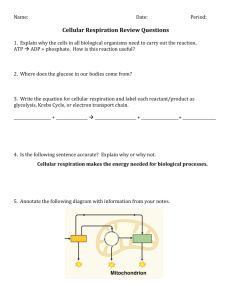
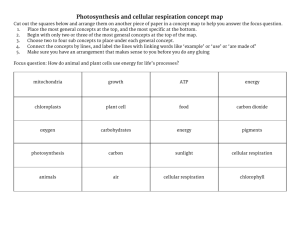
Quarter 2-3 Notebook - Foothill Technology High School
advertisement

Foothill Technology High School Biology Interactive Notebook Quarter Two DEPARTMENT STATEMENT: Students will actively experience science – both the concepts and practices of the disciplines. Science requires a student to understand facts, processes, interactions and the methods of discovering these things in an organized, yet creative fashion. Students will learn to operate various pieces of scientific equipment that serve as tools to improve the accuracy of measurement and analysis. Foothill Technology High School is dedicated to integrating concepts and projects across curricula. By integrating several subjects, we hope that each student gains a deeper understanding and appreciation of the fact that one subject cannot stand without the other. Page | 1 Table of Contents Lab Group Assignments Page 3 Course Expectations and Guidelines Pages 4 - 9 Notebook Requirements Pages 10 – 12 Student Grade Sheets Pages 13 – 17 Biochemistry Unit Pages 18 – 66 Cell Membrane and Transport Unit Pages 67- 98 Cell Structure and Function Unit Pages 99- 118 Cell Reproduction (Cycle) Unit Pages 119- 142 Cellular Energy (Energentics) Unit Pages 143- 222 Appendix A: Deadlines and Contacts Page Appendix B: Periodic Table Page 224 223 Page | 2 Lab Groups When you are assigned to a new lab group, write down your new lab partner’s names and e-mail addresses or phone numbers in the space provided. Make sure you write down the name of your group (i.e., “red group” or “cell group”) and your seat number for that group. Group: Lab Partner’s Name Seat Number: Phone Number Group: Lab Partner’s Name Seat Number: Phone Number Group: Lab Partner’s Name E-mail Address Seat Number: Phone Number Group: Lab Partner’s Name E-mail Address E-mail Address Seat Number: Phone Number E-mail Address Page | 3 COURSE EXPECTATIONS and GUIDELINES REQUIRED SUPPLIES: 1. Glue sticks (for putting items in Interactive Notebook) 2. Index cards (for concept cards) and at least 8 envelopes for storing these 3. College ruled, loose-leaf paper 4. Pencils, pens (blue or black ink), highlighters, erasers 5. Storage pouch for pencils, pens, glue sticks, unused index cards, etc. 6. Binder with three subject dividers reserved for Biology (The sections will be: Reference, Study Aides, and Assessments) 7. At least 10 divider to glue into your biology Interactive Notebook to separate units. 8. Calendar/Agenda for recording assignments 9. Suggested: Colored pencil set: 12 set 10. Suggested: Small ruler with metric measurement on one side UNITS OF STUDY Unit Fall Semester Summary Statement Scientific Concepts Maintaining an Interactive Notebook; Identifying Greek and Latin Roots to Scientific Words; Understanding and Applying the Scientific Method and Experimental Design; Employing the Metric System Unit Spring Semester Summary Statement Inheritance Exploring fundamentals of inheritance by examining cellular processes. This unit includes Meiosis and reproductive Biology. Ecology Investigating the interdependence of diverse living organisms and their interactions with the components of the biosphere. Understanding the role that humans play in the environment. Applied Genetics Exploring Mendelian inheritance, principles of molecular biology, as well as implications of applied genetics on individuals, society and the environment. Chemistry of Life Investigating the properties of biologically important compounds through guided and directed inquiry investigation. Nucleic Acids and Protein Synthesis Exploring similarities and differences between the structure and function of DNA and RNA. Understanding protein synthesis. Cells in Living Things Studying similarities and differences between prokaryotic and eukaryotic cells, modes of locomotion and movement, transport, forms of cellular reproduction, and cellular energetics. Evolution Exploring fundamentals of biological evolution through laboratory simulations and research of historical studies. Page | 4 COURSE EXPECTATIONS and GUIDELINES CLASS EXPECTATIONS Students are expected to: 1. Be on time. (Refer to Foothill’s Survival Guide for policies on tardiness and absenteeism) 2. Be prepared with appropriate materials (interactive notebook, pen, etc.) 3. Be involved in class discussions. 4. Be respectful of self, teacher, classmates, guest speakers, and school property. 5. Be aware of safety protocol in the lab and follow it. MARKING PERIOD GRADES Grading scale: A B C D* F 90 – 100% 80 – 89% 70 – 79% 60 – 69% 50 – 59% Work not attempted by the deadline date will be recorded as a zero. * Biology is a pre-requisite for Chemistry. You must earn at least a “C” in both semesters of Biology in order to go onto Chemistry the following year. All students with goals of attending a University after high school must consider taking at least Biology and Chemistry in order to fulfill University eligibility requirements. EXPECTED WORKLOAD Quizzes and unit tests – every 2-4 weeks Lab investigations and activities – 1 per week Homework (including online assessments) – 1 to 3 times a week Projects – one big project per quarter WEIGHTING OF TASKS and ZANGLE ASSIGNMENT CATEGORIES Tests and Quizzes Projects Daily Homework Online Quizzes and Classwork Semester Final 30% 20% 15% 15% 20% COURSE EXPECTATIONS and GUIDELINES Page | 5 HOMEWORK Homework will be related to the curriculum and will be assigned to strengthen skills, reinforce concepts, and/or prepare for a lesson, unit, or activity. Students should expect some homework on a daily basis. There are two general categories of homework: 1. Peer evaluated: In cooperative groups, students will evaluate work in Interactive Notebooks as either following or not following the guidelines. This grade will be totaled and entered when the IntNB is collected. 2. Instructor evaluated: This includes online assessments, projects, conceptmastery worksheets, and lab conclusions. LATE WORK AND /OR MISSING WORK: • • • Each assignment will have a due date. This is the date by which you are expected to submit the assignment. Missing work will be recorded in the grade book as a zero. If a student is absent from class, that student is responsible for making up missed work. EXTRA CREDIT 1. Individual Extra Credit: There are select opportunities for a small amount of individual extra credit to be earned. These will be discussed in class. 2. Group Extra Credit: Large assignments for extra credit will be considered on a class-level basis, not an individual basis, and will only be offered to students who have all assignments completed. There will be one group extra credit option each semester. RENAISSANCE INCENTIVES Tickets: Tickets will be awarded on a merit basis. Outstanding behavior can earn a student and/or an entire class a ticket. Tickets may also be earned for outstanding work (i.e., quiz and test scores). Students will drop their tickets in a drawing for various incentives. Other Incentives Include: Action Incentive A or B on a project Cheat sheet on a test 1 month of homework completion HW late pass 1 month no tardies 5 minute late or leave early pass Date Earned Date Used Page | 6 Safety Rules in the Laboratory Notebook Copy 1. During experiments, listen carefully to instructions and follow them as exactly as you can. In case of confusion or uncertainty, ask questions! Performing unauthorized experiments will not be tolerated. 2. No horseplay will be tolerated. 3. All students will wear safety goggles/safety glasses in the laboratory whenever experiments are being performed that warrant eye protection. Notify your teacher if you are wearing contact lenses. Circumstances when safety goggles will always be worn are: When anything is heated, when students handle chemicals, or when there is the potential for chance flying debris. 4. Never eat in lab or drink out of glassware. Never taste or smell anything unless you are given specific directions to do so. 5. Any injury, major or minor (cut, burn, etc.), must be reported to the teacher at once. Put cold water on burns first. Chemical spills should be washed off immediately with water. Know the location of all laboratory safety equipment. Chemicals in the eyes must be flushed immediately with the eyewash. Hold the eye open, rotate the eyeball, and continue washing for 10-15 minutes. 6. Do not touch equipment without permission. All unauthorized experimentation is strictly forbidden. This includes all chemicals, models, or apparatus. Complete all pre-lab work. Don’t do any lab procedures until approved by instructor. 7. When heating a test tube, always point the open end away from you and others. It should be at a slant rather than straight up in most cases. 8. If the fire alarm should sound during a lab, turn off all heat sources and proceed quietly from the classroom to the assembly area outside. 9. If glass is broken, the teacher should be informed immediately. Students should not handle broken glass; teachers will dispose of broken glass. 10.Be careful not to contaminate the class supply bottle. Use disposable papers with solids. Use the same supply spoon with the same chemical. Don’t put any excess material back into the supply bottle. 11.Dispose of all materials as per instruction. Do not assume that all liquids go down the sink! 12.Dissection equipment can only be used on specimens. Any misuse of dissection tools will result in disciplinary action. 13.Before class will be dismissed, all equipment and sinks must be clean, and desktops must be clean and dry. If the above guidelines are not followed, you may not be allowed to participate in the lab and may not receive credit for the activity. I have read the guidelines and understand what is expected of me in the laboratory. _________________________ __________________________ _________ _____ Print Student’s Name Student’s Signature Date Period Page | 7 BIOLOGY INTERACTIVE NOTEBOOK REQUIREMENTS The Biology Interactive Notebook contains most of the information that will be explored during each semester. A few of the activities are listed: Right-brained activities Reflections on in-class activities Usually contain images and in color Examples o Graphic Organizers o Concept Cards & Maps o Flow-charts o Venn Diagrams & Foldables o Unit Cover and Back Pages Left-brained activities In-class activities and notes Usually recorded in pen or pencil Examples o Cornell Notes o Lab Procedures and Data o Lab Conclusions o Worksheets o Fill in the blank Notes The following are specific instructions for Left-side Assignments that will be assigned on a frequent basis in Biology. CONCEPT CARDS DIRECTIONS FRONT OF CARD: Illustration: Draw or paste an illustration of the vocabulary word or concept Link: Link another word or idea that helps you remember the original vocabulary word on card BACK OF CARD: Students add the following information the back of the concept card where appropriate: Word: Write the vocabulary word at the top of the card. Definitions: Should be in students own words (check with dictionary or text) Characteristics or features: If appropriate, write down a short list of characteristics for the words. What prefixes or suffixes make up the word? Examples from the text and/or personal experiences: If appropriate, students provide examples of the words on their concept cards. These can be in written or pictorial form. Personal sentences: Students writes sentences using the words. Page | 8 CONCEPT CARDS EXAMPLE FRONT OF CARD: hairy Link: another word or idea that helps you remember the original vocabulary word Illustration of vocabulary word or concept BACK OF CARD: Vocabulary Word CILIA Definition 1. Small hair-like projections from the surface of cells 2. Noun Characteristics 3. Cilia, unlike flagella, are shorter. Sentence from your textbook that uses this word 4. Many bacteria use cilia to move around. Your own Sentence Page | 9 BIOLOGY INTERACTIVE NOTEBOOK REQUIREMENTS FLOW-CHARTS Before performing any lab or activity in class, each student must be prepared for the lab. This preparation is reflected in a flow-chart of the procedure for the day’s lab. Flow-charts must meet the following requirements: 1. Each step must be included 2. Each step must be represented as a picture depicting the step 3. Each step must also include text, briefly describing that point in the procedure 4. Arrows must connect each step The flowchart to the left is an example of a lab procedure flowchart from Promega.com. Page | 10 BIOLOGY INTERACTIVE NOTEBOOK REQUIREMENTS FOLDABLES Concept Map Book Instructions: 1. Fold a sheet of paper along the long or short axis, leaving a two-inch tab uncovered. 2. Fold in half or in thirds. (Additional tabs can be created by folding into more parts.) 3. Unfold and cut along the inside fold lines to create tabs. 4. Identify the concept by writing key words or using pictures on the two-inch tab. Draw arrows from the central idea to the tabs, data will be recorded underneath each tab. Concept maps demonstrate relationships between ideas. They help you understand concepts by clarifying ideas and terms, and by dividing complex concepts or processes into smaller parts. You can use concept maps to relate, define, brainstorm, and sequence. Venn Diagram Instructions: 1. Fold a sheet of paper in half like a hotdog. 2. With the paper horizontal, fold the right edge toward the center, trying to cover one half of the paper. 3. Fold the left side over the right and crease to form three tabs. 4. Draw two overlapping ovals on the front. 5. Cut up the two valleys on one side only. Step 1 Step 3 Step 2 Step 4 Page | 11 BIOLOGY INTERACTIVE NOTEBOOK REQUIREMENTS Student Design Unit Cover and Back Pages At the beginning and end of each unit, students will design a unit cover and back page, respectively. The guidelines for each follow: Unit Cover Pages: Cover pages are worth five points, and must follow each guideline listed below. Each guideline is worth one point. 1. Colorful: It must be in four or more colors 2. Neat: It must be neat (last-minute work will cost you points!) 3. Unit Focused: Each unit cover page must include the name of the unit 4. Unit Relevant: The image on the unit cover page must be unit-related 5. Tabbed: Each unit cover page must include a tab with the name of the unit on it Unit Back Page Concept Maps: Unit back pages are worth five points and must follow each guideline listed below. Each guideline is worth one point. 1. Unit Focused: The central theme of the unit must be the central “bubble” of the concept map. 2. Include Vocabulary: There must be at least ten concept “bubbles” in the entire concept map. 3. Unit Relevant: The ideas bubbled in on the concept map must be ideas or terms from the unit covered. 4. Linked: The ideas must be connected by lines or arrows. 5. Correct: The ideas linked must be correctly drawn together. Page | 12 STUDENT GRADE SHEET Page# Date Assignment Title Grade Peer Initials / / / / / / / / / / / / / / / / / / / / / / / / / / / Page | 13 STUDENT GRADE SHEET Page # Date Assignment Title Grade Peer Initials / / / / / / / / / / / / / / / / / / / / / / / / / / Page | 14 STUDENT GRADE SHEET Page # Date Assignment Title Grade Peer Initials / / / / / / / / / / / / / / / / / / / / / / / / / / Page | 15 STUDENT GRADE SHEET Page # Date Assignment Title Grade Peer Initials / / / / / / / / / / / / / / / / / / / / / / / / / / Page | 16 STUDENT GRADE SHEET Page # Date Assignment Title Grade Peer Initials / / / / / / / / / / / / / / / / / / / / / / / / / / Page | 17 Biochemistry Unit Student Design Cover Page (see guidelines on page 12) Page | 18 Biochemistry Unit Front Page At the end of this unit, I will: Know the six elements that are commonly found in living things. Recognize the different types of chemical bonds that are important to biology. Know the importance of water to biological systems. Understand why enzymes are important and how they work in biological systems. Know the four categories of organic compounds, what monomers make them up, and what bonds hold them together. Roots, Prefixes and Suffixes I will understand are: Group One: neu-, iso-, -tope, co-, -valent Group Two: -lyst, Group Four: macro-, poly-, -mer, carbo-, -hydrate The terms I can clearly define are: Inorganic Chemistry: atom, nucleus, proton, neutron, electron, element, isotope, compound, covalent bond, molecule, ion, ionic bond, van der Waals force Water: polar molecule, nonpolar molecule, hydrophilic, hydrophobic, hydrogen bond, acid, base, pH, buffer Chemical Reactions: chemical reaction, reactant, product, activation energy, catalyst, enzyme, substrate, active site Organic Chemistry: macromolecule, polymer, carbohydrate, lipid, protein, amino acid, nucleic acid, nucleotide, monomer, hydrolysis, dehydration synthesis The assignments I will have completed by the end of this unit are: Unit Cover Page Completed vocabulary cards for the terms underlined above Atoms Family Worksheets and Song (sung with class) Periodic Table Basics Worksheets Covalent Bonding and Ionic Bonding Worksheets Balancing Act Worksheet Bonding with Classmate Worksheet and Activity Tested for the Presence of Organic Compounds in various foods Filled out the Lipids section of Organic Compounds table Completed the Concept Map for the Unit Back Page (this one is started for you) Page | 19 Page | 20 Inorganic Chemistry Fill in the Blank Notes Who named the atom and when was it named? _________________________________________________________ _________________________________________________________ _________________________________________________________ What does Dalton’s Atomic Theory say? _________________________________________________________ _________________________________________________________ _________________________________________________________ Fill in the blank Chemical actions happen when ________________________________ _________________________________________________________ _________________________________________________________ What part of Dalton’s theory has been rejected? _________________________________________________________ _________________________________________________________ _________________________________________________________ Who discovered the electron? When did he discover it? Page | 21 How did he discover it? _________________________________________________________ _________________________________________________________ _________________________________________________________ What is the mass of an electron? Who discovered the proton? _________________________________________________________ _________ When was it discovered? What is its mass of a proton? ____________________________ Who discovered the neutron? _________________________________________________________ When was it discovered? _________ What did Rutherford propose? _________________________________________________________ _________________________________________________________ _________________________________________________________ Page | 22 Draw a Bohr model for Oxygen? Define atomic number _________________________________________________________ ______________________________________________________ _________________________________________________________ Fill in the blank Sometimes there are more or less neutrons in a nucleus, these are called _________ What is chemical bonding? _________________________________________________________ _________________________________________________________ _________________________________________________________ What does an atom do to achieve stability? _________________________________________________________ _________________________________________________________ What is a covalent bond? _________________________________________________________ _________________________________________________________ Page | 23 What is a single bond? What is a double bond? What is a triple bond? What is a chemical reaction? _________________________________________________________ _________________________________________________________ _________________________________________________________ _________________________________________________________ _________________________________________________________ _________________________________________________________ _________________________________________________________ Page | 24 Page | 25 Page | 26 Page | 27 Page | 28 Periodic Table of Biologically Significant Elements Tape or Glue your Folded Periodic Table below Acrostic Poem for the six common elements of life Carbon C Hydrogen H Oxygen O Nitrogen N Phosphorus P Sulfur S Page | 29 Building an Atom: Use your notes to help you fill in the blanks below. To build an atom you need to know the atomic number, which is the number of _________________, and the atomic mass, which is the number of ____________________ and ___________________. The number of protons equals the number of negatively charged subatomic particles or __________________. Using the Periodic Table of Elements you discover that Carbon has an atomic number of _____ and an atomic mass of (round off) _____. Since you know that protons and neutrons are located in the _________________ of the atom and that moving around the nucleus are the ________________ , you could draw the Carbon atom as shown in the space below. 6P 6N or 6P 6N 2 4 Using the above models and your Periodic Table of Elements, draw an oxygen atom. or Page | 30 Page | 31 Page | 32 Page | 33 Making a Covalent Bond In a covalent bond, electrons are shared. By sharing electrons, atoms can become more stable. A stable atom is one in which the outermost energy level is _________________. How many electrons does hydrogen (H) have? _________ How many electrons does helium (He) have? _________ Which atom is stable, hydrogen or helium? __________________ Two hydrogen atoms can become stable by sharing electrons and forming a covalent bond as shown below. 1P 1P If carbon bonds with hydrogen, it needs four hydrogen atoms (one electron each) to fill its outer energy level. The resulting molecule is called CH4 or methane. Draw a carbon atom in the space below. Then attach 4 hydrogen atoms to the carbon atom to form a methane, CH4, molecule. Page | 34 Making an Ionic Bond When an atom gains or loses an electron, it becomes an electrically charged atom and is called an ____________. The atom that gains an electron becomes a ( positive / negative ) ion. The atom that loses an electron becomes a ( positive / negative ) ion. Oppositely charged ions are attracted to each other and an _______________ bond is formed. Draw a potassium (K) atom and a chlorine (Cl) atom below. To become stable, what could the potassium atom do? _____________________________________________________________ To become stable, what could the chlorine atom do? _____________________________________________________________ What kind of bond would join K+ and Cl-- together to form potassium chloride? _____________________________________________________________ Page | 35 Page | 36 Page | 37 Page | 38 Page | 39 Page | 40 Page | 41 Properties of Water Draw the Bohr Model of Water over the Space-filling model to the right (Use your teacher’s example as a guideline.) What is water’s chemical formula? Which element has more electrons? What is the partial charge on the Hydrogens? on the Oxygen? How does this charge affect the way water molecules bond with other water molecules? Page | 42 Biochemistry: The unique properties of water Water molecules are ____________________________________ They are attracted to other water molecules through relatively weak _____________________________. ________, at a very small rate (1 out of 500,000,000!) Can water form ions? How would you quantify (count) this ionic disassociation? The _________ scale The “____________ of Hydrogen” scale Besides water, what elements and compounds are essential to life? Organic compounds 1. Mostly contain Carbon, Hydrogen, Oxygen, Nitrogen, Phosphorus and Sulfur* 2. Four categories of organic compounds: 1. C 2. L 3. P 4. N 3. All formed and separated in similar ways 1. Formation (Polymerization): Dehydration Synthesis 2. Separation: Hydrolysis Page | 43 Polymerization _________________ _____________________: A bond is formed and a water molecule is lost. ______________________: A bond is broken when a water molecule is added. Page | 44 • Polymerization (definition): – Forming of large organic_______________________ by the joining of smaller repeating units called _____________________ Carbon = The element of life • • • Carbon’s Valence has ____ electrons Can bond with ____ elements Can form chains, rings, branches, & isomers Lipids • • Polymerization A lipid is any molecule that DOES ________ mix with water. Lipids function in: – Energy (E) storage, – forming cell membranes, – and as chemical messengers (e.g., hormones) • ____________________ (hydrophobic) • Made up mostly of Carbon and Hydrogen (with a few Oxygen) 1. Fats (Triglycerides) – Glycerol + 3 Fatty Acids – _______________ = No Double Bonds (solid) – _______________ = Double Bonds (liquid) 2. Phospholipids a. Glycerol with Phosphate Head + 2 Fatty Acid Chains b. ___________________ (“Both” “lover”) i. Hydrophilic head ii. Hydrophobic tail c. Forms 2 layers in water d. Makes up cell membranes 3. Sterols a. Lipids whose Carbon Skeleton consists of 4 fused rings b. Includes: ______________________________ ______________________________ ______________________________ c. Makes up cell membranes Page | 45 Organic Compounds What elements make up Proteins? _____, _____, _____, _____ and sometimes _____ What are the functions of Proteins? 1. ________________________________________ 2. ________________________________________ 3. ________________________________________ 4. ________________________________________ What elements make up Lipids? _____, _____, and _____ What are the functions of Lipids? 5. ________________________________________ 6. ________________________________________ 1. ________________________________________ 7. ________________________________________ 2. ________________________________________ 8. ________________________________________ 3. ________________________________________ 9. ________________________________________ What indicator would one use to test for the presence of Lipids? _________________________________________ What indicator would one use to test for the presence of Proteins? What elements make up Carbohydrates? _____, _____, and _____ What are the functions of Carbohydrates? 1. ________________________________________ ________________________________________ What elements make up Nucleic Acids? _____, _____, _____, _____ and _____ What is the function of Nucleic Acids? ______________________________________________ 2. ________________________________________ __ What indicator would one use to test for the presence of Simple Sugars? ___________________________________ What indicator would one use to test for the presence of Starch? _________________________________________ Page | 46 Page | 47 Page | 48 Page | 49 Testing for the Presence of Organic Compounds ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Page | 50 Testing for the Presence of Organic Compounds BACKGROUND The foods you eat are made of organic compounds. You can perform chemical tests to learn what foods contain carbohydrates, lipids, and/or proteins. HYPOTHESES There will be several hypotheses written for this lab. You will write one for each type of organic compound being tested. Use the prediction table to help you form your hypotheses. Part I Hypothesis: If the food substance contains starch and is tested with the indicator ____________________, then the solution will change colors to _________________. Part II Hypothesis: If the food substance contains sugar and is tested with the indicator ____________________, then the solution will change colors to _________________. Part III Hypothesis: If the food substance contains lipid and is tested with the indicator ____________________, then the solution will change colors to _________________. Part IV Hypothesis: If the food substance contains protein and is tested with the indicator ____________________, then the solution will change colors to _________. Prediction Table FOOD LIPIDS PROTEINS STARCH SUGAR APPLE BUTTER EGG WHITE FISH POTATO SPINACH VEGETABLE OIL WATER Page | 51 Create a Matrix: How are you going to set up this experiment? This matrix will help you keep track of which substances have been added to your paper, well-plate, or test-tube. Be sure to label each substance that is being tested. Fill this in DURING the experiment. Apple Butter Egg White Fish Potato Spinach Vegetable Oil Water Paper (test for lipids) Biuret’s Solution (test for protein) Lugol’s Iodine (test for starch) Benedict’s Solution (test for sugar) Pre-lab Questions: 1. Two of these indicators cannot be used with the plastic well-plate. Which two are these, and why can’t you use the plastic well-plate for either of these? _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ 2. Will the data collected during this experiment be qualitative or quantitative? Explain. _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ Page | 52 Testing for the Presence of Organic Compounds (Procedure Flowchart) Page | 53 Testing for the Presence of Organic Compounds MATERIALS 8 test tubes test-tube rack lab apron safety goggles plastic gloves Pipette stirring rod distilled water test-tube holder Biuret reagent Benedict’s solution Well plate Lugol's solution Paper Bag or other paper water bath and hot plate Food substances: apple juice melted butter raw egg white raw fish raw potato raw spinach vegetable oil PROCEDURE 1. Read all the directions for this activity before you begin your work. Answer the prelab questions on page 231. Draw a flowchart for the entire procedure on page 232. 2. Put on your safety goggles, lab apron, and plastic gloves. 3. Put 8 test tubes in your test-tube rack. Label each test tube by putting masking tape near the top edge of the test tube. Use a pencil to write one of the seven food substances on each label. Mark the eighth label water. The water is your control. 4. Collect two well-plates and lay the well plates on a sheet of paper. Trace the wellplate, and label where you will put each of the food substances to be tested. The matrix on page 200 should serve as a guideline. The only two indicators to be tested in the well-plates are Lugol’s iodine and Biuret’s solution. 