Name
advertisement

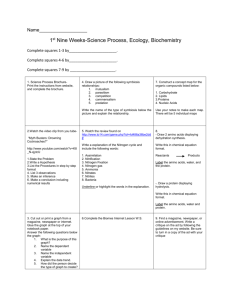
Name___________________________ Date___________ Period___________ Classifying Organic Molecules Problem: What are living things made of? There may be as many as 10,000 different kinds of molecules in living things. But are there a few common patterns? Part 1: Classifying 1. Look at your molecule cards. What do they all have in common? _____________________________________________________________________ 2. What differences do they have? _____________________________________________________________________ 3. What are the 4 categories of organic molecules? _____________________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ _____________________________________________________________________ Part 2: Interpreting Molecular Structure 4. Sort out all cards that contain nitrogen (N) into a “nitrogen” pile. Take this pile and sort out those that also contain phosphorus (P). How many cards do you have that contain both nitrogen and phosphorus? ___________ Group 1: Nucleotides 5. Nucleotides combine to form nucleic acids including DNA (deoxyribonucleic acid), RNA (ribonucleic acid), and ATP (adenosine triphosphate). Where is DNA found? _____________________ 6. What is DNA’s function for the cell? ___________________________________ 7. What is RNA’s function for the cell? ___________________________________ 8. What is ATP’s function for the cell? ___________________________________ 9. Each nucleotide contains three parts; a nitrogenous base, a five carbon sugar, and a phosphate group. Circle and label the three parts below: 10. Which of the CHNOP elements are found in nucleic acids? _________________ 11. List 2 differences between your nucleotide cards. ________________________ __________________________________________________________________ Group 2: Amino Acids 12. Amino acids are the building blocks of proteins. Proteins are molecules that play many important roles in the body; muscle structure, hormones, antibodies, hemoglobin for carrying oxygen, transport proteins for carryin molecules across the cell membrane, chemical messengers in the nervous system, and enzymes to control chemical reactions. Each amino acid contains an amino group: NH3 and a carboxyl group: COO or COOH. Label both, circle the amino group and put a square around the carboxyl group. 13. Find the cards with an amino group and a carboxyl group. How many amino acids do you have in your set? ________________ 14. Which of the CHNOP elements are contained in ALL amino acids?___________ There should be 9 other cards left over in your “nitrogen” pile. Set these aside, we will come back to them later. Group 3: Sugars 15. Sugars are the building blocks of Carbohydrates. They are hydrates of carbon, having the general formula “Cn(H2O)n. Sugars are burned “oxidized” to release energy in what cellular process? _______________________________________ 16. If n = 6, how many carbons are there? __________ How many hydrogens? _________ How many oxygens? ___________ 17. Take your “no nitrogen” pile and sort out those cards that have OH attached to most carbons. Be aware that organic chemists use many shortcuts in drawing complex molecules. They often do not include the letter C for carbon in ring structures. How many cards did you find? ______________ 18. What are the two forms of sugars? _____________________________________ 19. Which of the CHNOP elements are found in sugars? _______________________ 20. Sugars can be joined together in long chains to form large molecules called starch, cellulose, and glycogen. Starch and glycogen are easily broken down into sugars for energy. Cellulose, on the other hand, which is made in plants, can be broken down only by a few organisms in the world (primarily the bacteria in the guts of termites). What happens to the cellulose (fiber) you eat? __________________________________________________________________ Group 4: Lipids 21. Fatty acids are monomers of lipids. They are long chains with a carboxyl group at one end and have an absence of oxygen except in the carboxyl group. How many fatty acids did you find? _______________ 22. Fatty acids that contain no double bonds between carbons are saturated. An unsaturated fatty acid contains one or more double bonds between carbons. How many saturated fatty acids do you have? __________ Unsaturated? __________ 23. To form a lipid at least one fatty acid must bind to what other monomer? _____________________ 24. These lipids form the cell ____________________________. 25. If you have three remaining cards in the “no nitrogen” pile you have been successful in classifying molecules. These three cards are steroids which are used as chemical messengers throughout the body. Which CHNOP elements are found in lipids? _____________________ There are still 9 cards left in your nitrogen pile. Those cards are not polymers or monomers. See if you can figure out which molecules these are. Label the back of all the molecule cards. Quiz your lab members. Organize your information on this table: Monomer Polymer Elements Examples Cellular Function Nucleotides Amino Acids Monosaccharide Glycerol & Fatty Acids 26. Write a paragraph explaining the similarities and differences between the organic molecules. Include information on their structure and how it relates to their function. __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________ __________________________________________________________________