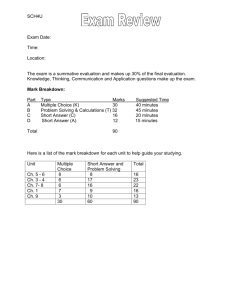
SAMPLE FINAL EXAM - SOLUTION Statistical Mechanics and

SAMPLE FINAL EXAM - SOLUTION
Statistical Mechanics and Chemical Dynamics (Chem 130C)
(Dated: Jun 2, 2008)
1. A first-order rxn, A → P, goes only forward, with a rate constant of 10 s
− 1 . How long before
90% of A has turned into P? How would your answer change if there were a back rxn at rate 2 s
− 1
? Hint: In the second part, an equilibrium is established.
If the reaction goes only forward:
[ A ] = 0 .
1[ A ]
0
= [ A ]
0 e
− kt
; t = 0 .
23 s .
If there is a backward reaction:
K = k
1 k
− 1
=
[ P ]
[ A ]
= 5;
[ A ] eq
=
1
6
[ A ]
0
= 0 .
17[ A ]
0
.
So A never gets 10% of the original concentration.
2. For a first-order reaction, the activation barrier is calculated to be 100 kJ/mol, and 10% of the reactant vanishes in 20 minutes when performed in an ice bucket. How much would have been gone if it had been done at room temp (20 o C)? Hint: Arrhenius.
I use subscript 1 to denote room temperature and 2 for ice bucket.
1
293
−
1
273
!
= 20 .
23;
For first-order reaction: k
1 k
2
= exp −
100 × 10 3
8 .
314
[ A ] ln
[ A ]
0
= − kt ;
At ice bucket, the left hand side is ln 0 .
9 , so we know k
2
= 0 .
00527 min
− 1 temprature, k
1
= 20 .
23 k
2
= 0 .
107 min
− 1
, and for room
, plug in the above equation, we know [ A ] = 0 .
118[ A ]
0
, which means that 88 .
2% have been gone.
3. A rxn has the following simple mechanism: A → B + C, B → A, A+C → P, with rate constants k, k
0
, k
2 respectively. Use the steady-state approximation to show that this is first-order in [A], and find the effective rate constant for the rate of consumption of A. What conditions must the k ’s satisfy to make your answer accurate?
Use steady state approximation on B and C: d [ B ]
= k [ A ] − k
0
[ B ] = 0; dt d [ C ]
= k [ A ] − k
2
[ A ][ C ] = 0; dt
2
And we can solve for [B] and [C]:
[ B ] = k [ A ]
; k 0
Plug in the differential rate law for [A]:
[ C ] = k k
2
; d [ A ]
= − k [ A ] + k
0
[ B ] − k
2
[ A ][ C ] = − k [ A ] .
dt
This means that the effective rate constant is k . The steady state approximation is more accurate when the consumption rate is much greater than the formation rate, which means that k
0 k and k
2 k .
4. The reversible rxn A → P has rate constants 0.1 min
− 1 forward and 0.033 min
− 1 backwards. If
I start with 0.1 moles of A, what will be in my tank after an hour? Hint: This can be thought of as relaxing toward equilibrium.
I use k
1 to denote the rate constant for forward reaction, and k
2 for backward reaction.
d [ A ]
= − k
1
[ A ] + k
2
[ P ] = − k
1
[ A ] + k
2
([ A ]
0
− [ A ]); dt where the second equal sign used mass conservation. And we can do the integration: d [ A ]
( k
1
+ k
2
)[ A ] − k
2
[ A ]
0
= − ( k
1
+ k
2
) dt ;
( k
1 ln
( k
1
+ k
2
)[ A ] − k
+ k
2
)[ A ]
0
− k
2
2
[ A ]
0
[ A ]
0
= − ( k
1
+ k
2
) t ;
We plug in [ A ]
0
= 0 .
1 mol, and we get [ A ] = 0 .
0248 mol. (volume is the same)
Note: you can also use equilibrium as your reference point (and use an x to denote the deviation from equilibrium concentration), just as what we did in class.
5. I have 10 moles of Argon in 1 liter at room temperature, and I suddenly double the volume.
How is the mean velocity after the change related to that before? Repeat for, the collision frequency and the mean free path.
T does not change, so kinetic energy does not change, hence mean speed does not change.
λ = √ kT
2 σp
= kV
√
2 σnR
=
¯
; z
So we know as V is doubled, the mean free path becomes twice the original value; and the collision frequency decreases to half the original value (because ¯ does not change).