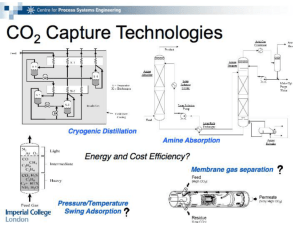
CHM 235 Summer 2002
advertisement

CHM 235 Course Outline and Homework in McMurry (6th ed.) Fall 2006 Chapter 1: 1.19-1.26, 1.28-1.31, 1.34, 1.36-1.39, 1.46, 1.47 Atomic orbitals (s,p), electronic configuration (through calcium) Covalent vs ionic bonds, Lewis structures, valence electrons Valence bond theory, hybridization (sp3, sp2, sp, N and O) Molecular shape (tetrahedral, trigonal planar, linear), molecular orbital theory Chapter 2: 2.25-2.31, 2.35-2.37, 2.39-2.41, 2.43-2.46, 2.49, 2.55, 2.56 Polar covalent bonds, electronegativity, dipole moments Formal charge Resonance Acids and bases (B-L, and Lewis), pKA Drawing condensed and skeletal structures Chapter 3: 3.23-3.28, 3.30, 3.32, 3.34, 3.36, 3.39, 3.44-3.49, 3.53, 3.54 Functional groups (alkanes, alkenes, alkynes, halides, alcohols, ethers, amines, nitriles, aldehydes, ketones, carboxylic acids, esters, amides) Constitutional isomers Nomenclature of straight-chain alkanes (up to dodecane), branched alkanes, alkyl groups, and cycloalkanes (including cis/trans) Chapter 4: 4.24-4.26, 4.29-4.32, 4.38, 4.41, 4.42, 4.46 Sawhorse and Newman projections Conformations of ethane, propane, butane (staggered, eclipsed, gauche, anti) Ring strain, steric strain Conformations of cyclohexane (axial and equatorial hydrogens, ring flip, mono and disubstituted cyclohexanes) Chapter 5: 5.19, 5.21, 5.23, 5.24, 5.27, 5.28, 5.30, 5.32, 5.37, 5.39-5.42, 5.47 Overview to addition, elimination, substitution, and rearrangement reactions Free radical halogenation reactions (initiation, propagation, termination) Polar reactions (electrophile, nucleophile) Curved arrow formalism (“arrow-pushing”) to show reaction mechanisms Thermodynamics (Go = Ho - TSo) (exergonic, endergonic, exothermic, endothermic) Bond dissociation energies (Ho = energy used to break bonds–energy gained by making bonds) Energy diagrams (reaction coordinates, transition states, reaction intermediates, RDS) 1 Chapter 6: 6.23-6.32, 6.39-6.44, 6.78, 6.48 Degree of unsaturation in alkenes Naming alkenes Cis/trans isomerism and E, Z designation Alkene stability (most substituted alkene is the most stable) and hyperconjugation Electrophilic addition of HX across the double bond (Markovnikov’s rule) Carbocation stability (30 more stable than 20 more stable than 10) and rearrangements Hammond postulate Rearrangement reactions (H- and CH3- transfer) Chapter 7: 7.23-7.30, 7.32-7.36, 7.40-7.44 Preparation of alkenes: elimination reactions (reactions 1a, 1b) Addition of HX (rxn 2a), X2 (rxn 2b), X2 + H2O (2c) Addition of H2O: acid catalyzed hydration, oxymercuration (rxn 2d), hydroboration (rxn 2e) Syn addition of H2 (hydrogenation) (rxn 2f) Syn addition of OH (hydroxylation) (rxn 2g) Addition of carbenes to alkenes (rxns 2h1 and 2h2) Oxidative cleavage reactions: ozonolysis (rxn 3a), aqueous acidic KMnO4 (rxn 3b), and HIO4 (rxn 4) Polymerization Chapter 8: 8.19-8.29, 8.31-8.37 Electronic structure and IUPAC names for alkynes Preparation of alkynes: elimination reactions (rxn 1a) Addition of HX (rxn 2a) and X2 (rxn 2b) Addition of H2O: Hg(II)-catalyzed hydration (rxn 2c) and hydroboration/oxidation (rxn 2d) Addition of H2 (reduction) (rxn 2e1 and 2e2) Oxidative cleavage (rxn 2h) Alkyne acidity and formation of acetylide anions (R—CC) (rxn 1b) Alkylation of acetylide anions, a substitution reaction (rxn 1b) Introduction to organic synthesis (the fun part!) Chapter 9: 9.34-9.36, 9..38-9.46, 9.51-9.55, 9.65, 9.67, 9.79, 9.80 Enantiomers, chirality, specific rotation Sequence rules for R and S Diastereomers and meso compounds Racemic mixtures and their resolution (chiral drugs!) Fischer projections Stereochemistry in reactions (addition of HBr, Br2) 2 Chapter 10: 10.17-10.25, 10.28, 10.31-10.33, 10.36-10.38, 10.40 Naming alkyl halides Preparation of alkyl halides using free radical halogenation Preparation of alkyl halides by addition to alkenes (rxn 1b), allylic bromination of alkenes (rxn 1a); substitution reactions with alcohols (rxns 1c1-1c3) Stability of the allylic radical Grignard reagent formation (rxn 2a), Gilman reagent formation (rxn 2b) Reactions of Grignards and Gilman reagents (rxns 2c and 2d) Oxidation/reduction reactions in organic chemistry Chapter 11: 11.25-11.31, 11.34-11.38, 11.41, 11.45-11.47, 11.50, 11.54, 11.55, 11.5911.60, 11.68 The SN2 substitution reaction (stereochemistry, kinetics, the substrate, the attacking nucleophile, the leaving group, the solvent) (rxn 1b) The SN1 substitution reaction (stereochemistry, kinetics, the substrate, the attacking nucleophile, the leaving group, the solvent) (rxn 1a) The E2 elimination reaction and Zaitsev’s rule (rxn 2b) The E1 elimination reaction (rxn 2a) Chapter 12: 12.17-12.19, 12.28-12.30, 12.34-12.40 Introduction to mass spectrometry Introduction to IR spectroscopy Chapter 13: 13.31-13.32, 13.35-13.44, 13.46, 13.49-13.52 (and maybe some others) Introduction to NMR and chemical shift 13C NMR and DEPT 13C NMR 1H NMR (proton equivalence, chemical shifts, integration, spin-spin splitting, complex spin-spin splitting) 3