Osmosis
advertisement

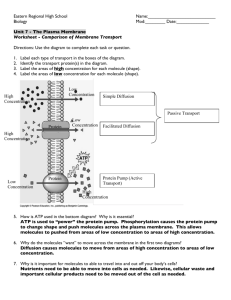
Notes to go with the Sept 29 class. Membrane transport Active – movement through membrane against a concentration gradient due to energy input (ATP) and transport proteins in the membrane. Passive – principle means is diffusion – movement from a high concentration to a low concentration until equilibrium is established. Movement is away from a high concentration until the molecules hit the sides of the container where they bounce back and cross to the other side of the container. This goes on until equilibrium is reached. As long as diffusion is occurring, equilibrium has not been reached. Factors influencing diffusion – kinetic energy of molecules: temperature, size, charge, media, etc. Diffusion can be through a membrane. Small molecules like H2O, O2, CO2 can diffuse easily. Example: cells in sea water. O 2 dissolves easily in sea water so there is O2 available. Cells will have less O2, so diffusion of O2 will occur. As the O2 is used up, CO2 is produced. The CO2 concentration will be higher inside the cell, so CO2 moves out. 1. Osmosis – special case of diffusion where you have large molecules that would like to move out of the cell to establish equilibrium, but they can’t get through the membrane. However, H2O molecules can , so they move into the cell to try a equalize concentrations. The starch interferes with H2O movement so more water flows in than flows out. The membrane is said to be semipermeable in a case like this. Net water movement into the cell can cause the cell to swell and burst. If the larger molecule is outside the cell, then net water flow will be out of the cell and the cell will crenate. (This is how pickling works!) The membrane that you are using is regenerated cellulose which doesn’t absorb solutes and isn’t charged. The MWCO (molecular weight cut-off) is 13,000 Daltons. (a Dalton is the approximate size of a H atom). All dialysis membranes are listed by the MWCO instead of a pore diameter size. Some membranes have straight channels for molecules to move through and some have convoluted channels to differentially hold out wider molecules. So, even though a molecule may be relatively small molecular weight-wise, it may not get through a convoluted- channel membrane. Long, linear molecules may get through easier than shorter, wider molecules. It would help in understanding your results if you had knowledge of the shape of your molecule, but then again, charge and other factors affect the transport of molecules through the membrane. See handouts attached). I am thinking that it may be that the membranes pores might not be exactly the same size over the whole membrane, but the weight of the molecule that transports through can be determined with some level of confidence. Handouts that go with this: Diffusion, Osmosis, and Cell Membranes 3 pg Diffusion, Osmosis, and Cell Membranes - Teacher Background. 6 pg Diffusion and Osmosis – 6 page lab Dialysis: an overview. Technical resource with info on dialysis tubing and specs. SpectraPore dialysis tubing with information on different sizes of tubing defined by the MWt in Daltons. Testing for sugars O Reducing sugars have aldehyde groups ( C ) that are oxidized to carboxylic O R R Acids ( C ) Reducing sugars are monosaccharides. R O They reduce Cu+2 to Cu+. The cyclic ring must be open to expose the aldehyde group, so since this form is in equilibrium with the open form, any monosaccharide molecule that is in a ring form with be in the open form in a fraction of a second. The Cu+2 is a blue color and the Cu+ combines with oxygen to make an oxide which is rust colored. See handout with the equations on it. Testing for starches. The information for starches is in a handout, but it is not easy to find out what causes the color change. Starch molecules insert in between the molecules in the chains and cause a change in the absorption spectra which causes a color change. There are a number of handouts: Reducing Sugars 1 pg Background Information on Glucose, Fructose, and Cellulose 4 pg Sugars and Fats 3 pg Diffusing Glucose Molecules INTERACTIVE STUDENT ACTIVITY Starch Structure 2 pg Starch 4 pages Enzymes Enzymes are catalysts that speed up reactions. 2H2O2 →2H2) + O2 Catalase promotes conversion of H2O2 to water and oxygen. H2O2 is a harmful oxidizing agent. Catalase uses H2O2 to oxidize toxins like phenols, formic acids, and alcohols. It is found in peroxisomes (cell organelles) which are involved in oxidation of fatty acids and synthesis of cholesterol and bile acids. H2O2 is a by-product of fatty acid oxidation. White blood cells produce H2O2 to kill bacteria (bacteria have no peroxisomes). Catalase prevents H2O2 from harming the cell itself. The rate at which catalase works depends on the concentration of H2O2, temperature, pH, salt concentration and presence of inhibitors or activators. Every enzyme has an optimal range; activity is less outside the range. The lower the concentration of substrate, the faster the reaction will occur. The higher the temperature, the more the reaction. There is also an optimum pH for enzymatic activity. The optimum pH for catalase is around 7.0, and for salivary amylase, it is 4.6 to 5.2. Handout Catalase FAQ 1 ½ pages, both sides Testing Enzyme Activity 1 page, but a great lab. Cell structure and function Slides that I have: Handouts – Animal Tissue Review 2 pg Hyaline cartilage – Handouts: Cartilage 4 pages Skeletal muscle – Handouts: Muscle Types 3 pages Online slides – you can look at the three types of muscle with large photos and good information http://www.bioedonline.org/slildes/slide01.cfm?q+cardiac%20muscle... Neurons – Handouts – Nervous tissues 4 pg Nervous tissue 1pg Nerve tissue 2 pg Intestinal epitheliumHandouts - Epithelial Tissue 1 pg Absorption 1 pg The Small Intestine 2 pg, but it’s a powerpoint slide show of 11 slides with good pictures, diagrams, and easy to understand information There are two computer activities that you can do. One is Animal Cells and Tissues which is just looking at and reading a very good site for tissue and cell information. On your handout, the URL is given. The next site is really tremendous. Go to http://phschool.com/science. You’ll get the Prentice Hall Book site, BUT scroll down to the Biology Place and click on “more”. Then click on BioCoach. Go down to Cardiovascular System II: the Vascular Highway. You can click on instruction and read that or I suggest you head to the next concept which is blood. Here, you can click on Practice (requires you to load Macromedia Shockwave Player to do the activity) or you can click on Review and get another interactive activity. For each concept (there are four or five) you can either Practice with interactive activities or Review with them. At the end, you can take the test. You can print at any time and go back at any time. This is just for one topic – cardiovascular system, but there are others and this would be a great tool for a science class.