5. Pick up a piece of paper bag, and label the spaces where you will be placing the food substances. Paper is your indicator, no other indicator needs to be added to the paper bag test. PART I: Testing for Carbohydrates (STARCH) 1. Use a medicine dropper to put ~10 drops of each food in the well in the well-plate with the matching label. Add 3-4 drops of Lugol's solution (iodine) to each well. 2. Starch is one form of carbohydrate. If the substance in your test tube contains starch, it will turn a blue-black color when it mixes with the iodine solution. 3. Observe the contents of your test tubes and Record the amount of starch present (0, +, ++, +++, ++++) in your data table. The food which contains the most starch should be recorded as ++++. 4. Empty and wash each well thoroughly, and return to the stock supply table. Page | 54 PART II: Testing for Carbohydrates (SUGAR) 1. Use a pipette to put ~10 drops of each food into the test tube with the matching label. Add 10 drops of Benedict's solution to each test tube. CAUTION: Benedict’s solution is poisonous. Do not get any in your mouth and do not swallow any! 2. Use a test-tube holder to carefully place the test tubes in the hot water bath your teacher has prepared. Heat the test tubes for 2 to 3 minutes. CAUTION: Use a testtube holder to handle hot test tubes. Point the open end of a test tube away from yourself and others. 3. Use a test -tube holder to return the hot test tubes to the test-tube rack. If the substance in your test tube contains sugar, Benedict's solution will change color. See Table 1 below: Table 1: Appearance of Substance after Adding Benedict's Solution Amount of Sugar in Food 0 none + trace Color blue blue/green ++ +++ little sugar some sugar green yellow ++++ much sugar orange/red 4. Observe your test tubes (using white paper as a background). Record the amount of sugar present, in your data table. 5. Empty your test tubes, clean them thoroughly, and return them to the test tube rack. PART III: TESTING FOR LIPIDS 1. Use a pipette to put ~1 drop of each food onto the newsprint. 2. Observe and compare/contrast the translucence of each food substance. Record the information, in order of translucence (0, +, ++, +++, ++++) in your data chart. The food which contains the most lipids should be recorded as ++++. PART IV: TESTING FOR PROTEIN 1. Use a pipette to put ~10 drops of each food into the well of the well-plate with the matching label. Use a pipette to carefully add 10 drops of Biuret reagent to each test tube. CAUTION: Biuret reagent can burn your skin. Wash off spills and splashes immediately with plenty of water while calling to your teacher. 2. Observe the contents of each test tube (using white paper as a background). If the food contains proteins, it will turn a pinkish purple. Record the amount (0, +, ++, +++, ++++) of protein for each food substance in your data table. The food which contains the most protein should be recorded as ++++. 3. Empty the well plate and clean them thoroughly. Before leaving the laboratory, clean up all materials and wash your hands thoroughly. Page | 55 Data Table FOOD LIPIDS PROTEINS STARCH SUGAR APPLE BUTTER EGG WHITE FISH POTATO SPINACH VEGETABLE OIL WATER ANALYSIS QUESTIONS/CONCLUSION 1. Were you able to accept your entire hypothesis? _______________________________________ If not, what surprised you? _____________________________________________________________ _____________________________________________________________ _____________________________________________________________ 2. Which compound is most common in foods that come from plants? _____________________________________________________________ Which compound is most common in foods that come from animals? _____________________________________________________________ Does water contain any of the organic compounds you tested? _______ Explain why water was used as the control. _____________________________________________________________ _____________________________________________________________ Would you call this a positive or negative control? ______________ Page | 56 3. If you wanted to reduce the amount of fat in your diet, what foods would you avoid? _____________________________________________________________ _____________________________________________________________ 4. Which foods tested would your body use for a quick burst of energy? _____________________________________________________________ _____________________________________________________________ For energy when no carbohydrates are available? _____________________________________________________________ _____________________________________________________________ For building body parts? __________________________________________________________ Page | 57 Lipids 1. Looking at the structure of the _____-glyceride above, do you think this lipid is a big or a small organic compound?___________________________________________ 2. According to the structure above, and what you have learned about bonds. What do you think one of the major functions of lipids are in terms of living systems?___________________________________________________________ 3. Without gaining or losing any atoms, how could you make this structure take up more space?_________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ 4. Draw what you think that structure would look like below. Page | 58 Phospholipids Follow your teacher’s instructions to draw a “flirtatious” phospholipid and water droplet in the space provided. 1. Which part “likes” water? _________________________ What is this “like” for water called? ___________________ 2. Which part doesn’t like water? _________________________ What is this “dislike” for water called? ___________________ 3. How would a group of phospholipids arrange themselves if placed in water? Draw this type of arrangement below. Chemistry Unit Test Study Guide Page | 59 Chemistry Unit Test Study Guide What is the difference between an organic and inorganic compound? Is NaCl organic or inorganic? Is C6H12O6 organic or inorganic? What is dehydration synthesis? What is hydrolysis? How are monomers and polymers related? Is this an example of dehydration synthesis or hydrolysis? Is this an example of dehydration synthesis or hydrolysis? A lipid called triglyceride has what monomers? A lipid called phospholipids has what monomers? How is a saturated fat different from an unsaturated fat (in regards to the bonds between the carbon atoms in the fatty acid structure)? Page | 60 What particles are found in the nucleus of an atom? If an atom has more protons than electrons, it has a ___ charge. If an atom has more electrons than protons, it has a ___ charge. The smallest unit of matter is The smallest unit of life is What are ions and how are they formed? How do you find the number of protons in an atom? The number of neutrons? Electrons? How many electrons can the first orbital of an atom hold? Second? Third? For atoms to be “stable”, the orbitals must be… What is a covalent bond? What is an ionic bond? Page | 61 Page | 62 Biochemistry Unit Concept Map (The Concept Map has been started for you, only fill in the information on Lipids at the end of the Biochemistry Unit. The rest will be filled in by you as the information is covered at a later date.) Nucleic Acids Proteins Organic Compounds Carbohydrates Composed of the elements: Lipids Monomer or Building Blocks: Monomer Structure: Polymer or Important Groups: Functions of Macromolecule: Page | 63 Biochemistry Unit Concept Cards Page | 64 Parent/ Significant Adult Review Dear Parent/ Significant Adult: This Interactive Notebook represents your student’s learning to date and should contain the work your student has completed. Please take some time to look at the unit your student just completed, read his/ her reflection and respond to any of the following The work we found most interesting was ____________________________ because… What does the notebook reveal about your student’s learning habits or talents? My student’s biggest concern about this class is… Parent/ Significant Adult Signature: Comments? Questions? Concerns? Feel free to email: Mr. Duston at ryan.duston@venturausd.org Ms. Perez at darcy.perez@venturausd.org Page | 65 Biochemistry Unit Back Page The California State Standards I have come to use and understand are: How to relate the position of an element in the periodic table to its atomic number and atomic mass. How to use the periodic table to determine the number of electrons available for bonding. The nucleus of the atom is much smaller than the atom yet contains most of its mass. Atoms combine to form molecules by sharing electrons to form covalent bonds or by exchanging electrons to form ionic bonds. How to draw Lewis dot structures. How to use the pH scale to characterize acid and base solutions. The definitions of solute and solvent. Large molecules (polymers), such as proteins, nucleic acids, and starch, are formed by repetitive combinations of simple subunits. The bonding characteristics of carbon that result in the formation of a large variety of structures ranging from simple hydrocarbons to complex polymers and biological molecules. Most macromolecules (polysaccharides, nucleic acids, proteins, lipids) in cells and organisms are synthesized from a small collection of simple precursors. Page | 66 Cell Membrane and Transport Unit Student Design Cover Page (see guidelines on page 12) Page | 67 Cell Membrane and Transport Unit Front Page At the end of this unit, I will: Be familiar with different types of cell transport Work in cooperative groups to identify different methods of cell transport in different types of cells Roots, Prefixes and Suffixes I will understand are: Phospho-, bi-, trans-, -port, lyso-, -some, chloro-, -plast, -um (-a) Equi-, iso-, -tonic, hypo-, hyper-, macro-, phage The terms I can clearly define are: Selective permeability, phospholipid bilayer, transport protein, fluid mosaic model mixture, solution, solvent, solute, isotonic solution, hypotonic solution, hypertonic solution, diffusion, dynamic equilibrium, facilitated diffusion, osmosis, flaccid, turgid, plasmolysis Active transport, endocytosis, exocytosis, vesicle, macrophage The assignments I will have completed by the end of this unit are: Unit Cover Page Unit Concept Cards for terms underlined above Worksheets on Cellular Transport and Different Types of Solutions The Swell Cell Challenge Lab Real Life Application Jigsaw Activity Elodea Plasmolysis Lab Section Review Worksheet Unit Back Page Page | 68 WANTED The Cell Membrane (a.k.a., “The Plasma Membrane,” “Selectively Permeable Membrane” “Semipermeable Membrane,” “Phospholipid Bilayer” and “Fluid Mosaic Model”) Wanted for aiding and abetting certain molecules across the membrane. For this, the cell membrane has earned its alias, “’Selectively ‘ or ‘Semi’permeable Membrane.” Only certain molecules are allowed across the membrane, while others are not. Height: Between 3 and 10 nanometers. If you are on the lookout, be warned: You will need an electron microscope to see the membrane. Known Accomplices: The cell membrane was last seen surrounding every living cell. It is known to surround bacterial cells, as well as plant and animal cells. In addition to making up the outer membranes of cells, phospholipids surround every membrane-bound organelle. Identifying Features: The cell membrane primarily consists of Phospholipids, always arranged so that the ___________________________ (water-“fearing”) tails of the phospholipid face another tail from another phospholipid. The phospholipids are fairly slippery, and do not stick to neighboring phospholipids. This property gives a “plasma-like” or “fluid” appearance to the membrane. Embedded within the membrane (which is mostly made up of phospholipids) are cholesterols (making the membrane less fluid) and large proteins that help with many different functions. Carbohydrate side chains are also often found on the outer surface of the membrane. Page | 69 Cell Membrane Reading Comprehension Read the Wanted column on the previous page to answer the questions below. How did the cell membrane earn each of the following aliases? 1. Plasma membrane: 2. Selectively permeable membrane: 3. Semipermeable membrane: 4. Phospholipid bilayer: 5. Fluid mosaic model: Use the features listed below to identify the structures that are found in cell membranes: Phospholipid Carbohydrate side-chain Glycoprotein Membrane Protein Inside of Cell Outside of Cell Hydrophobic Region Hydrophilic Region Page | 70 Different Types of Cell Membrane Proteins TRANSPORTERS RECEPTORS ENZYMES SIGNAL/RECOGNITION Y 1. What are the four general types of proteins found anchored in the cell membrane? ________________,___________________, ___________________and ___________________ a. The _________________ protein identifies the cell type and to whom the cell belongs. b. _________________ found in cell membranes help speed up the rate of reactions. For instance, converting one substance into another. c. The _________________ protein receives information from outside the cell and passes it into the cell. d. The _________________ protein is a passageway through the cell membrane. Page | 71 What types of substances are “permitted” across the semipermeable membrane? Use the image below the table to help fill in this table. Check your answers with your teacher’s. Can Pass through Membrane Cannot Pass through Membrane Solubility? Size? Charge? Page | 72 Egg-Osmosis Demonstration In this demonstration, you will observe osmosis, the movement of water across a semipermeable membrane, from an area of higher concentration to an area of lower concentration. Sketch the set-up that your teacher demonstrates. After a minimum of 24 hours, sketch the results of the experiment, then reflect on why this happened by answering the follow up questions. Then, write an abstract of this demonstration. BEFORE AFTER 1. Is the water in the beaker hypotonic or hypertonic compared to the egg? 2. Is the egg hypertonic or hypotonic compared to the water in the beaker? 3. Did the water enter the egg? ____________ Why or why not? 4. Why is it important to use multiple eggs for this demonstration? Page | 73 Outline details of this demonstration that you need to write your abstract. Use the acronym below to assist you with your outline. I M R a C Based on the above outline, write your abstract below. ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ Page | 74 Cellular Transport: Different Types of Solutions Read the passage and then answer the questions that follow. Whenever the concentration of a dissolved substance is higher on one side of a membrane than on the other, there is a concentration gradient. Movement of water across the membrane depends on this concentration gradient. In osmosis water flows into and out of a cell until the concentration of water molecules is equal on each side of the cell membrane. At this point the flow of molecules into and out of the cell is in a state of equilibrium. In an isotonic solution the concentration of solute outside the cell is the same as that inside the cell. If a cell is placed in an isotonic solution, the rate of diffusion of water into the cell is exactly the same as the rate of diffusion of water out of the cell. In a hypotonic solution the concentration of solute outside the cell is lower than that inside the cell. Cells placed in a hypotonic solution swell up because water moves from the solution into the cell until the solutions inside and outside the cell are equal in concentration. In a hypertonic solution the concentration of the solute outside the cell is greater than that inside the cell. Cells placed in a hypertonic solution shrivel up and lose their shape because more water flows out of the cells than into them. Page | 75 The diagrams below represent the three types of solutions: isotonic, hypotonic, and hypertonic. 