Molecule Madness Online Learning Activity
advertisement

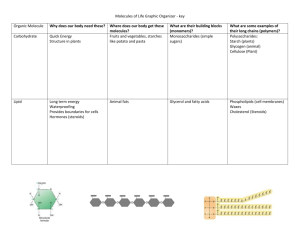
Molecule Madness Online Learning Activity NAME_____________________ Go to each of the websites that are given and then answer the related questions or fill in the charts. 1. http://www2.nl.edu/jste/bonds.htm http://bcs.whfreeman.com/thelifewire/content/chp02/02020.html Describe an ionic bond – use the example given on the website: Describe a covalent bond – use the example given on the website: What is the difference between a polar and a nonpolar covalent bond? 2. http://programs.northlandcollege.edu/biology/Biology1111/animations/hydrogenbonds.html http://www.johnkyrk.com/H2O.html The two websites above deal with hydrogen bonds and the polarity of water. Explain why water forms weak hydrogen bonds between its molecules. How many water molecules can one water molecule hydrogen bond to? 3. http://programs.northlandcollege.edu/biology/Biology1111/animations/dissolve.html http://www.mhhe.com/physsci/chemistry/essentialchemistry/flash/molvie1.swf Explain how salt (sodium chloride) dissolves in water. When sodium chloride dissolves what particles are floating around in solution? 4. http://www.johnkyrk.com/pH.html Use the pH scale (by moving the arrow back and forth) find the pH of the following substances: Strong HCl (hydrochloric acid): _____ Digestive juices: _____ Tomatoes: _______ Chicken: _____ Distilled water: ______ Urine: _____ Sea water: _______ Baking Soda: _____ Oven cleaner: ______ 5. http://www.zerobio.com/drag_oa/coolermolecules/drag_and_drop_cooler_molecules.html Use the “drag and drop molecules to figure out the 10 structural formulas of the given molecules. Put the name and formulas below. How did you figure out the formulas? What “rules” did you use? 6. http://www.mcgrawhill.ca/school/applets/abbio/quiz/ch06/how_enzymes_work.swf Draw a diagram of enzyme action showing the enzyme, the substrate, the active site, the binding of the substrate at the active site creating an enzyme-substrate complex, the product and the original enzyme unchanged. Go to the following websites to fill in the chart below and to answer the questions on the next page. http://www.cst.cmich.edu/users/baile1re/bio101fall/atmolorga/molecu/moleprez/sld001.htm http://www2.visalia.k12.ca.us/eldiamante/science/biology/powerpoints/biochem.pdf http://bcs.whfreeman.com/thelifewire/content/chp03/0302002.html http://www.wisc-online.com/objects/index_tj.asp?objID=AP13104 (carbohydrates) http://www.wisc-online.com/objects/index_tj.asp?objID=AP13204 (lipids) http://www.wisc-online.com/objects/index_tj.asp?objID=AP13304 (proteins) Type of Molecule Elements Monomers General structure function Examples Test for 1. Describe the relationship between a monomer and a polymer and give an example from one of the macromolecules in the chart. 2. Which macromolecule is not a polymer? 3. What is the chemical process that links the monomers together? 4. What is the chemical process that breaks monomers apart? 5. What essential protein is involved in the breaking and linking of monomers? 6. Describe the differences between a monosaccharide, a disaccharide, and a polysaccharide? 7. Name three important polysaccharides. Describe their functions. 8. What type of molecule is glucose? 9. How many different amino acids are there? 10. One of the most important roles of proteins is to function as enzymes. Generally, describe the function of enzymes. 11. What does it mean to say that a protein functions by a “lock and key” model? 12. What is the active site of a protein? 13. What determines the shape of a protein? 14. What type of molecule is insulin? What is the function of insulin? 15. What type of molecule is hemoglobin? What is the function of hemoglobin?