Work-sheet 1 - Chemistry With BT
advertisement

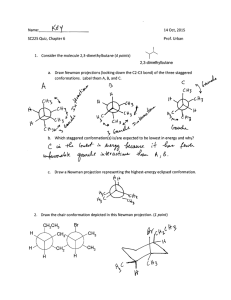
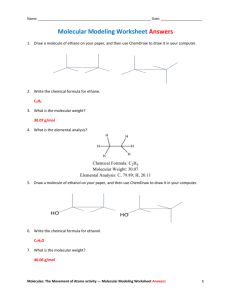
ORGANIC CHEMISTRY I LAB WORK-SHOP 1 Week 4 I. Alkanes: Structures and Models 1. Write the names and structures of straight chain alkanes from C1 to C10 2. Construct butane using models 2.1. Identify all primary (1o), secondary (2o), and tertiary (3o) carbons (if any) 2.2. Place a methyl group on one of the secondary carbons. Draw the structure. Name the structure. 2.3. Place a methyl group on the other secondary carbon. Name the structure. Discuss with your group members whether the name of the new molecule should be different (review the naming rules) 2.4. If the names of the structures in 2.2 and 2.3 are the same, what is the relationship between the two secondary carbons? 3. Each group should build one of the structures below using models (no need to place any hydrogens on carbons) C C C C-C-C-C-C-C-C-C C C CC C-C-C-C-C-C C (1) 3.1. 3.2. 3.3. 3.4. C C C C C-C-C-C-C C C-C-C-C (2) (3) Name your group’s molecule assuming that all missing bonds are hydrogens. Compare the name of your structure with those of the other groups. After confirming the name of each structure, compare your structures as well. Write a short paragraph summarizing your conclusion Under what conditions would the three structures be different? (refer to question 3.5 before answering question 3.4) 3.5. Are the structures on the right different from each other? If so, when are they different? 4. Give a definition for the term conformation 4.1. Can “conformational isomers” be isolated from one another? 4.2. Build a model for ethane as in question 3.5 4.3. How many conformational isomers of ethane are there? 4.4. Draw the two “extreme” conformations of ethane in 3.5 using Newman Projection Representations 4.5. Give a definition of a dihedral angle as it applies to ethane 4.6. What is the dihedral angle in the staggered conformation of ethane 4.7. What is the dihedral angle in the eclipsed conformation of ethane 5. Construct a model of propane (similar to the models in 3.5, but substitute one of the white balls (H atom) with a methyl group 5.1. Make a model of the eclipsed conformation. Draw its Newman Projection Representation 5.2. Make a model of the staggered conformation. Draw its Newman Projection Representation 6. Draw an energy diagram (similar to that given in the text), and draw the correct conformations (in Newman Projection Representation) of ethane in the minima and maxima points of the graph. 7. Construct a model of 1,2-dibromoethane. 7.1. Make a model of the eclipsed conformation (note that there will be two eclipsed conformations, one of which will be less stable than the other). Draw their Newman Projection Representations. 7.2. Make a model of the staggered conformation (note that there will be two staggered conformations, one of which will be less stable than the other). Draw their Newman Projection Representations. Identify the names given to the two different staggered conformations. 7.3. Construct an energy diagram similar to the one for question and draw the correct conformations (in Newman Projection Representation) of 1,2-dibromoethane in the minima and maxima points of the graph. 8. Construct a model of butane and follow the directions as in 7.1 to 7.3. When you draw a Newman Projection Representation for butane, assume as if you are working with ethane (refer to question 3.5); except that replace one hydrogen on each carbon with a –CH3 group. 9. Draw ethane below in “zig-zag” form (in skeletal structures); however, show all hydrogens in “dashed” and “wedged” forms to illustrate the distinguishing aspects of the eclipsed and staggered conformations. 10. The type of representation you have drawn for question 9 is referred to as “saw-horse”. Show “saw-horse” representations of 10.1. Gauche conformation of butane 10.2. Anti conformation of 1,2-dibromoethane 10.3. The less unstable eclipsed conformation of butane