1. Indicate which type of solution is shown in each of the three diagrams above. A. _______________ B. _______________ C. _________________ 2. Which diagram, A, B, or C, shows no concentration gradient? ________ 3. Which diagram, A, B, or C, represents a situation in which the cell will decrease in size and lose its shape? _________ 4. In each of the diagrams, use an arrow to indicate if there is a net flow of water into or out of the cell. Page | 76 Cell Transport: Different Types of Solutions Use the activity on the previous page and your notes on page 258 – 260 to label each of the types of solutions in which the cells are submerged in below. A. ________________ B. C. This is a type of passive transport, but since it specifically is focused on the transport of water, what type of passive transport is this? Page | 77 What is an isotonic solution? • [Water] inside cell = [Water] outside cell • Cell is at ____________________ – Molecules are equally distributed in end The amount of water entering the cell = the amount of water leaving the cell • What is a hypotonic solution? What is a hypertonic solution? What direction does water flow? What can and can not pass through the membrane? Define selectively permeable • A solution that has___________ water, and ________ solute • The cell can lyse or burst if left in a ______________________ solution • A solution that has _________ water and _____________ solute • The cell will ____________________ a) Water ________________ flow at all. b) Water flows from an area of__________ concentration to an area of _____________ concentration. c) Water flows from an area of ________ concentration to an area of _______ concentration. CAN PASS THROUGH CAN NOT PASS THROUGH _____________________________________________________________ _____________________________________________________________ What are the 3 types of passive transport? What are 3 examples of active transport? Page | 78 What is a protein pump? _____________________________________________________________ _____________________________________________________________ What is endocytosis? _____________________________________________________________ _____________________________________________________________ What are the 2 types of endocytosis? What is exocytosis? _____________________________________________________________ _____________________________________________________________ Page | 79 Page | 80 Checking for Understanding Identify the images on the lines or symbols provided. The terms to fill in the blanks: Active Transport Facilitated Diffusion Passive Transport ATP (Cell Energy) Diffusion Page | 81 Warm-up: The following image follows the path of particles as they enter and leave the cell. Describe the image above using the vocabulary words: Endocytosis, exocytosis, plasma membrane and vesicle. Page | 82 The Swell Cell Challenge ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Purpose: To understand the types of solutions that will move across a semipermeable membrane. Hypothesis: If a cell is placed in a ____________________ solution, then the cell’s mass will increase. Prelab for “Diffusion through a Semipermeable Membrane” 1. Steps 1 and 2 in the procedure for this lab are instructions for making a dialysis tube (semi-permeable membrane) filled with glucose/starch. When you are finished making this tube, will it be flaccid (soft) or turgid (hard)?____________________ Why?____________________________________________________________ 2. In step 2 of the procedure, it tells you to rinse off the outside of the filled tube with water. Why do you think you are asked to do this? ________________________________________________________________ 3. In step 3 of the procedure, how will you test for the presence of glucose? ___________________________________________________________ 4. Iodine indicates the presence of what compound?_ ___________ Page | 83 5. Make a hypothesis for the 25-minute observations for the glucose/starch tube. Water (will or will not)___________diffuse into the dialysis tube filled with glucose and starch. Glucose (will or will not)_________________ diffuse from the tube into the water. Large molecules (will or will not) ______________________ diffuse across the dialysis tube and small molecules (will or will not) ___________ diffuse. Glucose is a (monosaccharide or disaccharide) _________________________. Starch (will or will not)_________________ diffuse from the tube into the water. Iodine (will or will not)___________diffuse into the dialysis tube filled with glucose and starch. Starch is a (monosaccharide or polysaccharide) _________________________. 6. What cell structure does the dialysis tubing represent in this experiment? _____________________________ 7. What is the function of this cell structure? ________________________________________________________________ Page | 84 The Swell Cell Challenge Flowchart Page | 85 The Swell Cell Challenge Materials: dialysis tubing I2KI solution string 100-mL Beaker 15% glucose/1% starch solution graduated cylinder Procedure: 1. Obtain a 15 cm piece of 2.5-cm dialysis tubing that has been soaking in water. Tie off one end of the tubing to form a bag. To open the other end of the bag, rub the end between your fingers until the edges separate. 2. Test the 15% glucose/1% starch solution in your graduated cylinder with glucose Testape. Record the results in Table 1. 3. Place 10 mL of the 15% glucose/1% starch solution in the bag. Tie off the other end of the bag leaving sufficient space for the expansion of the contents in the bag. Rinse the bag, pat it dry, and record the color of the solution in Table 1. Place the dried bag onto a balance to record the mass. Record this in Table 2. 4. Fill a 100-mL beaker two-thirds full with I2KI (potassium iodide + iodine) solution and record the color of the solution in Table 1. Test this solution with glucose Testape and record the results in Table 1. 5. Immerse the bag in the iodine solution in the beaker and record the time. 6. Allow your setup to stand for 5 minutes, then remove the tube to record the mass. To do this, rinse the tube and pat it dry. Record the mass in Table 2. Return the tube to the iodine solution and let it sit in the solution for five minutes. 7. Repeat step 6 five times. 8. Once you have finished recording the final mass of the tube, record the final color of the solution in the bag and of the solution in the beaker in Table 1. 9. Test the liquid in the beaker and in the bag with glucose Testape. Record the results in Table 1. Table 1 Initial Contents Bag Testape Results (Presence of Glucose) solution color 15% glucose & 1% starch Initial . Final . Initial Final . . Beaker H2O + IKI . . . Page | 86 Table 2 First Mass of bag (g) Second . Third . Fourth Fifth . Graph of Data Graph the data from Table 2 in the space provided below. Make sure to include a title and label your axes. Page | 87 Analysis of Results: 1. Which substance(s) are entering the bag and which are leaving the bag. What experimental evidence supports your answer? ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ 2. Explain the results you obtained. Include the concentration differences and membrane pore size in your discussion. ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ 3. Based on your observations, rank the following by relative size, beginning with the smallest: glucose molecules, water molecules, IKI molecules, membrane pores, starch molecules. ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ ___________________________________________________________________ Page | 88 Real Life Application of Cell Transport A report in the 23 April 1998 issue of The New England Journal of Medicine tells of the life-threatening complications that can be caused by an ignorance of osmosis. Large volumes of a solution of 5% human albumin (a protein in the blood) are injected into people undergoing a procedure called plasmapheresis. The albumin is dissolved in physiological saline (0.9% NaCl) and is therefore isotonic to human plasma (the large protein molecules of albumin have only a small osmotic effect). If 5% solutions are unavailable, pharmacists may substitute a proper dilution of a 25% albumin solution. Mixing 1 part of the 25% solution with 4 parts of diluent (a substance that something else is dissolved in) results in the correct 5% solution of albumin. BUT, in several cases, the diluent used was sterile water, not physiological saline. SO, the resulting solution was strongly hypotonic to human plasma. The Result: Massive, life-threatening hemolysis (releasing blood into the surrounding body fluids) in the patient. Draw a picture of what is happening in a patient’s cells in the space provided below, use arrows to show the net flow of water: Patient’s red blood cell in physiological saline Patient’s red blood cell in sterile water Based upon this, what do you think the symptoms of hemolysis would be? Page | 89 UNDERSTANDING DIFFUSION AND PERMEABILITY: PLASMOLYSIS ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ OBJECTIVE: To understand how materials move from areas of greater concentration to areas of lower concentration through diffusion, through a permeable membrane. MATERIALS: Microscope glass slide 2 coverslips 2 Elodea leaves water 6% salt solution HYPOTHESIS: If a freshwater leaf is placed in salt water, then water will flow (into or out of) the leaf cells. PROCEDURE 1. Prepare a wet mount of two elodea leaves on the same slide. Use 2 drops of tap water on one leaf, and 2 drops of salt water on the other leaf. Cover each with a cover slip. Make sure the two liquids on the slide do NOT run together. If they do, discard the leaves and start over. 2. Wait at least 3 minutes. Observe each under both low and high powers. Carefully observe the location of the chloroplasts (the small green structures) in relation to the cell wall of both leaves. Page | 90 3. Draw a diagram of the normal cell and the plasmolyzed cell in the circles below. Normal cell Plasmolyzed cell ANALYSIS: Read the following four statements before answering the questions: a. Elodea cells normally contain 1% salt and 99% water on the inside. b. Tap water used in this investigation contains 1% salt and 99% water. c. Salt water used in this investigation contains 6% salt and 94% water. d. Salt water has a higher concentration of salt than fresh water or Elodea cells. 1. Describe the location of chloroplasts in a normal Elodea cell (in tap water). _________________________________________________________________________ 2. Describe the location of chloroplasts in a plasmolyzed cell (in salt water). _________________________________________________________________________ Answer the following questions about the cell in tap water: 3. What is the percentage of water outside the cell? ______________ 4. What is the percentage of water inside the cell? ____________ Page | 91 Answer the following questions about the cell in salt water: 5. What is the percentage of water outside the cell at the start of this lab? ____________ 6. What is the percentage of water inside the cell at the start of this lab? ____________ 7. Which way did the water move, from inside the cell to outside, or from outside to inside the cell? _________________________________________________________________________ 8. Describe how the shape of the cell changed after being put in salt water. _________________________________________________________________________ 9. How would you define plasmolysis? _________________________________________________________________________ _________________________________________________________________________ Page | 92 Page | 93 Page | 94 Cell Membrane and Transport Unit Concept Cards (paste your envelopes for “Cell Transport” cards on this page) Page | 95 Cell Membrane and Transport Unit Concept Map (Instructions on page 12) Page | 96 Parent/ Significant Adult Review Dear Parent/ Significant Adult: This Interactive Notebook represents your student’s learning to date and should contain the work your student has completed. Please take some time to look at the unit your student just completed, read his/ her reflection and respond to any of the following The work we found most interesting was ____________________________ because… What does the notebook reveal about your student’s learning habits or talents? My student’s biggest concern about this class is… Parent/ Significant Adult Signature: Comments? Questions? Concerns? Feel free to email: Mr. Duston at ryan.duston@venturausd.org Ms. Perez at darcy.perez@venturausd.org Page | 97 Cell Membrane and Transport Unit Back Page The California State Standards I have come to use and understand are: Cells are enclosed within semipermeable membranes that regulate their interaction with their surroundings. 1. The functions of the nervous system and the role of neurons in transmitting electrochemical impulses. 2. The kidneys are responsible for the removal of nitrogenous wastes and the liver detoxifies blood maintains glucose balance. 3. Muscle contractions involve actin, myosin, Ca+2 , and ATP. Page | 98 Cell Structure and Function Unit Student Design Cover Page (See guidelines on page 12) Page | 99 Cell Unit Front Page At the end of this unit, I will: Have studied the similarities and differences between prokaryotic and eukaryotic cells Be familiar with the modes of cellular locomotion and movement Roots, Prefixes and Suffixes I will understand are: Phospho-, bi-, trans-, -port, lyso-, -some, chloro-, -plast, -um (-a) Equi-, iso-, -tonic, hypo-, hyper-, macro-, phage The terms I can clearly define are: Cell, cell theory, plasma membrane, organelle, eukaryotic cell, nucleus, prokaryotic cell Cytoplasm, cytoskeleton, ribosome, nucleolus, endoplasmic reticulum, Golgi apparatus, vacuole, lysosome, centriole, mitochondrion, chloroplast, cell wall, cilium, flagellum The assignments I will have completed by the end of this unit are: Unit Cover Page Unit Concept Cards for terms underlined above Complete the table of organelles TIME Organelle of the Year When Good Organelles Go Bad Cell Hyperlink Cooperative Group Project Cell City Analogy Cell Unit Back Page Page | 100 Cell Structure and Function Who was Robert Hoooke? ______________________________________________________________ Who coined the term “nucleus”? _______________________________________________________________ What is the cell theory? What did Rudolph Virchow discover? 