Naming Covalent/ Molecular Compounds
advertisement

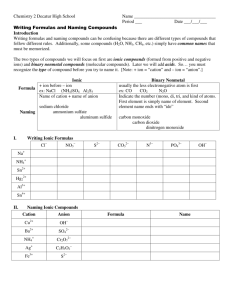
Naming Covalent/ Molecular Compounds Naming Covalent Compounds Hydrogen compounds are handled differently and will be looked at first. Nomenclature: 1) Name the hydrogen that appears in the formula. 2) Name the second element that appears in the formula, changing its ending to –ide. (Similar to ionic compounds) Examples: HCl HI hydrogen chloride hydrogen iodide Covalent Bonds use prefixes to indicate the number of atoms involved in the compounds 1= mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7= hepta 8 = octa 9 = nona 10 = deca RULES 1- The name of the first element does not get changed, only a prefix is added if it is more than one. Ex: Trisulfur tetrafluoride S3F4 2- The second element gets a different ending, as well as a prefix. Ex : phosphorus trichloride PCl3 (NOT: phosphorus trichlorine) RULES continued.. 3- If the first element in the compound is single you do not write mono: ex. CO2 - carbon dioxide (NOT monocarbon dioxide) 4- If the element’s name starts with a vowel, and the prefix ends with a vowel, you can drop the last vowel on the prefix: ex. S5O10 - pentasulfur decoxide (NOT pentasulfur decaoxide) RULES continued.. 5- Covalent compounds do not get simplified. Name the compound exactly as you see it: Ex. H2F2 = dihydrogen difluoride (NOT hydrogen monofluoride HF) Naming Covalent Compounds What is the name of SO3? STEP 1 The first nonmetal is S sulfur. STEP 2 The second nonmetal is O, named oxide. STEP 3 The subscript 3 of O is shown as the prefix tri. SO3 → sulfur trioxide The subscript 1(for S) or mono is understood. 7 Naming Covalent Compounds (Continued) Name P4S3 STEP 1 The first nonmetal P is phosphorus. STEP 2 The second nonmetal S is sulfide. STEP 3 The subscript 4 of P is shown as tetra. The subscript 3 of O is shown as tri. P4S3 → tetraphosphorus trisulfide 8 Names of Covalent Compounds Remember Prefixes are used in the names of covalent compounds because two nonmetals can form two or more different compounds Examples of compounds of N and O: NO nitrogen oxide NO2 nitrogen dioxide N2O dinitrogen oxide N2O4 dinitrogen tetroxide N2O5 dinitrogen pentoxide 9 Learning Check Select the correct name for each compound. A. SiCl4 1) silicon chloride 2) tetrasilicon chloride 3) silicon tetrachloride B. P2O5 1) phosphorus oxide 2) phosphorus pentoxide 3) diphosphorus pentoxide C. Cl2O7 1) dichlorine heptoxide 2) dichlorine oxide 3) chlorine heptoxide 10 Solution Select the correct name for each compound. A. SiCl4 3) silicon tetrachloride B. P2O5 3) diphosphorus pentoxide C. Cl2O7 1) dichlorine heptoxide 11 Learning Check Write the correct formula for each of the following: A. phosphorus pentachloride B. dinitrogen trioxide C. sulfur hexafluoride 12 Solution Write the correct formula for each of the following: A. phosphorus pentachloride 1P penta = 5Cl PCl5 B. dinitrogen trioxide di = 2N tri = 3 O N2O3 C. sulfur hexafluoride 1S hexa = 6F SF6 13 TRY THESE: a) Diphosphorus pentoxide b) Nitrogen monoxide c) Xenon hexafluoride d) Tetrarsenic decoxide More Examples a) SiO2 b) N2S5 c)P4O10 d)BrCl6 Formulas, Names and Uses of Some Covalent Compounds 16 Learning Check Write the name of each covalent compound. CO _____________________ CO2 _____________________ PCl3 _____________________ CCl4 _____________________ N2O _____________________ 17 Solution Write the name of each covalent compound. CO carbon monoxide CO2 carbon dioxide PCl3 phosphorus trichloride CCl4 carbon tetrachloride N2O dinitrogen oxide 18 Review of Covalent Compounds HI hydrogen iodide NF3 nitrogen trifluoride SO2 sulfur dioxide N2Cl4 dinitrogen tetrachloride NO2 nitrogen dioxide N2O dinitrogen monoxide Ionic and Covalent Identify each compound as ionic or covalent, and give its correct name. A. SO2 B. BaCl2 C. (NH4)3PO4 D. Cu2CO3 E. N2O4 20 Solution Identify each compound as ionic or covalent, and give its correct name. A. SO2 covalent; sulfur dioxide B. BaCl2 ionic; barium chloride C. (NH4)3PO4 ionic; ammonium phosphate D. Cu2CO3 ionic; copper(I) carbonate E. N2O4 covalent; dinitrogen tetroxide 21 Learning Check Name the following compounds: A. Ca3(PO4)2 B. FeBr3 C. SCl2 D. Cl2O 22 Solution Name the following compounds: A. Ca3(PO4)2 ionic Ca2+, PO43− calcium phosphate B. FeBr3 ionic Fe3+, Br − iron(III) bromide C. SCl2 covalent 1S, 2Cl sulfur dichloride D. Cl2O covalent 2Cl, 1 O dichlorine oxide 23 Homework Page 249 Problems: 14, 15, 16 and 17 Page 274 Problems: 93, 94 and 95 (a-c)