Carboxylic Acids
advertisement

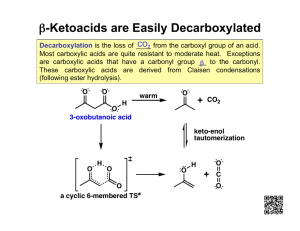
Lecture 10 Carboxylic Acids 1.Introduction: Carboxylic acids are characterized by the presence of the carboxyl group –C=O .That gives an acid the general formula RCOOH.typically | O-H These molecules are weak acids in aqueous solution. 2. Nomenclature: Organic acids are named from the parent alkane by dropping the final-e and adding –oic acid .thus CH3COOH commonly called acetic acid,has the systematic name (IUPAC) ethanoic acid.other examples of carboxylic acids are shown in the following examples: O O O ║ ║ ║ H-C-OH CH3-C-OH CH3CH2C-OH Systematic(IUPAC):methanoicacid ethanoic acid Common name: Formic acid acetic acid O ║ CH3CH2CH2C-OH Butanoic acid Butyric acid propanoic acid propionic acid Carboxylic acids containing six or fewer carbons are frequently called by their common names. In the systematic nomenclature the position of substituent is designated by a number. The carbonyl of carboxylic acid always the C-1 carbon .in common nomenclature,the position of a substituent is designated by a lower case Greek letter,and the carbonyl carbon is not given a designation.The carbon adjacent to the carbonyl carbon is the αcarbon,the carbon adjacent to the α-carbon is the ß-carbon and so on. O O ║ ║ EX(1) : CH3CH2CH2CH2CH2C-OH CH3CH2CH2CH2CH2C-OH 6 5 4 3 2 1 є б γ ß α Systematic Common 55 CL O | ║ EX(2): CH3CH2CHCH2CH2C-OH Systematic:4-chlorohexanoic acid Common: γ-chlorocaproic acid 3- Preparation of carboxylic acids: These are several methods for preparing carboxylic acids: (1) primary alcohols can be oxidized to give carboxylic acids: O CrO3 ║ CH3(CH2)8CH2OH CH3(CH2)8COH H2O,H2SO4 Decan-1-ol Decanoic acid (2) Aldehydes can be oxidized to give carboxylic acids: O O ║ AgNO3 ║ CH3CH2CH2CH2CH2CH CH3CH2CH2CH2CH2COH Hexanal NH4OH Hexanoic acid (Tollens′ reagent) (3) oxidation of alkenes: KMno4 a- CH3-CH=CH2 CH3COOH+CO2+H2O Tollen′s reagent b- RCH=CHR′+O3 RCOH+R′COH dil.acid Aldehyde RCOOH+R′COOH Carboxylic acids (4) Hydrolysis of esters: RCOOR′ H3O,∆ RCOOH+R′OH 4- Physical properties: Like alcohols ,carboxylic acids form strong intermolecular hydrogen bonds,which has notice value effect on boiling points. Carboxylic acids normally boil at much higher than alkenes or alkyl halide of similar molecular weight.(acetic acid at 118◦c,chloropropane at 46.6◦c).Carboxylic acids are often foul in odor ,and miscible in water (till 56 4 carbon atoms),and higher than (5)are insoluble .solubility like alcohols; in less polar solvent(ether, alcohol).although the carboxylic acids are much weaker than mineral acids ,but much stronger acids than alcohols. 5-Chemical properties (1) Decarboxylation (loss of CO2) RCOOH ∆ RH+CO2 (2) Formation of acid chlorides 3RCOOH+PCL3 3RCOCl Acid chloride The net effect is substitution of the -OH group by -Cl (3) Esterification : RCOOH+R′OH RCOOR′+H2O O ║ CH3C-OH +H-OCH2CH3 O ║ CH3C-OCH2CH3 +H2O Acetic acid+ ethanol ethyl acetate+ water 57