1._______________________________________________________________ 2._______________________________________________________________ 3._______________________________________________________________ _______________________________________________________________ _______________________________________________________________ What does TEM stand for? _______________________________________________________________ _______________________________________________________________ What does SEM stand for? _______________________________________________________________ _______________________________________________________________ What are some differences between prokaryotic and eukaryotic cells? _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ Draw a picture of a prokaryotic cell and label it Draw a picture of a eukaryotic cell and label it Page | 101 Page | 102 Page | 103 Page | 104 Page | 105 Page | 106 Page | 107 Page | 108 Cell City Analogy In a far away city called Foothill City, the main export and production product is the steel widget. Everyone in the town has something to do with steel widget making and the entire town is designed to build and export widgets. The town hall has the instructions for widget making, widgets come in all shapes and sizes and any citizen of Grant can get the instructions and begin making their own widgets. Widgets are generally produced in small shops around the city, these small shops can be built by the carpenter's union (whose headquarters are in town hall). After the widget is constructed, they are placed on special carts which can deliver the widget anywhere in the city. In order for a widget to be exported, the carts take the widget to the post office, where the widgets are packaged and labeled for export. Sometimes widgets don't turn out right, and the "rejects" are sent to the scrap yard where they are broken down for parts or destroyed altogether. The town powers the widget shops and carts from a hydraulic dam that is in the city. The entire city is enclosed by a large wooden fence, only the postal trucks (and citizens with proper passports) are allowed outside the city. Match the parts of the city (underlined) with the parts of the cell. 1. Mitochondria ________________________________________ 2. Ribosomes ________________________________________ 3. Nucleus ________________________________________ 4. Endoplasmic Reticulum ________________________________________ 5. Golgi Apparatus 6. Protein _______________________________________ ________________________________________ 7. Cell Membrane ________________________________________ 8. Lysosomes ___________________________________________________ 9. Nucleolus ________________________________________ Page | 109 Page | 110 Page | 111 Page | 112 Page | 113 Page | 114 Cell Structure and Function Unit Concept Cards (paste your envelopes for “Cell” cards on this page) Page | 115 Cell Structure and Function Unit Concept Map (Instructions on page 12) Page | 116 Parent/ Significant Adult Review Dear Parent/ Significant Adult: This Interactive Notebook represents your student’s learning to date and should contain the work your student has completed. Please take some time to look at the unit your student just completed, read his/ her reflection and respond to any of the following The work we found most interesting was ____________________________ because… What does the notebook reveal about your student’s learning habits or talents? My student’s biggest concern about this class is… Parent/ Significant Adult Signature: Comments? Questions? Concerns? Feel free to email: Mr. Duston at ryan.duston@venturausd.org Ms. Perez at darcy.perez@venturausd.org Page | 117 Cell Structure and Function Unit Back Page The California State Standards I have come to use and understand are: How prokaryotic cells, eukaryotic cells (including those from plants and animals), and viruses differ in complexity and general structure. The role of the endoplasmic reticulum and Golgi apparatus in the secretion of proteins. How eukaryotic cells are given shape and internal organization by a cytoskeleton or cell wall or both. Page | 118 Cell Reproduction Unit Student Design Cover Page (see guidelines on page 12) Page | 119 Cellular Reproduction Unit Front Page At the end of this unit, I will: Know that cells go through a life cycle that includes interphase, mitosis, and cytokinesis. Roots, Prefixes and Suffixes I will understand are: Inter-, -phase, -kinesis, chromo- (chroma-), centro-, -mere, telo-, carcino-, -gen The terms I can clearly define are: Cell Cycle, Interphase, Mitosis, Cytokinesis, Chromosome, Chromatin Prophase, Sister Chromatid, Centromere, Spindle Apparatus, Metaphase, Anaphase, Telophase Cyclin, cyclin-dependent kinase, cancer, carcinogen, apoptosis, stem cell The assignments I will have completed by the end of this unit are: Unit Cover Page Unit Concept Cards for terms underlined above Gelatin Cube Wet Lab and Questions Mitosis Flip Book Mitosis Timing Computer Lab Mitosis Microscope Observation Lab “Your Body is Younger than You Think,” Article and Reading Comprehension Cell Reproduction Unit Back Page Page | 120 Diffusion and Cell Size ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Purpose: To determine why cells are small and what role diffusion has in cell size. Background: Cells from different or organisms vary greatly in size from one another. Eukaryotic cells tend to be larger than prokaryotic ones. For example, the largest known cell is a giraffe nerve cell (see below) and the smallest known cells are bacteria (prokaryotes). Page | 121 To give you an even better idea of variations in cell size, look at the “length-line” below: In this lab, you will investigate how the size of a cell is related to its ability to get molecules and other substances in and out. Using different sized cubes of special agar (a gelatin-like substance), you will measure the amount of sodium hydroxide diffusion (NaOH) that occurs in each cube. The agar has phenolphthalein mixed into it. Phenolphthalein is an indicator for bases and will turn pinkish purple in the presence of the base NaOH. By looking at the color change, you will be able to determine how much diffusion has occurred. Page | 122 Gelatin Cube Wet Lab Pre-lab questions: 1. Define Surface area in your own words. 2. Define Volume in your own words. Overview of Activity: Agar cubes of various sizes have been prepared with NaOH (an acid or base?) and phenolphthalein (a pH indicator). The cubes were initially a pink color due to the phenolphthalein. They will soak in a dilute HCl solution for 10 minutes before students remove the cube from the solution, and slice the cube in half. Analysis Questions 1. Based on your understanding of random motion, explain or sketch what the clearing line in the gel actually looks like if we could see the particles at the molecular level. 2. In the graphic to the right of two different size spheres, approximately how many times larger are the radii, the surface areas and the volumes? Connect the graphic to what you have learned from the experiment. Page | 123 3. Bryophytes are a phylum of the plant kingdom that lacks a vascular system. They have no specialized tubes for transporting water and organic products of photosynthesis. Instead, they rely upon diffusion. Examples of bryophytes are the mosses. Explain why mosses cannot grow tall. 4. The following image is a representational graphic of an idealized small intestinal cell. What unusual feature do you notice about this cell? One of the functions of the intestinal cell is to allow the passage of digested nutrients from the interior of the small intestine to the blood capillaries. One of the principles of biology is: “Form follows function.” Explain how this is illustrated in the intestinal cell. 5. What are some other examples of structures within the circulatory, respiratory, excretory systems, which have extensive branching to increase diffusionary surface area? Page | 124 Warm-up: Designing a Cell with a Large Surface Area to Volume Ratio Notice how both cells (above) have the same surface area to volume ratio. Cells have many adaptations that increase the surface area to volume ratio. In the space provided below, design a cell with a high surface area to volume ratio. Include the organelles, as it has to be functional! Page | 125 The Cell Cycle Page | 126 Mitosis Fill in the Blank Notes What are the stages of mitosis? _______________________________________________________________ What is the problem _______________________________________________________________ with a low surface to _______________________________________________________________ volume ratio? What are the key roles of cell division? _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ Fill in the blank DNA molecules are packed into _____________________________________. What are gametes? _______________________________________________________________ _______________________________________________________________ What are somatic cells? What are chromosomes made of? What are histones? What are nucleosomes? What does a centromere do? _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ Page | 127 Describe Prophase _______________________________________________________________ _______________________________________________________________ Describe Metaphase _______________________________________________________________ _______________________________________________________________ Describe Anaphase _______________________________________________________________ _______________________________________________________________ Describe Telephase _______________________________________________________________ _______________________________________________________________ What happens in cytokinesis? What is the end product? _______________________________________________________________ _______________________________________________________________ Page | 128 Page | 129 Page | 130 Page | 131 Page | 132 ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Page | 133 Page | 134 Observing Mitosis in Onion Root Tips Rapid cell division occurs in rapidly growing regions of organisms, like plant roots. In this lab, you will observe and record cells at various stages of mitosis within preserved onion root tips. Use the images to help you and your lab partner find the stages of mitosis in your slide. Record your observations in the space provided. In the space provided, use your notes to fill in the details of each stage of mitosis. Image Observation What’s Happening during this phase? Interphase Prophase Metaphase Anaphase Telophase Page | 135 Your Body Is Younger Than You Think by Nicholas Wade New York Times, August 2, 2005 Whatever your age, your body is many years younger. In fact, even if you're middle aged, most of you may be just 10 years old or less. This heartening truth, which arises from the fact that most of the body's tissues are under constant renewal, has been underlined by a novel method of estimating the age of human cells. Its inventor, Jonas Frisen, believes the average age of all the cells in an adult's body may turn out to be as young as 7 to 10 years. But Dr. Frisen, a stem cell biologist at the Karolinska Institute in Stockholm, has also discovered a fact that explains why people behave their birth age, not the physical age of their cells: a few of the body's cell types endure from birth to death without renewal, and this special minority includes some or all of the cells of the cerebral cortex. It was a dispute over whether the cortex ever makes any new cells that got Dr. Frisen looking for a new way of figuring out how old human cells really are. Existing techniques depend on tagging DNA with chemicals but are far from perfect. Wondering if some natural tag might already be in place, Dr. Frisen recalled that the nuclear weapons tested above ground until 1963 had injected a pulse of radioactive carbon 14 into the atmosphere. Breathed in by plants worldwide and eaten by animals and people, the carbon 14 gets incorporated into the DNA of cells each time the cell divides and the DNA is duplicated. Most molecules in a cell are constantly being replaced but the DNA is not. All the carbon 14 in a cell's DNA is acquired on the cell's birth date, the day its parent cell divided. Hence the extent of carbon 14 enrichment could be used to figure out the cell's age, Dr. Frisen surmised. In practice, the method has to be performed on tissues, not individual cells, because not enough carbon 14 gets into any single cell to signal its age. Dr. Frisen then worked out a scale for converting carbon 14 enrichment into calendar dates by measuring the carbon 14 incorporated into individual tree rings in Swedish pine trees. Having validated the method with various tests, he and his colleagues have reported in the July 15 issue of Cell the results of their first tests with a few body tissues. Cells from the muscles of the ribs, taken from people in their late 30's, have an average age of 15.1 years, they say. The epithelial cells that line the surface of the gut have a rough life and are known by other methods to last only five days. Ignoring these surface cells, the average age of those in the main body of the gut is 15.9 years, Dr. Frisen found. The Karolinska team then turned to the brain, the renewal of whose cells has been a matter of much contention. Prevailing belief, by and large, is that the brain does not generate new neurons after its structure is complete, except in two specific regions, the olfactory bulb that mediates the sense of smell, and the hippocampus, where initial memories of faces and places are laid down. This consensus view was challenged a few years ago by Elizabeth Gould of Princeton, who reported finding new neurons in the cerebral cortex, along with the elegant idea that each day's memories might be recorded in the neurons generated that day. Dr. Frisen's method will enable all regions of the brain to be dated to see if any new neurons are generated. So far he has tested only cells from the visual cortex. He finds these are exactly the same age as the Page | 136 individual, showing that new neurons are not generated after birth in this region of the cerebral cortex, or at least not in significant numbers. Cells of the cerebellum are slightly younger than those of the cortex, which fits with the idea that the cerebellum continues developing after birth. Another contentious issue is whether the heart generates new muscle cells after birth. The conventional view that it does not has recently been challenged by Dr. Piero Anversa of the New York Medical College in Valhalla. Dr. Frisen has found the heart as a whole is generating new cells, but he has not yet measured the turnover rate of the heart's muscle cells. Although people may think of their body as a fairly permanent structure, most of it is in a state of constant flux as old cells are discarded and new ones generated in their place. Each kind of tissue has its own turnover time, depending in part on the workload endured by its cells. The cells lining the stomach, as mentioned, last only five days. The red blood cells, bruised and battered after traveling nearly 1,000 miles through the maze of the body's circulatory system, last only 120 days or so on average before being dispatched to their graveyard in the spleen. The epidermis, or surface layer of the skin, is recycled every two weeks or so. The reason for the quick replacement is that "this is the body's saran wrap, and it can be easily damaged by scratching, solvents, wear and tear," said Elaine Fuchs, an expert on the skin's stem cells at the Rockefeller University. As for the liver, the detoxifier of all the natural plant poisons and drugs that pass a person's lips, its life on the chemical-warfare front is quite short. An adult human liver probably has a turnover time of 300 to 500 days, said Markus Grompe, an expert on the liver's stem cells at the Oregon Health & Science University. Other tissues have lifetimes measured in years, not days, but are still far from permanent. Even the bones endure nonstop makeover. The entire human skeleton is thought to be replaced every 10 years or so in adults, as twin construction crews of bone-dissolving and bone-rebuilding cells combine to remodel it. About the only pieces of the body that last a lifetime, on present evidence, seem to be the neurons of the cerebral cortex, the inner lens cells of the eye and perhaps the muscle cells of the heart. The inner lens cells form in the embryo and then lapse into such inertness for the rest of their owner's lifetime that they dispense altogether with their nucleus and other cellular organelles. But if the body remains so perpetually youthful and vigorous, and so eminently capable of renewing its tissues, why doesn't the regeneration continue forever? Some experts believe the root cause is that the DNA accumulates mutations and its information is gradually degraded. Others blame the DNA of the mitochondria, which lack the repair mechanisms available for the chromosomes. A third theory is that the stem cells that are the source of new cells in each tissue eventually grow feeble with age. "The notion that stem cells themselves age and become less capable of generating progeny is gaining increasing support," Dr. Frisen said. He hopes to see if the rate of a tissue's regeneration slows as a person ages, which might point to the stem cells as being what one unwetted heel was to Achilles, the single impediment to immortality. Page | 137 Your Body Is Younger Than You Think Reading Comprehension 1. Which parts of your body are the “youngest” (they are replaced more than the other parts)? 