Chemical Reactions: Exothermic, Endothermic, and Oxygen Reactions
advertisement

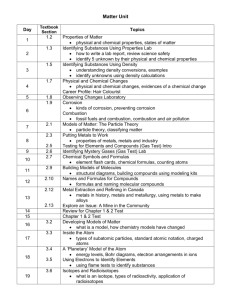
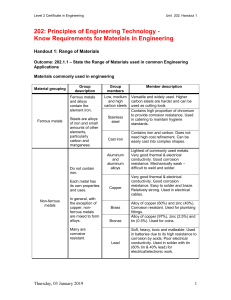
Chemical Reactions Exothermic Reactions reactions that release heat – gives off energy in the form of heat… therefore feel hot happens when you burn an object in the presence of oxygen when your body metabolizes food it gives off heat… this is an exothermic reaction Endothermic Reactions reactions that absorb heat – take up energy in the form of heat… therefore feel cold Chemical Reactions Involving Oxygen the most common type of chemical reactions involve oxygen Combustion: a chemical reaction that occurs when oxygen reacts with a substance to form a new substance and give off energy (heat) this is a highly EXOTHERMIC reaction combustion required FUEL, OXYGEN and FUEL – combustion triangle – if one is missing there is no reaction fire is the most common example of a combustion reaction wood + oxygen → heat + light + carbon dioxide + water fire is perhaps the first chemical reaction used by humans combustion also occur in the engine of a car or in your barbeque gasoline or propane + oxygen → energy + water + carbon dioxide Corrosion: a slow chemical reaction that occurs when oxygen in the air reacts with a metal the reaction is enhanced when there is moisture in the air this is called oxidation of metals (or rocks) a common example is rust such as what we see on a car iron 4 Fe (s) + + oxygen 3O2 (g) → → iron oxide 2Fe2O3 (s) once corrosion starts it can accelerate… because there is more and more surface area for the reaction to occur on one way to protect metal from corrosion is to apply a thin coat of paint which eliminates or slows down the contact between the iron and the oxygen some metals are coated with zinc, which is more resistant to corrosion … this is called galvanizing sometimes harder metals like chromium are used as a coating to protect softer metals like iron … the process of coating one metal with another is called electroplating Cellular Respiration: a chemical reaction that takes place in the cells of our body food (glucose) + oxygen → energy + water + carbon dioxide C6H12O6 (s) + O2 → energy + H2O + CO2