Retrosynthesis Solutions
advertisement

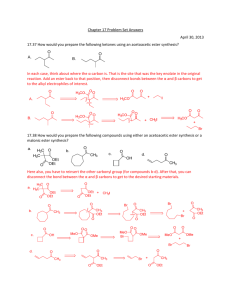
Retrosynthesis Practice Problems Answer Key October 1, 2013 1. Draw a retrosynthesis for how to make the compound shown below from starting materials with eight or fewer carbon atoms. The first step is to convert the OH to a carbonyl group so that you can obtain the key a,b‐unsaturated carbonyl motif that is the product of an aldol reaction: This is a good point to retrosynthetically disconnect the ester to give you the alcohol (which in the forward direction undergoes a Fisher esterification reaction): From here we know how to an aldol reaction retrosynthetically – add an OH to the beta carbon, and then break the bond between the alpha and beta carbon to form a dicarbonyl: I can count three saturated carbons between the two carbonyls in my intermediate, which means that we can use a Michael reaction as the next retrosynthetic step: The alpha beta unsaturated ketone now has less than 8 carbons, so that has been disconnected sufficiently. We need to do a bit more work on the cyclohexenone fragment. Luckily that has an alpha,beta unsaturated carbonyl already in it, which we can disconnect via an aldol reaction: Luckily this intermediate has three saturated carbons between the carbonyls, which again points to a Michael reaction: And now all of the starting materials have 8 or fewer carbon atoms, so we have succeeded. 2. Make the following molecule from starting materials with twelve or fewer carbons (hint: you can leave the heterocycle intact): This is a great molecule, because there are so many places along the macrocycle that can be retrosynthetically disconnected. I show a few examples of that below: We are going to focus on the disconnections that we have talked about in class – the Fisher lactonization and the two aldol reactions, and use each one in turn to break apart the ring. Fisher esterification: Aldol reaction: Second aldol reaction: This is where this problem starts to get a little tricky. The middle OH could have been a carbonyl that was attacked by an organometallic nucleophile. However, you would need to first protect the aldehyde group before doing that sequence so that the aldehyde doesn’t also get attacked by the organometallic nucleophile: In the forward direction, this is a broadly applicable way to protect carbonyl compounds – by treating them with a diol to generate a cyclic acetal: With the protected aldehyde in hand, we are set to do the next key retrosynthetic disconnection: This gives me two starting materials, each of which has 12 or fewer carbon atoms, so we have succeeded here as well. 3. How would you synthesize the compound shown below from an aldol reaction? Draw the structures of both precursors (8 points). We have a procedure for this: break the double bond, put a hydroxyl on the β‐carbon, and then break the bond between the α and β carbon to form two carbonyl starting materials. One thing to note here is that the starting material does not have the double bond in the position that you would expect – i.e. it is not between the alpha and beta carbons. however, if you break that bond, you can still put the OH on the beta carbon, and that gets you right back to an intermediate that is straightforward to disconnect via an aldol retrosynthesis. 4. Provide a retrosynthetic pathway for the following target from “simple, readily available” starting materials: Here the trick is finding where the alpha,beta unsaturated ketone is. We can get to the alpha beta unsaturated ketone in two steps. Step 1: Oxidizing the OH to a carbonyl compound: Step 2: Getting rid of the cyclopropane ring and converting it to the desired alkene: In the forward direction, we can accomplish this transformation with a Simmons‐Smith cyclopropanation reaction. You are responsible for this transformation, but not for the mechanism. The mechanism involves a carbene intermediate: Getting back to the retrosynthesis, now we have an alpha beta unsaturated carbonyl that we can disconnect via an aldol retrosynthesis: And we can follow that with a Michael retrosynthesis: 5. The structure of testosterone acetate is shown below. Draw a retrosynthetic pathway to synthesize this compound from starting materials that have no more than 10 carbon atoms (30 points). This is pretty complicated, so let’s take it one step at a time. Step 1: The easiest thing to get rid of is the ester, to bring it back to an alcohol (that was esterified via Fischer esterification): We also have an alpha beta unsaturated ketone that we know how to deal with retrosynthetically: Next step is a Michael reaction: In the forward direction, we will be able to form the enolate at the position that we want (the more substituted alpha carbon), because that is the thermodynamic enolate. Now I can put a double bond between the alpha and beta carbons to give me another alpha, beta unsaturated ketone: To answer the question about why I am allowed to do this, in a retrosynthesis, you can do anything in the reverse direction that is feasible in the forward direction. So ask yourself, can you selectively hydrogenate the double bond on that alpha, beta unsaturated carbonyl to give the saturated product? Sure. In which case, it is an allowed transformation in the retrosynthetic direction. Once I do that, I can deal with this like any other alpha, beta unsaturated carbonyl: And we can do a Michael reaction too: