Overall Standard Heat of Reactions
When doing an energy balance around a reactor, we use the heat-of-formation method and not the heat-ofreaction method. In the heat-of-formation method, the molar enthalpy of any mixture is calculated by
starting with the heat of formations for each pure compound. Because of this fact, the heat-of-formation
method automatically accounts for multiple reactions that may be occurring within the system boundary.
A chemical reaction can be either exothermic (giving up energy) or endothermic (requiring energy). For
multiple reactions, how do you determine the overall energy change for the combined reactions?
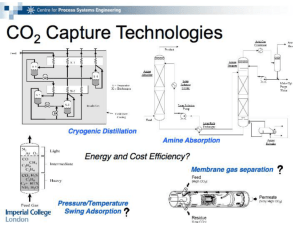
You only need to use the standard heat of reaction ( H rxn ) for each chemical reaction at 25°C and 1 atm
to calculate the overall standard heat of reactions. If you have multiple reactions, four possibilities exist for
the overall standard heat of reactions as follows:
H rxn for each reaction
-
Rxn I:
Rxn II:
Rxn III:
etc.
+
+
+
+
-
+
-
For the first column, the overall would be exothermic.
For the second column, the overall would be endothermic.
For the third column, the overall could be exothermic or endothermic.
For the fourth column, the overall could be endothermic or exothermic.
For the third and fourth column, the overall will depend upon how many Rxn I's are occurring, how many
Rxn II's are occurring, how many Rxn III's are occurring, etc. You will not know that until you solve for
the extent of reactions ( R l ’s) in the material balances.
After finding the
Rl ’s, you can then calculate the overall standard heat of reactions as follows:
Overall H rxn
o
Hˆ rxn
,l
RI H rxn, I
nc
l, j
Hˆ of , j
RII H rxn, II
etc.
for l 1, 2, , nr
j 1
If the overall ΔH of reaction is negative, than it is exothermic; otherwise, it is endothermic.
where
nc
nr
Rl
is
is
is
o
Hˆ rxn
,l
is
l, j
is
Hˆ of , j
is
number of chemical compounds within the system boundary.
the number of chemical reactions within the system boundary.
the number of reaction events, often called the extent of reaction, for
the l-th reaction in units of (mass-reaction of l )/time. Some
example units are: g-rxn/s, kg-rxn/h, and lb-rxn/min.
the standard heat-of-reaction for the l-th chemical reaction at the
standard condition of 25ºC and 1 atm. The units are (energy of
l/mass-rxn of l).
the stoichiometric coefficient for the j-th component in the l-th
reaction. It is positive (+) for a product and negative (−) for a
reactant. It has units of (mass-mole of j)/(mass-reaction of l ), like
g-mol/g-rxn, kg-mol/kg-rxn, or lb-mol/lb-rxn.
the standard heat-of-formation for pure component j-th at the
standard condition of 25ºC, 1 atm, and stable phase ( Ph oj ) of j-th.
The units are (energy of j/mass-mole of j).
v11.01.24
© 2011, Michael E. Hanyak, Jr., All Rights Reserved
1 of 1