBS Lett. 2005 Oct 31;579(26):5932-9. Epub 2005 Aug 9. Review. Yigit E. et al. Analysis of the C. elegans Argonaute family reveals that distinct Argonautes act sequentially during RNAi. Cell. 2006 Nov 17;127(4):747-57. Paper for the discussion : Parry DH, Xu J, Ruvkun G. A whole-genome RNAi Screen for C. elegans miRNA pathway genes. Curr Biol. 2007 Dec 4;17(23):2013-22. and Figure 1A from supplemental data Resources Vella, M.C. and Slack, F.J. C. elegans microRNAs (September 21, 2005), WormBook, ed. The C. elegans Research Community, WormBook, doi/10.1895/ wormbook.1.26.1, http://www.wormbook.org. B.J. Reinhart, F.J. Slack, M. Basson, A.E. Pasquinelli, J.C. Bettinger, A.E. Rougvie, H.R. Horvitz and G. Ruvkun, The 21-nucleotide let-7 RNA regulates developmental timing in Caenorhabditis elegans, Nature 403 (2000), pp. 901– 906. Boutros M, Ahringer J. The art and design of genetic screens: RNA interference. Nat Rev Genet. 2008 Jul;9(7):554-66.