FINAL FLY PAPER
advertisement

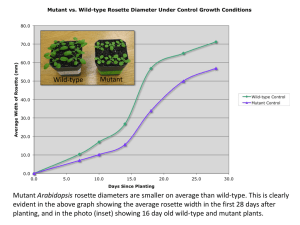
Determination of gene location, order, and linkage in Drosphila melanogaster for body color, eye color, bristle shape, and wing shape Shannon Ceperich, Lindsey Levine, and Marybeth Norton Introduction Heredity is the passing of traits, controlled by genes, from parents and ancestors to offspring. It can lead to species evolution and adaptation. Thomas Hunt Morgan showed that genes are linked in a series on chromosomes and are responsible for identifiable, hereditable traits (Morgan 1916). The purpose of this experiment was to examine the inheritance of Drosophila melanogaster and the genes responsible for ebony body, white eyes, miniature wings, and singed bristles. The wild-type eye color of D. melanogaster is red (w+) and a mutant phenotype is white (w). White eyes can result from a sex-linked mutation on the X chromosome. This was first observed by Morgan in 1910 when he saw a male fly with white eyes. He crossed that whiteeyed male with a red-eyed female and both sexes of the F1 progeny had red eyes. He crossed the F1 progeny and found that half of the F2 males had red eyes and the other half had white eyes. The F2 females had red eyes. The eye color mutation occurred in males, leading him to believe that eye color was sex-limited. Further breeding proved that the white-eyed mutation occurred in a sex-linked gene (Morgan 1910). Because the F1 females were heterozygous and expressed the red eye phenotype, the wild type red eyes trait is dominant and the mutant white eye trait is recessive (Reed and Reed, 1948). The "w" gene that leads to white eye color is located at 1.5 MU from the end of the X chromosome (Morgan 1910.) The heterozygous F1 female progeny resulting from reciprocal crosses between female wild-type, pure breeding, red-eyed (w+), normal-bristled (sn+), long-winged (m+), tan-bodied (e+) flies and male mutant, pure breeding, white-eyed (w), singed-bristled (sn), miniature-winged (m), ebony-bodied (e) flies are expected to show the dominant red eye trait and the hemizygous F1 male progeny are expected to have the same eye color phenotype as the mother because it is an X-linked trait. The mutant ebony body is due to an autosomal recessive mutation in the ebony gene (e) located on the third chromosome in Drosophila melanogaster at 70.7 mu (Bridges 1923). This mutation was first discovered by E. M. Wallace in 1990 (Wallace 1990). The wild-type (e+) allele of the gene results in a tan-colored body. The ebony mutation affects the quantity of pigments formed as well as the time of their appearance making the adult fly body black (Waddington, 2009). The wild- type tan body allele is dominant and the mutant ebony body allele is recessive. The F1 progeny resulting from the reciprocal crosses of this experiment are expected to show the dominant tan body trait because all flies, both male and female, are heterozygous for this autosomal gene. Wing size is another phenotype being observed. Morgan was the first to discover the miniature wing (m) mutant in 1910. Miniature wings are a recessive, X-linked trait that is controlled by a gene located at 36.1 mu on the X chromosome (Bridges and Morgan 1923). The mutant miniature wings are smaller in size than the wild-type wings, but can be similar in shape (Waddington, 2009). The flies in this experiment that have miniature wings are expected to have rounder wings. The heterozygous F1 female progeny of this experiment are expected to express the dominant wild-type wing and the hemizygous F1 male progeny are expected to have the same wing phenotype as the mother because it is an X-linked trait. Mohr was the first to discover the singed bristle mutant in 1922 and found the responsible gene (sn) to be X-linked and mutant allele recessive. The location of this gene is 20.9 mu from the chromosome end (Mohr 1922). The singed bristle phenotype appears gnarled and kinky and is due to the lack of actin filament bundles in both large and small bristles. The DNA of flies with singed bristles contains a TATA-box deficient (TATA-less) promoter (Roiha 1988). Reciprocal crosses between pure-breeding wild-type females and hemizygous mutant males and pure-breeding mutant females and hemizygous wild-type males were used to determine what were dominant traits and if a gene was sex-linked or autosomal (Figure 1). The F1 females would express the dominant traits because they were heterozygous. For example, if the F1 females all have normal wings, then normal wings are dominant and miniature wings are recessive. Females have two X chromosomes and males have one X chromosome and a Y chromosome. This means that the male inherits his X chromosome from his mother. In this experiment, F1 male flies resulting from pure breeding mutant females and pure breeding wildtype males were observed to determine sex linkage. The F1 male progeny resulting from this experiment’s reciprocal crosses between pure breeding flies (Figure 1) should have the same phenotype as his mother if the gene causing the phenotype is X-linked as his only X chromosome is from her. If the gene causing the phenotype was autosomal, then the F1 male progeny had a 75% chance of expressing the dominant phenotype and a 25% chance of expressing the recessive phenotype. A test cross between F1 heterozygous virgin females and hemizygous recessive males who were heterozygous for the autosomal (e/e+) gene, was used to determine map distances between genes as well as whether genes were linked to each other or followed The Law of Independent Assortment (Figure 2). The Law of Independent Assortment, first described by Gregor Mendel, the father of genetics, states that allele pairs separate independently of other allele pairs during gamete formation. On the other hand, genes may be linked and inherited together during meiosis because they are located near each other on the same chromosome. These linked genes are less likely to be separated by a crossover event. In heterozygotes, if two phenotypes are correlated, or always appear together, they can be assumed to be resulting from linked genes (Sutton 1903); therefore, the F2 progeny were screened for gene linkage. As shown in Figure 2 and based on the hypothesis that for both male and females all Xlinked traits are linked and will not separate in a crossover event, 3/8 of the F2 progeny are expected to be completely wild-type, 1/8 are expected to be wild-type for the X-linked traits and to express the mutant ebony body, 3/8 are expected to be mutant for the X-linked traits and to express the wild-type body, 1/8 are expected to be completely mutant, 1/2 of are expected to express the wild-type of the X-linked genes, and 1/2 are expected to express the wild-type of the autosomal body color gene. A) w sn m P1 e+ X w sn m e+ e w+ sn+ m+ e+ w e F1 B) w+ sn+ m+ e w sn m e+ & sn m w+ sn+ m+ e+ w+ sn+ m+ e+ w+ sn+ Mv e+ P2 e w sn m e X F1 e w+ sn+ m+ e+ & w sn m e e Figure 1. Results expected from reciprocal crosses between wild-type, pure breeding, red-eyed (w+), normal-bristled (sn+), long-winged (m+), tan-bodied (e+) flies and mutant, pure breeding, white-eyed (w), singed-bristled (sn), miniature-winged (m), ebony-bodied (e) flies. The w,sn, and m genes are X-linked and the e gene is autosomal. A) In the P1 cross, pure breeding mutant females (ww snsn mm ee) were crossed with the pure breeding wild-type males (w+ sn+ m+ //\ e+e+). The resulting F1 females were expected to be heterozygous for all traits and to express the dominant allele. The resulting F1 males were expected to be hemizygous mutant for the X-linked traits as they inherited their X chromosome from their homozygous wild-type mothers. They were expected to be heterozygous for the autosomal body color gene and thus express the dominant wild-type of the gene. B) In the P2 cross, pure breeding wild-type females (w+w+ sn+sn+ m+m+ e+e+) were crossed with pure breeding mutant males (w sn m //\ ee). The resulting F1 females were expected to be heterozygous for all traits and to express the dominant allele. The resulting F1 males were expected to be hemizygous wild-type for the X-linked traits. They were expected to be heterozygous for body color and thus express the dominant wild-type. e+ w sn m + + + m e m+ e+ F1 F2 w sn m e+ X w sn w+ sn+ e w+ sn+ m+ e+ & 3/8 w sn m e or e+ w+ sn+ m+ e e or e+ w+ sn+ m+ e & 1/8 w sn m e w sn m e+ e w sn m e+ & 3/8 w sn m e or e+ w sn m e e or e+ w sn m e & 1/8 w sn m e e Figure 2. An F1x F1 cross was conducted between the heterozygous F1 females (w+w sn+sn m+m e+e) and the F1 males (w sn m //\ e+e) resulting from the P1 reciprocal cross (Figure 1). This cross will help determine map distances between the genes if genes are linked to each other. Based on the hypothesis that for both male and females all X-linked traits are completely linked and will not separate in a crossover event, 3/8 of the F2 progeny are expected to be completely wild-type, 1/8 are expected to be wild-type for the X-linked traits and to express the mutant ebony body, 3/8 are expected to be mutant for the X-linked traits and to express the wild-type body, 1/8 are expected to be completely mutant, 1/2 of are expected to express the wild-type of the X-linked genes, and 1/2 are expected to express the wild-type of the autosomal body color gene. Results Table 1. Progeny resulting from reciprocal crosses between wild-type, pure breeding, red-eyed (w+), normal-bristled (sn+), long-winged (m+), tan-bodied (e+) flies and mutant, pure breeding, white-eyed (w), singed-bristled (sn), miniature-winged (m), ebony-bodied (e) flies. In the P1 cross, pure-breeding mutant females (ww snsn mm ee) were crossed with the pure breeding, wildtype males (w+ sn+ m+ // e+e+). In the P2 cross, pure breeding wild-type females (w+w+ sn+sn+ m+m+ e+e+) were crossed with pure breeding mutant males (w sn m // ee). Cross P1 P2 Parents Mutant Females and Wild-type Males Wild-type Females and Mutant Males Phenotypes of F1 Progeny Observed Number Observed Females Males Red eyes, normal bristles, long wings, and tan body 57 0 White eyes, singed bristles, miniature wings, and tan body 0 43 Red eyes, normal bristles, long wings, and tan body 55 45 Table 2. Progeny resulting from a cross between heterozygous red-eyed (w+), normal-bristled (sn+), long-winged (m+), tan-bodied (e+) F1 females (w+w sn+sn m+m e+e) and the F1 males (w sn m // e+e) resulting from the P1 reciprocal cross and expressing white-eyes (w), normalbristles (sn), long-wings (m), and tan-bodies (e+) carrying the autosomal recessive allele for mutant ebony bodies (e). F2 Progeny Phenotypes Origin Body color Eye color P P P P DCO DCO DCO DCO SCOI SCOI SCOI SCOI SCOII SCOII SCOII SCOII Tan Ebony Tan Ebony Tan Ebony Tan Ebony Tan Ebony Tan Ebony Tan Ebony Tan Ebony Red Red White White Red Red White White Red Red White White Red Red White White Bristle Shape Normal Normal Singed Singed Singed Singed Normal Normal Singed Singed Normal Normal Normal Normal Singed Singed Numbers Expected If all 4 genes Total independently assorting ** 107 38 39 13 4 38 52 13 6 38 11 13 57 38 7 13 30 38 7 13 23 38 25 13 20 38 9 13 8 38 0 13 Numbers observed Wing Shape Long Long Miniature Miniature Long Long Miniature Miniature Miniature Miniature Long Long Miniature Miniature Long Long Males Females 49 18 0 15 1 6 24 1 14 5 11 8 3 5 4 0 58 21 4 37 5 5 33 6 16 2 12 17 17 4 4 0 ** If all four genes were independently assorting, then there was a ½ chance that the F2 progeny would express red eyes, a ½ chance they would express normal bristles, and a ½ chance they would express long wings. There was a ¾ chance they would express a tan body and a ¼ chance they would express a mutant white body color. Statistical Analysis and Conclusion Reciprocal crosses between pure-breeding wild-type females and pure-breeding males who were mutant for white-eyes, singed-bristles, miniature-wings and ebony body color or purebreeding white-eyed, singed-bristled, miniature-winged, ebony bodied females and purebreeding wild-type males were used to determine if a gene was sex-linked or autosomal and which allele was dominant. All of the F1 females resulting from both reciprocal crosses (Table 1) were heterozygous for all traits and were tan-bodied, red-eyed, normal-bristled, and longwinged; therefore, the wild-type phenotype for all traits was dominant. The progeny resulting from the reciprocal crosses also showed that the gene controlling body color was autosomal and the genes for eye color, bristle shape, and wing shape were X-linked. The genes for eye color, bristle shape, and wing shape were determined to be X-linked because the hemizygous F1 males had the same phenotypes for these traits as their mother. Mothers with red eyes, normal bristles, and long-wings had sons with red eyes, normal bristles, and long-wings. Mothers with white eyes, singed-bristles, and miniature-wings had sons with the same phenotypes for those traits. The gene controlling body color was determined to be autosomal because a cross between a pure-breeding ebony female and a pure-breeding tan male resulted in F1 male progeny with a tan body color. Because the gene controlling body color is autosomal and the genes controlling eye color, bristle shape, and wing type are on the X chromosome, the body color gene is not linked to the other genes. A cross between F1 heterozygous females and F1males hemizygous for white eyes, singed-bristles and miniature-wings and heterozygous for body color (Figure 2) was used to determine whether all four genes were linked to each other or followed the Law of Independent Assortment. The number of progeny in each phenotypic category was predicted based on the hypothesis that all four genes assorted independently (Table 2). The observed number of progeny differed significantly from expectations. A chi square test with 15 degrees of freedom resulted in a value of 306.69, which indicates a probability less than 0.01; therefore, all four genes were not independently assorting and some may be linked. The progeny resulting from the same F1 x F1 cross were used to determine gene order, distances between the genes, and where crossovers occurred if the genes controlling eye color, bristle shape, and wing shape were X-linked. The genes controlling eye color, bristle shape, and wing shape were determined to be in the same order as previously reported (Figure 3); however, the map distances were not at the same distances as reported previously in the literature (Morgan 1910; Bridges and Morgan 1923; Mohr 1992). Using a three point test cross and previously recorded map distances of genes, the expected number of progeny in each phenotypic category was predicted and compared to the observed progeny (Table3). There was a significant difference that was reflected in a large chi square value of 168.34, of less than 0.01; therefore, the hypotheses that the genes would be separated by the same map distances as previously reported in the literature was not supported. The difference between predicted progeny and resulting progeny was likely due to a screening error for bristle shape, which caused a higher count of flies in the double cross category. The screening error was distinguishing flies with singed-bristles versus flies with normal bristles. The least frequent pair of categories should have been double crossovers (w+w snsn m+m and ww sn+sn mm), but we observed 64 progeny in one category (ww sn+sn mm) of double crossovers when the expected value was 12. If the number of progeny with white eyes, normal bristles, and miniature-wings was reduced significantly to match the expected value, the chi-square value would be lower and support the hypothesis better. The F2 progeny were reevaluated by looking at the number as a two point test cross involving the genes controlling eye color and wing shape. Expected frequencies were again predicted using previously reported map distances and compared to the observed progeny (Table4). This two gene comparison eliminated the distortion of the double cross over category. The chi square value was 11.4 with 3 degrees of freedom, which gives a probability less than 0.01. This probability is too low meaning there was a significant difference between observed and expected map units. This may have been due to the fact that there was a larger number of red-eyed progeny than white-eyed progeny (229:176) when there should have been a 1:1 ratio of progeny with red eyes and white eyes. This means that red-eyed flies were surviving more than white-eyed flies. If the number of progeny with red eyes and white eyes was the same, then the map units would have been more similar to the map distances reported previously in the literature (Morgan 1910; Bridges and Morgan 1923; Mohr 1992). The map distances may have been different due to interference. There was a high incidence of double crossovers observed as the value of interference was -2.375. Overall, the gene controlling body color was determined to be autosomal and the genes controlling eye color, bristle shape, and wing shape were X-linked. The wild-type allele was dominant for all traits. All four genes were not independently assorting. The distances between genes controlling eye color, bristle shape, and wing shape were calculated and chi square analysis showed that they were different than the expected distances recorded in previous literature, but the order of the genes was correct (Figure 3). Table 3. Three point test cross evaluation of progeny resulting from a cross between heterozygous red-eyed (w+), normal-bristled (sn+), long-winged (m+). F1 females (w+w sn+sn m+m) and the F1 males(w sn m) resulting from the P1 reciprocal cross and expressing white-eyes (w), normal-bristles (sn), and long-wings (m). Phenotypes Category Eye color Bristle shape Numbers Wing shape Observed Expected * P Red Normal Long 146 140 P Ebony Singed miniature 56 140 DCO Red Normal Long 17 12 DCO Ebony Singed miniature 64 12 SCO I Red Normal Long 37 32 SCO I Ebony Singed miniature 48 32 SCO II Red Normal Long 29 26 SCO II Ebony Singed miniature 8 26 *Numbers expected if genes controlling eye color, bristle shape, and wing shape were linked and did not assort independently. Values were obtained using a distance of 19.4 mu between w and sn and a distance of 15.2 mu between sn and m. Table 4. Two point cross analysis for genes controlling eye color and wing shape using progeny resulting from a cross between heterozygous red-eyed (w+) and long-winged (m+) F1 females (w+w m+m) and the F1 males (w m) resulting from the P1 reciprocal cross and expressing whiteeyes (w) and long-wings (m). Phenotypes Category Eye color Wing shape Number Observed Expected * Parent Red Long 163 132 Parent White Miniature 120 132 Recombinant Red Miniature 66 70 Recombinant White Long 56 70 *Numbers expected if genes controlling eye color and wing shape were linked and did not assort independently. Values were obtained using a distance of 34.6 mu between w and m. A) ___w___19.4___sn___15.2___m___ __e__ B) ___w____41____sn___29.1___m___ __e__ Figure 3. Map distances between genes investigated for inheritance of white eyes (w), singedbristles (sn), and miniature-wings (m). A) The expected map distances and order based on previous literature (Morgan 1910; Bridges and Morgan 1923; Mohr 1992). B) The observed and calculated map distances and order based on progeny resulting from a testcross between F1 heterozygous virgin females and hemizygous recessive males who were heterozygous for the autosomal (e)/(e+) gene. Literature Citations 1. Morgan T, Bridges C (1916) Sex-Linked Inheritance in Drosophila. The Carnegie Institution of Washington, 237 2. Sutton W (1903) The chromosomes in heredity. Biol Bull, 4: 231-251 3. Morgan T (1910) Sex-limited inheritance in Drosophila. Science, 32: 120-122 4. Reed S, Reed E (1950) Natural selection in laboratory populations of Drosophila. Society for The Study of Evolution, 4: 34-42 5. Waddington C (1942) Body-Colour Genes in Drosphila. Proceedings of the Zoological Society of London, 111: 173–180 6. Roiha H (1988) P elements and insertion and rearrangements at the singed locus of Drosophila melanogaster. Genetics, 119: 75-85 7. Bridges C, Morgan T (1923) The third-chromosome group of mutant characters of Drosophila melanogaster. Publs Carnegie Institute, 327: 1-251 8. Mohr (1922) Cases of mimic mutations and secondary mutations in the X-chromosome of Drosophila melanogaster. Abstamm VererbLehre, 28: 1-22 9. Wallace B (1990) Male-mate interactions and mating kinetics in Drosphila. Behav. Genet., 20: 405-421