SNC 2D Chemistry Unit Test Review
advertisement

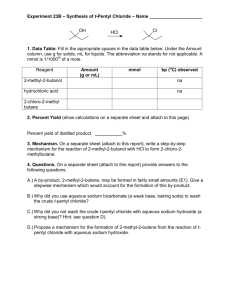
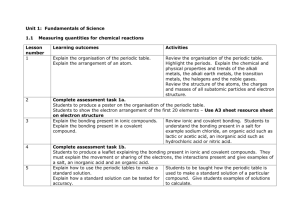
SNC 2D Chemistry Unit Test Review While doing this review please use your periodic table and table of polyatomic ions that was provided for the first quiz. Review from the beginning; periodic table and forming compounds. 1. What information can you get about an element by looking at the periodic table? Use Nickel as your example. 2. What is an ionic compound? A molecular compound? 3. What are the rules for naming each ionic compounds and molecular compounds/ 4. What do subscript numbers mean in a chemical formula? 5. What are the five major chemical families on the periodic table? List example elements for each family Review from the middle; balancing equations and acids and bases. 1. In a chemical equation what do the large numbers in front of chemical formulas mean? 2. What are the three states of matter and how are they represented in a chemical equation? 3. Please write the balanced chemical equation for the following reaction: Aqueous sodium chloride reacts with bromine gas to produce solid sodium bromide and chlorine gas. 4. Balance this equation and write a description of the reaction. KNO3(s) → KNO2(s) + O2(g) 5. Please create a table listing common characteristics of acids and bases 6. What is used to measure the acidity or basicity of a substance? What are the strongest acids and bases? 7. What does pH stand for 8. What are the rules for naming acids? Write the formula for nitric acid and the name of HF. 9. What are the reactants and products of a neutralization reaction? 10. Please write the balanced neutralization reaction that takes place between sulphuric acid and sodium hydroxide. 11. If the salt (ionic compound) produced from a neutralization is not soluble in water what will you see? 12. What compounds contribute to acid rain? 13. What is a natural stone that can neutralize acidic lakes? 14. Write the balanced chemical equation for the following neutralization: hydrochloric acid reacts with aqueous sodium hydroxide to produce aqueous sodium chloride and water. 15. Balance the following neutralization and write it as a word equation H2SO4(aq) + KOH(aq) → K2SO4(aq) + H2O Review from the end: Reaction types 16. What are the five types of chemical reactions and the general formulas to represent each. 17. Please balance each of the following chemical equations and state what type of reaction has occurred. Fe2(SO4)3 + Ba → BaSO4 + Fe HgO(s) → Hg(l) + O2(g) Na3PO4(aq) + CuSO4(aq) → Na2SO4(aq) + Cu3(PO4)2(aq) C3H8 + O2 → CO2 + H2O Mg(s) + F2(g) → Mg2F(s) 18. What would the balanced chemical equation and word equations be for the reaction between calcium chloride and sodium carbonate? Good Luck!!