Building Models: Molecular Compounds
advertisement

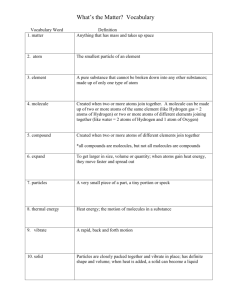
SNC1D – Chemistry Name: _____________________ BUILDING MODELS OF MOLECULES A molecule is a combination of two or more atoms. These atoms are held together by bonds. The bonds represent the electrons of the atoms. We draw the bonds of atoms using structural diagrams. A structural diagram represents a molecule with chemical symbols for atoms and lines for the bonds. You must know the number of bonds in selected atoms so you can draw or build the molecule. Know this! However, molecules may contain a different number of atoms & this will change the number of bonds that is present. Water (H2O) has 2 hydrogen & 1 oxygen atom. H O H Structural diagram # of connections per atom Atom Hydrogen Oxygen Nitrogen Carbon one two three four Hydrogen peroxide (H2O2) has 2 hydrogen & 2 oxygen atoms. H O O H Structural diagram Below are examples of ball and stick diagrams (all ball and stick diagrams can be written as structural diagrams). SNC1D – Chemistry Name: _____________________ Building Models You must know the number of bonds in the atoms so you can build molecules. All bonds must be used up for each atom. No bonds can be left undone. Water: H – O – H Model Atom Colour Hydrogen # of bonds per atom Oxygen Nitrogen Carbon Build each model below. Check it with the teacher, and then draw the ball and stick diagram for each one in the space provided. Use the correct colours on your drawings. If you finish all of the molecules try to create a glucose molecule (C6H12O6). Ammonia (NH3) Ethane (C2H6) Oxygen gas (O2) Nitrogen gas (N2) Carbon dioxide (CO2) Methanol (CH3OH) Formaldehyde (HCHO) Ethanol (C2H5OH) Ethylene (C2H4) Acetic Acid – vinegar (CH3COOH)