Naming Compounds cations and anions
advertisement

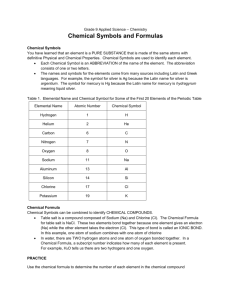
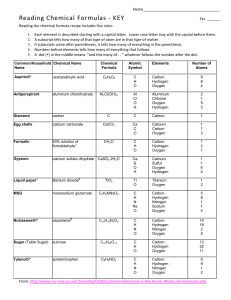
Naming Compounds, cations and anions Elements and symbols that you should know: Part 1 – The obvious ones: 1) Hydrogen H 8) Oxygen O 2) Helium He 9) Fluorine F 3) Lithium Li 10)Neon Ne 4) Beryllium Be 11) Magnesium Mg 5) Boron B 12)Aluminium Al 6) Carbon C 13)Silicon Si 7) Nitrogen N 14)Phosphorus P Some more obvious ones: 15)Sulphur S 18) Calcium Ca 16)Chlorine Cl 19) Zinc Zn 17)Argon Ar The less obvious ones: 1) Sodium Na 6) Tin Sn 2) Potassium K 7) Gold Au 3) Iron Fe 8) Mercury Hg 4) Copper Cu 9) Lead Pb 5) Silver Ag Polyatomic ions (-1 charge) • • • • • • • • • • • • • H2PO4 C2H3O2 HSO3 HCO3 NO2 NO3 CN OH MnO4 ClO ClO2 ClO3 ClO4 DiHydrogen phosphate Acetate Hydrogen Sulfite Hydrogen Carbonate Nitrite Nitrate Cyanide Hydroxide Permanganate Hypochlorite Chlorite Chlorate Perchlorate Polyatomic ions (-2 charge) • • • • • • • • HPO4 C2O4 SO3 SO4 CO3 CrO4 Cr2O7 SiO3 Hydrogen Phosphate Oxalate Sulfite Sulfate Carbonate Chromate Dichromate Silicate Polyatomic ions (-3 charge) • PO3 • PO4 Phosphite Phosphate Rule 1– If two identical elements combine then the name doesn’t change This happens with the following elements: 1) H2 4) F2 2) N2 5) Cl2 3) O2 6) Br2 These elements always go around in pairs (diatomic molecules). For example, hydrogen looks like this: Rule 2 – When two elements join and one is a halogen, oxygen or sulphur the name ends with ____ide e.g. Magnesium + oxygen magnesium oxide 1) Sodium + chlorine 6) KBr 2) Magnesium + fluorine 7) LiCl 3) Lithium + iodine 8) CaO 4) Chlorine + copper 9) MgS 5) Oxygen + iron 10)KF Rule 3 – When three or more elements combine and two of them are hydrogen and oxygen the name ends with hydroxide e.g. Sodium + hydrogen + oxygen Sodium hydroxide 1) Potassium + hydrogen + oxygen 2) Lithium + hydrogen + oxygen 3) Calcium + hydrogen + oxygen 4) Mg(OH)2 Rule 4 – When three or more elements combine and one of them is oxygen the ending is _____ate e.g. Copper + sulphur + oxygen Copper sulphate 1) Calcium + carbon + oxygen 6) AgNO3 2) Potassium + carbon + oxygen 7) H2SO4 3) Calcium + sulphur + oxygen 8) K2CO3 4) Magnesium + chlorine + oxygen 5) Calcium + oxygen + nitrogen Covalent formulae Ionic formulae H2O Water NaCl Sodium chloride CO2 Carbon dioxide CaCl2 Calcium chloride NH3 Ammonia MgO Magnesium oxide H2 Hydrogen HCl Hydrochloric acid Sulphuric acid O2 Oxygen H2SO4 HNO3 Nitric acid N2 Nitrogen NaOH Sodium hydroxide SO2 Sulphur dioxide Ca(OH)2 Calcium hydroxide CaCO3 Calcium carbonate Al2O3 Aluminium oxide Fe2O3 Iron oxide This powerpoint was kindly donated to www.worldofteaching.com http://www.worldofteaching.com is home to over a thousand powerpoints submitted by teachers. This is a completely free site and requires no registration. Please visit and I hope it will help in your teaching.







![Chemical Economics iMPORTANR CHEMISTRY[54]](http://s3.studylib.net/store/data/025216970_1-82ab8376e938e6941afbf5568df0ea97-300x300.png)