2. Which parts of your body are the “oldest” (they are not replaced often or at all)? 3. What do you think we would be like if the brain divided as frequently as the liver? 4. This article addresses normal cell division in a human body. How would you refer to abnormal cell division in an organism? 5. Chemotherapy is treatment that cancer patients undergo. It kills cells, and prevents cells from dividing, hopefully killing the cancerous cells in the patient’s body. Why do you think a person’s hair falls out when they are undergoing chemotherapy to treat cancer? Why do you think nausea is another common side effect? Page | 138 Cellular Reproduction Unit Concept Cards (paste your envelopes for “Cellular Reproduction” concept cards on this page) Page | 139 Cellular Reproduction Unit Concept Map (Instructions on page 12) Page | 140 Parent/ Significant Adult Review Dear Parent/ Significant Adult: This Interactive Notebook represents your student’s learning to date and should contain the work your student has completed. Please take some time to look at the unit your student just completed, read his/ her reflection and respond to any of the following The work we found most interesting was ____________________________ because… What does the notebook reveal about your student’s learning habits or talents? My student’s biggest concern about this class is… Parent/ Significant Adult Signature: Comments? Questions? Concerns? Feel free to email: Mr. Duston at ryan.duston@venturausd.org Ms. Perez at darcy.perez@venturausd.org Page | 141 Cellular Reproduction Unit Back Page The California State Standards I have come to use and understand are: Cells are enclosed within semipermeable membranes that regulate their interaction with their surroundings. Cells divide to increase their numbers through a process of mitosis, which results in two daughter cells with identical sets of chromosomes. Page | 142 Cellular Energy Unit Student Design Cover Page (see page 12 for guidelines) Page | 143 Cellular Energy Unit Front Page At the end of this unit, I will: Understand that Photosynthesis converts the Sun’s energy into chemical energy Understand that cellular respiration uses chemical energy to carry out life functions. Roots, Prefixes and Suffixes I will understand are: Thermo-, -dynamics, tri- an-, aero-, glyco- The terms I can clearly define are: Energy, Thermodynamics, Metabolism, Photosynthesis, Cellular Respiration, Adenosine Triphosphate (ATP) Thylakoid, Granum, Stroma, Pigment, NADP+, Calvin Cycle, Rubisco Anaerobic process, Aerobic respiration, Glycolysis, Krebs Cycle, Fermentation The assignments I will have completed by the end of this unit are: Unit Cover Page Unit Concept Cards for terms underlined above Tasty Model Activity Photosynthesis Storyboard Activity Floating Leaf Disc Assay: Measuring the Rate of Photosynthesis Lab Are Photons Needed for Photosynthesis? Lab Cellular Respiration: An Introduction Worksheet Cellular Respiration in Seeds Lab A “Swell” Activity Lab Observed or Performed in the Cellular Respiration Play Cellular Energetics Unit Back Page Page | 144 Tasty Models: Carbohydrates ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Understanding the arrangement of atoms within nutrient molecules often helps explain their chemical behavior, health attributes and role in weight balance. Although atomic representations do not accurately represent electron configurations, they can be used to show the arrangement of nuclei and bond type. In the following activities, you'll construct several different molecular models that represent substances that play an essential role in our nutritional needs. Carbohydrates Carbohydrates are a group of nutrients that include sugars and starches. Perhaps, the most familiar carbohydrate building block is glucose. Glucose is a monosaccharide, which means that it contains one sugar unit. Monosaccharides can be joined together to produce larger chains of carbohydrates. Starch is an example of a long chain of sugar molecules that are linked together. In this set of activities, you'll construct a single sugar (glucose) and observe the effects of a dehydration synthesis reaction. Materials Marshmallows Toothpicks Page | 145 Glucose Model 1. Examine the assortment of marshmallows that you will be using to assemble your molecular models. Now, consider the formula of glucose, C6H12O6. Based on this formula, how should you assign specific colors to the component atoms? Which atom will be represented with the most numerous color? 2. To build the ring version of glucose, let's construct a closed ring formed by five carbon atoms and one oxygen atom. 3. Now, let's add the sixth carbon atom. It is attached to the ring carbon that is immediately to the left of the oxygen atom. 4. The remaining five oxygen atoms are part of hydroxyl (OH) groups. They are added as shown here. 5. Finish the model by adding the remaining seven hydrogen atoms so that each carbon atom forms four bonds. Page | 146 Modeling Dehydration Synthesis: Making Polysaccharides To produce larger carbohydrate molecules, glucose is linked to other sugar molecules. During this bonding process, two atoms of hydrogen and one atom of oxygen are removed from the linking sugars. These atoms join together to produce a molecule of water. Hence, this type of sugar bonding is called dehydration synthesis. 1. Find another group with a completed glucose model. 2. Place both models side-by-side. Remove the two hydrogen atoms and one oxygen atom that are associated with dehydration synthesis (identified by the dashed line). 3. Join the free bond of the ring oxygen atom to the free bond of the carbon atom. Join the three removed atoms together to form a molecule of water. Questions 1. What type of molecule is removed during dehydration synthesis? ________________________________________________________ ___________________________________________________________ 2. What happens to the "open" bonds that are created as neighboring sugar molecules lose component atoms? 3. What might happen if you added water to a starch molecule? 4. When your group had just one glucose molecule, the ratio of Carbon to Hydrogen to Oxygen was 1:2:1. Was this ratio maintained when your group polymerized with another glucose molecule? What about three or more? Explain your observation. Page | 147 Carbohydrates Warm-up: Refer to the “Tasty Models” Activity to help you fill in the following. 1. Which CHONPS elements are included in Carbohydrates? ______________, ________________, and ___________________. 2. In what ratio are these elements found? ___ : ___: ___ 3. What is an example of a Carbohydrate monomer? 4. What is an example of a Carbohydrate polymer? 5. Why do you think Carbohydrates are important in a person’s diet? Page | 148 Carbohydrates 1. Carbohydrates are: – an important ________________________________ – Cellular _________________________ 2. Carbon, Hydrogen and Oxygen in a ratio of 1:2:1 3. General Formula ____________________ Types of Carbohydrates Monosaccharides (simple sugars) – Contain 3-7 Carbons each • Examples: ___________________________________________ Disaccharides (two sugars) • Examples: _____________________________________________ – Maltose = Glucose + Glucose – Lactose = Glucose + Galactose Page | 149 Page | 150 Energy Warm-up 1. Reflect on Energy Cycles (e.g., Carbon Cycle) from the beginning of the year. Are there places in these cycles in which energy can be created from nothing or completely lost in the cycle? Explain why or why not. 2. Refer to page 218 from your textbook to review the Laws of Thermodynamics. Restate them in your own words below. 1. First Law of Thermodynamics: 2. Second Law of Thermodynamics: 3. Can cells use any form of energy available? ____________ 1. What type of cells might use energy from the sun? 2. If a cell can’t use energy from the sun, what form of energy must they use? Explain. Page | 151 Page | 152 Page | 153 Photosynthesis Song I’m a little plant that grows and grows Photosynthesis is what I know Energy, 6 waters, & 6 CO2 Help me make glucose and 6 O2 Page | 154 Cell Energy and ATP Review The following is a diagram of an ATP molecule. Use the diagram to answer the questions that follow. 1. What does the “A” in ATP stand for? ________________ 2. What two molecules are used to build adenosine? ________________ and __________ 3. Adenine is a nitrogenous base and ribose is a 5-carbon sugar. 4. What does the “T” in ATP stand for? ________________ 5. What does the “P” in ATP stand for? ________________ 6. How many phosphate groups, P , are present in one ATP molecule? _____ 7. What is the complete name of ATP? 8. What is released when the chemical bonds between the last two phosphates of an ATP molecule are broken? Page | 155 1 2 3 4 8 5 7 6 8 Page | 156 Page | 157 Page | 158 Page | 159 Chlorophyll: The Photosynthetic Pigment According to the image of chlorophyll’s absorption below, why are leaves green? Page | 160 Page | 161 Page | 162 Page | 163 Page | 164 Photosynthesis: The Big Picture Fill in the spaces below with the chemical formula for Photosynthesis. The numbers link to clues listed below: 1. 2. + 4. 3. 5. + 1. This reactant is a critical part of the Carbon Cycle. It is released by heterotrophs (and autotrophs!) during respiration, and taken up by plants and algae during Photosynthesis. 2. This reactant is the fundamental component of the Water Cycle. Plants need this molecule for photosynthesis, while this is given off during respiration, it is also released by plants during a process called transpiration. 3. This is the catalyst that drives the entire reaction of photosynthesis. This is a form of energy that supports almost all life forms on Earth. This is the “photo” part of photosynthesis. 4. This product is the “synthesis” part of photosynthesis. This product is chemical energy that may go on to be consumed by a heterotroph. This is an organic compound, as it contains which element? __________________ 5. This product is often considered to be a waste product of photosynthesis, but we couldn’t be here without it. Page | 165 Photosynthesis Storyboard Page | 166 Photosynthesis Storyboard Activity In groups, you will begin the brainstorming process of creating a storyboard or cartoon of Photosynthesis. Make sure to check your sample storyboard (on the white-erase board) with your teacher prior to creating the final product. The guidelines are as follows: 1. There must be at least six “boards” or “squares.” a. Three of which must be dedicated to the light reaction b. Three must be dedicated to the Calvin Cycle. 2. The following terms must be used either in the image or in the caption of the storyboard: a. Photon b. Electron c. Water-splitting d. Oxygen e. ATP f. NADPH g. Light Reaction h. Calvin Cycle i. PGAL j. Glucose k. CO2 fixation l. Thylakoid m. Stroma 3. The following guidelines will be used when grading your Storyboard, so use the following as a checklist to make sure that your storyboard does the following: a. Underline the terms (a through m, listed above) as they are mentioned in the caption or in the cartoon. b. Label structures c. 4 or more colors must be used d. It must be neat. e. There must be at least six slides. Page | 167 The Floating Leaf Disk Assay for Investigating Photosynthesis ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Flowchart Page | 168 The Floating Leaf Disk Assay for Investigating Photosynthesis The biology behind the procedure: Leaf disks float, normally. When the air spaces are infiltrated with solution, the overall density of the leaf disk increases and the disk sinks. The infiltration solution includes a small amount of Sodium bicarbonate (NaHCO3). In this lab, the Bicarbonate ion (HCO3-) serves as the carbon source for photosynthesis. As photosynthesis proceeds, oxygen is released into the interior of the leaf which changes the buoyancy--causing the disks to rise. Since cellular respiration is taking place at the same time, consuming oxygen, the rate that the disks rise is an indirect measurement of the net rate of photosynthesis. Question: Will the leaf discs sink or rise when exposed to CO2 solution? Hypothesis: If ___________________________, then leaf discs should ___________because Materials: Sodium bicarbonate (Baking soda) Liquid Soap Plastic syringe (10 cc or larger) Leaf material Hole punch or straw Beakers Timer Light source Page | 169 Procedure: 1. Prepare 150 ml of bicarbonate solution for each trial. o The bicarbonate serves as an alternate dissolved source of carbon dioxide for photosynthesis. Prepare a 0.2% solution. (This is not very much it is only about 1/16 of a teaspoon of baking soda in 150 ml of water.) o Add 0.5 mL of liquid soap to this solution. The soap wets the hydrophobic surface of the leaf allowing the solution to be drawn into the leaf. It’s difficult to quantify this since liquid soaps vary in concentration. Avoid suds. If your solution generates suds then dilute it with more bicarbonate solution. 2. Cut 10 or more uniform leaf disks for each trial. o o o Single hole punches work well for this but stout plastic straws will work as well. Choice of the leaf material is perhaps the most critical aspect of this procedure. The leaf surface should be smooth and not too thick. Avoid plants with hairy leaves. We will use fresh spinach leaves. Avoid major veins. 3. Infiltrate the leaf disks with sodium bicarbonate solution. o o Remove the piston or plunger and place the leaf disks into the syringe barrel. Replace the plunger being careful not to crush the leaf disks. Page | 170 o o o o Push on the plunger until only 1mL of air and leaf disk remain in the barrel (< 10%). Pull 9mL of sodium bicarbonate solution into the syringe. Tap the syringe to suspend the leaf disks in the solution. Holding a finger over the syringe-opening, draw back on the plunger to create a vacuum. Hold this vacuum for about 10 seconds. While holding the vacuum, swirl the leaf disks to suspend them in the solution. Let off the vacuum. The bicarbonate solution will infiltrate the air spaces in the leaf causing the disks to sink. You will probably have to repeat this procedure 2-3 times in order to get the disks to sink. If you have difficulty getting your disks to sink after about 3 evacuations, it is usually because there is not enough soap in the solution. Add a few more drops of soap. 4. Pour the disks and solution into the solution remaining in the beaker. 5. For a control infiltrate leaf disks with a solution of only water with a drop of soap--no bicarbonate. 6. Place under the light source and start the timer. At the end of each minute, record the number of floating disks. Then swirl the disks to dislodge any that are stuck against the sides of the beakers. Continue until all of the disks are floating. Page | 171 Results Your Lab Group Number will correspond to the group number in the table. Fill in the Experimental Group Data in the column “E” and your Control Group Data in the column “C.” Number of discs floating Time (minutes) Group 1 Group 2 Group 3 Group 4 Group 5 Group 6 Group 7 Group 8 Group 9 Group Average E E E E E E E E E E C C C C C C C C C C 0 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 Page | 172 Graph: Title Page | 173 Conclusion: Answer in complete sentences 1. Did the results confirm or reject your initial hypothesis? Why or why not? _________________________________________________________________ _______________________________________________________________ 2. What gasses are being consumed and produced in photosynthesis? _________________________________________________________________ _________________________________________________________________ 3. How do the gasses consumed and produced in photosynthesis affect the buoyancy of the leaf? _________________________________________________________________ 4. Identify the following elements of experimental design in this experiment: a. Independent variable: __________________________________________________ ___________________________________________________________ b. Dependent variable: ___________________________________________________ ___________________________________________________________ c. Control group: ________________________________________________________ ___________________________________________________________ d. Experimental group: ___________________________________________________ ___________________________________________________________ e. Identify three constants: _______________________________________________ ___________________________________________________________ 5. What possible errors may have affected the results? (Identify at least one.) _________________________________________________________________ _________________________________________________________________ ____________________ Page | 174 6. Briefly explain how this experiment could be changed to test a different variable that may impact the rate of photosynthesis. _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ _________________________________________________________________ Page | 175 Photosynthesis Review 1. Ultimately, where does most of the energy come from for life on Earth? __________ 2. The energy in sunlight cannot be used directly by most living cells. How does radiant energy from the sun become ATP in a cell? 3. Green plants and some protists capture the energy in sunlight and use it to convert ____________ _____________ and _________ chemical energy in ___________________ _ into __, such as glucose. a. What is this process called? _________________________ b. In what cell organelle does this process occur? ______________________ 4. Cells can then use the process of _______________ _______________ to breakdown carbohydrates, such as glucose, and to convert the chemical energy into ATP. Use the terms below to fill in the graphic organizer. Hint: Energy is moving in this, and is represented with arrows. Photosynthesis Sun Glucose (Stored Energy) Radiant Energy ATP (Usable Energy) Cellular Respiration Page | 176 5. What is the relationship between photosynthesis and cellular respiration?________________________________________________ ___________________________________________________________ ___________________________________________________________ ___________________________________________________________ 6. What must be consumed by an organism undergoing cellular respiration? and 7. What is produced by an organism undergoing cellular respiration? and Page | 177 Cellular Respiration Warm-up: 1. Maria and Anthony both accidentally eat something containing a poison that blocks enzymes in the liver that break down lactic acid. Maria runs for help and Anthony sits down and starts crying. Who will have the lower blood pH? Who will experience the effects of the poison first? Explain your answers. 2. A drug that inhibits mitochondrial function would block which steps in cellular respiration? How would the cell generate ATP? What do you think the sideeffects of such a drug would be? 3. An apparatus was used to collect gases released by the freshwater plant, Elodea. In darkness, a gas is still collected in the test tube. Would this be the same gas or a different gas than the one collected under light conditions. If it is different what is the gas? Page | 178 Introduction One of the characteristics of life that you learned about in the beginning of this class was metabolism. In this worksheet we will be exploring the details of metabolism, and we will build on our new understanding of biological chemistry in order to comprehend how the body – and in particular the cell – is able to take complex molecules (like are in our food), and break them down, and then use them to build new structures, and generate energy to do work. Many students find it helpful to think of metabolism in a cell as an assembly line in a factory with different machines having different roles in the process. Activity B. Match the following. Write down the numbers in the space provided. Page | 179 Use the diagram of Cellular Respiration below to identify which process is responsible for the following, by writing “Glycolysis,” “Krebs Cycle,” or “Electron Transport Chain” next to each description. Fill in the “star” shapes with “ATP.” KREBS CYCLE H2O O2 1. Breaks up glucose into pyruvate (or pyruvic acid): 2. This is responsible for the release of CO2: 3. According to the images, which one of these do you think produces the most ATP? 4. This step uses Oxygen: 5. What do Glycolysis and the Krebs Cycle contribute to the Electron Transport Chain? and Page | 180 Page | 181 Page | 182 Page | 183 Page | 184 Page | 185 Page | 186 Page | 187 Page | 188 Page | 189 Tying things together Label this overview of Photosynthesis and Respiration together to see how these metabolic processes are interrelated. Page | 190 Cellular Respiration in Seeds Lab ABSTRACT: _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ _____________________________________________________ Page | 191 Cellular Respiration in Seeds Lab In cellular respiration, the glucose produced during photosynthesis must be broken down so that the energy trapped in the glucose molecule can be changed into ATP, a form of energy that can be used by the cell. In this laboratory investigation, you will be examining cellular respiration in the following three set-ups: Use only black beans. a) germinating seeds (seeds that are sprouting) b) dry seeds (seeds that are not sprouting but are dormant or “asleep”) c) glass beads. To test cellular respiration, a chemical called soda lime will be used. Remember that oxygen is used by the seeds during cellular respiration, and carbon dioxide is released. Soda lime absorbs the carbon dioxide gas that is released by the seeds. The more cellular respiration occurs, the more carbon dioxide gas is absorbed by the soda lime. When a test tube with the seeds and soda-lime are placed upside down in a beaker of water, water will move into the test tube when there is more respiration going on. Pre-lab Questions: (Answer these questions in the space provided) 1. What is the equation for cellular respiration? ___________________________________ 2. According to the equation, what materials are being used during cellular respiration? (In other words, what are the reactants?) ________________________________________ 3. According to the equation, what materials are being made during cellular respiration? (In other words, what are the products?) _________________________________________ 4. What chemical is used in this lab to absorb the carbon dioxide released by the seeds during cellular respiration? ______________________________________________________ Page | 192 Cellular Respiration in Seeds Lab Procedure Flowchart Page | 193 Problem: In which set-up will cellular respiration be the greatest: the germinating seeds, the dry dormant seeds, or the glass beads? Hypothesis: If the test tube contains ___________________________________, then cellular respiration will be the greatest. Procedure: (Read these procedures, then create a flow-chart on the left side of your Interactive Notebook for each of the steps below.) 1. Fill three test tubes with approximately 1 cm of soda lime. 2. Place a small cotton plug into each of the test tubes (the cotton plug serves to keep the materials in the tube when the tube is inverted.) 3. Place 10 germinating seeds in one test tube, 10 dry, dormant seeds in another test tube, then 10 glass beads in the last test tube. 4. Place another cotton plug on top of the seeds. 5. Use a rubber band to tie the four test-tubes together. 6. Fill a beaker with 1.5 centimeters of water. 7. Invert the test tubes into the beaker of water. Be careful to record the initial water line. 8. Wait 24 hours and record how much water entered each test tube in millimeters. Diagram: The set-up below will guide you as you set-up your experiment. Tube 1 Tube 2 Tube 3 Soda lime powder Cotton plug Seeds Cotton plug Ruler Rubber band Air space within tube Beaker with water Page | 194 Results: Experimental Set-up Germinating Seeds Dry, dormant Seeds Glass Beads Height of water inside test tube in mm Post-lab questions: 1. In this lab, what evidence do you look for that proves that cellular respiration is occurring? _____________________________________________________________ 2. Of the three set-ups, which is the control group? _____________________ Which were the experimental group(s)? _____________________________ _____________________________________________________________ 3. What was the independent variable? (Hint…What condition that differed in the three test-tubes?) _____________________________________________________________ 4. What was the dependent variable? (Hint…What changes are you measuring as a result of the experiment?) __________________________________________________________ 5. Which set-up demonstrated the most respiration occurring? Why? _____________________________________________________________ _____________________________________________________________ Page | 195 ACT I Glycolysis Participants and their Props: Narrator: Reads setting ATP/ADP 1: Magnet Board with ADP compound and one phosphate Glycolysis 1: Large Magnet Board with Glucose Glycolysis 2: Two small magnet boards with phophates, electrons Glycolysis 3: Two NAD+ buckets and and 2H stickers Glycolysis 4: no props Glycolysis 5: Pyruvate Magnet Krebs Cycle Bouncer: ACT II: Entering the Mitochondrion Participants and Their Props Krebs Cycle Bouncer 1: Acetyl-CoA Magnet Krebs Cycle Bouncer 2 One orange NAD+ bucket and H sticker ACT III: The Krebs Cycle Participants and Their Props: Krebs Cycle 1: magnet board with a 4 Carbons on it Krebs Cycle 2: Citric Acid Magnet Krebs Cycle 3: 2 NAD buckets, and 2 H stickers Krebs Cycle 4: no props Krebs Cycle 5: FAD bucket and 2H stickers Krebs Cycle 6: 1 NAD bucket and H sticker ATP/ADP: Magnet board with ADP Page | 196 ACT IV: The Electron Transport Chain Participants and Their Props: Glycolysis 3: 2 NADH buckets Krebs Cycle Bouncer 2: NADH bucket Krebs Cycle 3: NADH bucket Krebs Cycle 6: NADH bucket Krebs Cycle 5: FADH2 bucket Oxygen: Oxygen bucket with ATP sign and Pipette with water ETC: ETC ramp ATP/ADP 1, 2, 3: ATP magnets Page | 197 Page | 198 Page | 199 Page | 200 Page | 201 Page | 202 Page | 203 Page | 204 Page | 205 ACT IV: The Electron Transport Chain Participants and Their Props: Glycolysis 3: 2 NADH buckets Krebs Cycle Bouncer 2: NADH bucket Krebs Cycle 3: NADH bucket Krebs Cycle 6: NADH bucket Krebs Cycle 5: FADH2 bucket Oxygen: Oxygen bucket with ATP sign and Pipette with water ETC: ETC ramp ATP/ADP 1, 2, 3: ATP magnets (Narrator Steps Forward and ATP/ADP 1 – 3 prepare to enter stage. Krebs Cycle-5 continues searching for Electron Transport Chain while narrator speaks…) Narrator: “So far we’ve been successful at making a few ATPs. But there isn’t enough ATP to run all cellular activity.” ATP/ADP 1 – 3: (shows off ATP magnet boards and smile) Narrator: “The electrons from glucose that are now carried in FADH2 and NADH must be converted to additional ATP.” (Glycolysis 3, Krebs Cycle Bouncer 2, Krebs Cycle 3, and Krebs Cycle 6 all bump into Krebs Cycle 5 while he is searching for the ETC) Krebs Cycle 5: “Hey are you guys looking for the ETC?” Krebs Cycle 6: “Yeah, man! I’ve been carrying this electron around for a while. I’ll be glad to get rid of it. It’s so full of energy that I can barely contain it!” Glycolysis-3: “Well at least you haven’t been carrying your electrons around since Glycolysis!” Krebs Cycle-3: “I heard that the ETC will take the electrons from us. We can definitely lighten our load!” Page | 206 Krebs-Cycle Bouncer 2: “Well, I know exactly where to find the ETC. I see the ETC all the time while I’m watching the Krebs Cycle Club. The ETC hangs out around the perimeter of the club. C’mon! I’ll show you!” (Krebs-Cycle Bouncer 2 leads entire group to an area designated as the mitochondrion membrane) Krebs-Cycle Bouncer 2: (Taps ETC 1 on the shoulder) “Uh…are you part of the ETC?” ETC: “I thought I’d be waiting forever and that ya’ll would never get here! That must have been a pretty long cycle you were a part of! If you’re tired, we can take the load off of you, if you give us your electrons!” Glycolysis 3: “Well, good! I can’t wait to get rid of these” ETC: make a the ramp.” “The electrons will fall in energy as they go down the ramp. We can trap this energy to lot of ATP! But first, we need oxygen to pick up the electrons at the bottom of Narrator jumps in: “After all, cellular RESPIRATION requires Oxygen. That’s why all animals have gills, lungs and other organs to breathe oxygen and plants have stomata to let in Oxygen!” Oxygen: (Places his oxygen bucket at the bottom of the ramp). “Oh…you need me! I’ll take the left-over electrons that nobody wants once they have fallen in energy” Glycolysis 3: “Okay, here it goes!” (Glycolysis 3, Krebs Cycle Bouncer 2, Krebs Cycle 3, and Krebs Cycle 6 all give up their electrons and roll them down the ETC ramp) Krebs Cycle Bouncer 2: “Oh, I feel so much….lighter…like a weight has been lifted! I’d better get back to my post, guarding the Krebs Cycle Club” (Returns to original post) Oxygen: “Hmmm…a chemical reaction is going on! These electrons are helping to attract a lot of hydrogen. I have the urge to BOND!” Narrator: (creates drumroll and makes a “kaboom” sound, as if a chemical explosion has occurred). Oxygen: “What’s this?” (lifts up cup of water out of his bucket and squirts water from pipette) “Oxygen, electrons, and hydrogen all bonded to make water!! But that’s not all!” (lifts giant ATP magnet from bucket) “It looks like we’ve also made a total of 32 ATPs from these electrons! The cell now has enough energy to do cellular work! Tada!!!” Class: (Everyone cheers and whistles) Narrator: “So the task is done, we’ve completed our goal. Glucose with the sugar-blues successfully undergoes his make-over, and in this process called cellular respiration, glucose helps to create 36 ATPs for the cell to use as an energy source.” Page | 207 Photosynthesis Study Guide Stage 1: Capturing Light Energy 1. A photon is a tiny packet of light ______________. 2. True or False 3. Briefly explain why plants appear green. __________________________________________ All photons carry the same amount of energy. _________________________________________________________________________ 4. True or False A particular type of pigment can absorb a photon of any energy level. 5. The major light absorbing pigment in plants is _________________ which is found inside the organelle called the __________________. 6. True or False Plant and animals cells contain chloroplasts. 7. What happens when atoms in a pigment absorb light energy? _________________________ ________________________________________________ 8. Many reactions in photosynthesis are classified as ________________ - _________________ reactions. These reactions involve the transfer of electrons from one molecule to another. 10. True or False In cells, electrons travel alone. 11. In cells, electrons travel from one molecule to another attached to a ________________. 12. As review, what is the electrical charge on a proton? _______________ As review, what is the electrical charge on an electron? ______________ As review, what do oppositely charged particles do to each other? ______________________ 13. A proton and an electron together make up a _______________ atom. Page | 208 Stage 2: Using Light Energy to Make ATP and NADPH 14. Look at Figures in your notebook. Identify the location of a chlorophyll molecule. _____________________________________________ 15. True or False A thylakoid is a disk-like structure. 16. What happens to electrons in chlorophyll when photons of light strike the chloroplasts? _____ ________________________________________ 17. Each excited electron, traveling as part of a ________________ atom, leaves the chlorophyll and jumps to a nearby protein in the thylakoid membrane. 18. What does the proton pump do to the hydrogen that is carrying an “excited” electron? ______ _________________________________________________________________________ 19. What is the role of the energy that is released by the proton pump? _____________________ _________________________________________________________________________ 20. What molecule is produced when the protons diffuse back through a channel protein? ______ 21. Review - Study the following steps of photosynthesis to determine the order in which they take place. Write the number of each step in the blank provided. ____ a. An electron that is attached to a proton becomes excited by the light. ____ b. Protons inside the thylakoid are driven out through a protein channel. 1 c. Photons strike the chloroplasts. ____ d. The excited electron, with its attached proton, moves to a thylakoid membrane, where it powers the pumping of the proton across the membrane. ____ e. The force of the protons leaving the thylakoid adds a phosphate to ADP, forming ATP. Page | 209 22. The second type of chlorophyll releases an excited electron that combines with a hydrogen atom that is then attached to NADP. What molecule is formed from the hydrogen and NADP? ____________ 23. What are the ATP and NADPH used for? ________________________________________ _____________________________ Stage 3: Building Carbohydrates 24. What is the ultimate goal of photosynthesis? _______________________________________ _________________________________________________________________________ 25. The process of capturing carbon atoms and using them to build glucose and other compounds is called the ____________ cycle. 26. Is photosynthesis a more complicated process than you thought it was? _______ 27. In the space below, write the chemical equation for the overall process of photosynthesis. 28. What are the reactants of photosynthesis? _____________________________________ 29. What are the products of photosynthesis? _____________________________________ 30. How do plants store the sugars produced as a result of photosynthesis? __________________ _________________________________________________________ 31. What can plants do with the stored starch molecules? ________________________________ _________________________________________________________ Page | 210 32. Multiple Choice - Circle the correct answer. The ultimate goal of photosynthesis is to A. make ATP from carbon dioxide. B. construct carbon containing molecules that serve as an energy source. C. convert ADP to ATP by using energy from the sun. D. use enzymes to speed up chemical reactions. Page | 211 Cellular Respiration Study Guide Glycolysis and Respiration 1. What is the name of the process by which living organisms release the energy stored in carbohydrates and other food molecules? _______________ ________________ 2. What is the first result of cellular respiration? _______________________________________ 3. What happens to the energy that is released when the bonds of ATP are broken? ___________ __________________________________________________________________________ 4. The two stages of cellular respiration are ____________________ and __________________ ____________________. 5. True or False Glycolysis occurs in the cytoplasm of the cell and does not require oxygen. 6. True or False Oxidative respiration occurs in the mitochondria of plant and animal cells. 7. Looking at the name “oxidative respiration”, do you think oxidative respiration requires oxygen? _____ 8. True or False Glycolysis is more effective than oxidative respiration at recovering energy from food molecules. 9. True or False Glycolysis is an ancient biological process. 10. True or False The word “glycolysis” means the “splitting of glycogen”. 11. True or False The products of glycolysis are two pyruvate molecules. 12. The cell must use some ATP to begin glycolysis. Remembering what you learned in Section 5.1, what is the ATP used to begin glycolysis called? ____________________ _____________ Page | 212 13. What happens during glycolysis to convert NAD into the molecule NADH? _______________ __________________________________________________________________________ __________________________________________________________________________ 14. What are the two possible processes that follow glycolysis? ______________________ or ___________________ ___________________ 15. What are the two possible substances formed when the hydrogen atom of NADH is added to pyruvate? ___________ _______ or __________ _____________ 16. Microorganisms living in the absence of oxygen use the process of _____________________ to produce relatively small amounts of ATP. What does “in the absence of oxygen” mean? _________________________________________________ 17. Fermentation takes place in ___________ cells when they do not receive enough oxygen, resulting in the formation of a substance called ______________ _________. 18. When the blood cannot remove lactic acid fast enough, what happens in your muscles? ________________________________________________________ 19. How did the formation of oxygen in the atmosphere change the way hydrogen was used during respiration? __________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ _________________________________________________________________________ 20. Why is the production of water a better alternative? _________________________________ _________________________________________________________________________ _________________________________________________________________________ Page | 213 21. In the space below, write the equation for the breakdown of glucose by glycolysis and oxidative respiration. 22. Look at the figures in your notes. How many ATP molecules are released by glycolysis? _______ How many ATP molecules are released by fermentation? _______ How many ATP molecules are released by oxidative respiration? ______ 24. After each pyruvate is oxidized, a two-carbon fragment is left. What happens to the oxidized pyruvate if the cell has enough ATP? _____________________________________________ ________________________ If the cell does not have enough ATP, the remaining twocarbon fragment joins with a four carbon sugar to begin the ________ cycle. 25. True or False The Krebs cycle consists of nine reactions. 26. The high energy electrons in NADH are used to make ATP during the _______________ __________________ __________. 27. True or False Due to the presence of proton pumps in the membranes of mitochondria, a living cell is never without a supply of ATP. 28. What happens to the electrons after the proton pumps have used their energy? _____________ __________________________________________________________________________ Where do you think the oxygen gas comes from? ____________________________________ 29. True or False Glycolysis releases more ATP molecules than does oxidative respiration. Page | 214 Regulating Cellular Respiration 30. What is feedback inhibition? __________________________________________________ __________________________________________________________________________ __________________________________________________________________________ 31. True or False Excess ATP may bind to the regulatory site on an enzyme early in glycolysis and the Krebs cycle and shut down the processes the cell uses to make ATP. Review Fill in the blanks using the following terms. Write the letter of the term in the blank at the beginning of the question. A. glycolysis B. fermentation C. oxidative respiration D. feedback inhibition E. pyruvate ____ 32. The slowing or stopping of an early reaction is caused by ?????. ____ 33. The term ????? is used to refer to the breakdown of sugar when it occurs in the absence of oxygen. ____ 34. The process of ????? occurs in the mitochondria and uses oxygen. ____ 35. Glycolysis breaks down glucose into two molecules of ?????. ____ 36. The first stage of cellular respiration is ?????, which occurs in the cytoplasm of the cell. Page | 215 Photosynthesis Review After it is labeled, the diagram below will illustrate photosynthesis. Write each of the following terms on the correct numbered line. Then answer the questions that follow. Carbon dioxide Glucose Oxygen Water 5. a. In photosynthesis, what three things come in from outside the plant? ______________________________________________ b. What are products of photosynthesis?__________________________________ c. In what organelle does photosynthesis occur? _____________________ 6. Write the overall equation for photosynthesis. ______________________________________________________________________ 7. How does life on earth depend on the process of photosynthesis? ______________________________________________________________________ ______________________________________________________________________ Page | 216 Cellular Respiration Review Answer the following questions relating to cellular respiration. 1. The chemical bonds in glucose contain stored energy. The purpose of cellular respiration is to store the energy of chemical bonds of glucose in molecules of _________. 2. Write the formula that shows the release of energy by a molecule of ATP. __________________________________________________________ 3. The concept map below illustrates cellular respiration. Place a star under each of the boxes that show stored energy. Color the boxes in the concept map as directed: Use red for the boxes that show glycolysis. Use blue for the boxes that show the path taken during fermentation. Use green for the boxes that show the path taken during oxidative respiration. Page | 217 4. a. In the process of cellular respiration, what two substances come in from the outside?__________________________________ b. What are the products of cellular respiration?____________________________ c. Cellular respiration starts in the cytoplasm of the cell. In what organelle is cellular respiration completed? __________________________ 5. Write the overall equation for cellular respiration in the space below. ______________________________________________________________________ 6. Compare the equation for cellular respiration with the equation for photosynthesis. ______________________________________________________________________ _____________________________________________________________________ ______________________________________________________________________ Page | 218 Cellular Energy Unit Concept Cards Page | 219 Cellular Energy Unit Concept Map Page Page | 220 Parent/ Significant Adult Review Dear Parent/ Significant Adult: This Interactive Notebook represents your student’s learning to date and should contain the work your student has completed. Please take some time to look at the unit your student just completed, read his/ her reflection and respond to any of the following The work we found most interesting was ____________________________ because… What does the notebook reveal about your student’s learning habits or talents? My student’s biggest concern about this class is… Parent/ Significant Adult Signature: Comments? Questions? Concerns? Feel free to email: Mr. Duston at ryan.duston@venturausd.org Ms. Perez at darcy.perez@venturausd.org Page | 221 Cellular Energy Unit Back Page The California State Standards I have come to use and understand are: Usable energy is captured from sunlight by chloroplasts and is stored through the synthesis of sugar from carbon dioxide. The role of the mitochondria in making stored chemical-bond energy available to cells by completing the breakdown of glucose to carbon dioxide. How chemiosmotic gradients in the mitochondria and chloroplast store energy for ATP production. Page | 222 Biology Semester One: Deadline and Contact Information Due Date: Partner’s Name E-mail/Phone Due Date: Partner’s Name E-mail/Phone Page | 223 Page | 224