cornell notes properties of water
advertisement

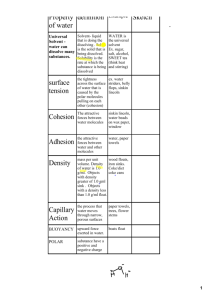
CORNELL NOTES TOPIC/OBJECTIVE: NAME: MR. IRWIN, BIOLOGY CLASS/PERIOD: DATE: ESSENTIAL QUESTION: QUESTIONS: NOTES: You will be reading the paragraphs below about waters stickiness (polarity). You will then reply to me, giving a summary of how the pictures on the board represent Cohesion, Adhesion, and Surface tension. Go through each paragraph and highlight or underline information that supports your summary. Try to limit your response to 40 words or less, still using proper sentence structure, spelling, punctuation and grammar!!! In the questions section come up with 3 questions about the unique properties of water. Water Properties Cohesion and adhesion: The hydrogen bond between water molecules is the reason behind two of water's unique properties: cohesion and adhesion. Cohesion refers to the fact that water sticks to itself very easily and does not spread out into a completely flat sheet when coming into contact with a surface. Adhesion means that water also sticks very well to other things. When water comes into contact with these surfaces, the adhesive forces are stronger than the cohesive forces. Surface tension: Water also has a high level of surface tension. The cohesive forces that hold together molecules of water are stronger between the molecules on the surface. Surface molecules do not have molecules above them to bond with. The bonds of cohesion between a surface molecule and its neighbors are stronger than the forces that hold the molecules together below the surface. The stronger cohesive forces create a film on the surface of the liquid that makes it more difficult to pass through than if already submerged. Summary / response: QUESTIONS: SUMMARY: NOTES: