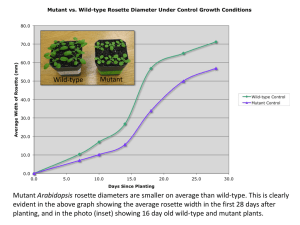
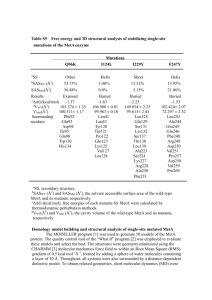
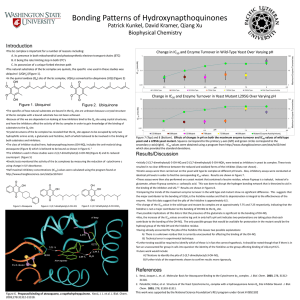
Supplementary Figures 1 and 2
advertisement

Supplementary Figure 1. The termination defect in the rat1-1 mutant is not due to protein instability or defects in polyadenylation. A. Polyadenylation of a precleaved substrate (the Gal7 3' end) was assayed in wild-type, rat1-1, and rna14-1 cells. Extracts were prepared from cells grown at room temperature or shifted to 37oC for 30 minutes as indicated. Reactions were done with and without ATP. The first lane of each panel contains the untreated precursor substrate. Precursor and polyadenylated product are indicated to the right. B. Immunoblotting of Rat1 proteins. Strains in which wild-type Rat1 or rat1-1 proteins are TAP-tagged were grown at room temperature and shifted to 37 oC for the indicated amount of time. Extracts were separated by gel electrophoresis, blotted to membranes, and probed with anti-PAP/horse radish peroxidase. The blot was also probed with anti-Rpb3 as a loading control. Supplementary Figure 2. The Rat1 D235A mutant lacks exonuclease activity. Immunoprecipitates from yeast expressing epitope-tagged wild-type Rat1, the D235A mutant, or empty vector (see Figure 4D) were incubated with body labeled RNA for the indicated amount of time. Products were analyzed by gel electrophoresis.