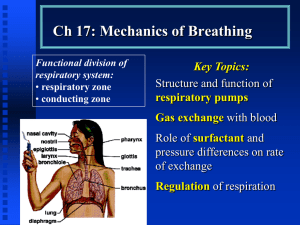
Development of Respiratory System and Body Cavities
advertisement

Development of the Respiratory System Thomas A. Marino, Ph.D. Competencies: Upon completion of this section of the course, you must be able to: • • • • • Define the segments of the primitive gut tube. Describe the embryological movements of the respiratory diverticulum as it develops into the trachea, bronchi, and lungs. Explain the origin of cells that develop into the lung tissue. Compare and contrast morphology of the lungs during the four stages of lung development. Describe the primitive body cavity and how it becomes subdivided into pleural, pericardial and peritoneal cavities. Introduction • Development of the lungs begins at 4 weeks. • The epithelium of the respiratory system develops from endoderm. • The connective tissue, cartilage and muscle develop from splanchnic mesoderm. Early Embryonic Morphology • Early vertebrate body plan • At 26 days a small opening in the foregut appears. • At 28 days it evaginates to form a laryngotracheal diverticulum. Separation of the Laryngotracheal Diverticulm • Longitudinal folds tracheoesophageal ridges develop • Form tracheoesophageal septum Notochord Noggin Sox2 Esophagus Nkx2-1 Lung Bud FGF10 Wnt2 Bmp RA Significance • Lack of – Shh – Retinoic acid receptors – FGF10 – Sox2 – Nkx2-1 – Bmp4 – Noggin – Wnt • Result – Trachoesophageal fistula – Esophageal atresia Tracheoesophageal septum separates • Trachea and lung buds ventral • Esophagus - dorsal Development of the Larynx • Epithelium develops from endoderm of laryngotracheal tube. • Connective tissue and cartilage develops from splanchnic mesoderm. • Cartilages develop from neural crest cells. Development of the Trachea • Epithelium develops from endoderm of laryngotracheal tube • including glands • Cartilage, connective tissue and muscle from splanchnic mesoderm Splanchnic Mesoderm Endoderm Cartilage Smooth muscle Epithelial/Mesenchymal Interactions ! ! Endoderm Mesoderm In FGF10 deficient mice there are no lung buds. FGF2 FGF10 Development of the Lungs • 4th week the lung bud develops • divides into two lung buds Development of the Lungs • Two lung buds divide: • The right one into three main bronchi • The left one into two main bronchi https://syllabus.med.unc.edu/ courseware/embryo_images/ unit-digest/digest_htms/ digest012a.htm Development of the Lungs • Bronchi continue to divide. • By 6 months there have been 17 generations of subdivisions. • After birth there are an additional 6 divisions of the bronchial tree. • As growth occurs there is a caudal development of the lungs. • At birth the tracheal bifurcation is at the level of the 4th thoracic vertebra. Maturation of the Lungs • There are 3 Stages of Lung Maturation • 1. Pseudoglandular Period ( 5 - 16 weeks) • 2. Canalicular Period (16 - 26 weeks) • 3. Terminal Sac (Saccular) Period (26 weeks to birth) • There are 4(5) Stages of Lung Maturation 1. Embryonic ( 4 – 11 weeks) 2. Pseudoglandular Period ( 5 - 16 weeks) 3. Canalicular Period (16 - 26 weeks) 4. Saccular (Terminal Sac) Period (26 weeks to after birth) 5. Alveolar Period (late fetal period to childhood) Maturation of the Lungs • Pseudoglandular Period - 5 to16 weeks • All elements of the lungs are developed except those elements involved in gas exchange. • Branching morphogenesis is prominent. • Terminal Bronchioles present no respiratory bronchioles Branching morphogenesis • Bud elongation • Elongation stops • Tip of the bud widens • Bifurcation FGF10 FGF2 Factors involved • Retinoic acid forms gradient with highest levels proximally – RA inhibits FGF10 • SHH promotes BMP4 which inhibits FGF10 • Wnt3b regulates BMPs which promote proliferation of mesoderm. • What BMPs are regulating is not precisely known at the cellular level. Maturation of the Lungs • Canalicular Period - 16 - 26 weeks • Overlap as cranial segments mature faster than caudal ones. • Lumen of bronchi and bronchioles become large relative to tissues • Bronchial tree branches become narrower. • Respiratory bronchioles and alveolar ducts develop. • Tissue becomes more vascular. Maturation of the Lungs • Canalicular Period (16 - 26 weeks) – Note the cuboidal epithelium of the airway. – The blood vessels are not close to the epithelium Maturation of the Lungs • Terminal Sac Period ( 26 weeks - birth) – Now the epithelium is much thinner. – The blood vessels abut the epithelium • What regulates the switch from pseudoglandular to canalicular an to terminal sac stage? – Alveolar sacs begin to form – Blood vessels become closely associated with the alveolar cells. Maturation of the Lungs • Terminal Sac Period (26 weeks to birth) • • • • • Terminal sacs develop Epithelium becomes very thin Capillaries bulge into the alveoli Type I alveolar cells develop Capillary network develops rapidly Formation of Alveoli • PDGF • Fgf – Fgf2 and Fgf18 important for late alveolar development • Retinoic acid – High and low levels can disrupt lung development. Multipotential cell Bronchiolar cells Non neuroendocrine cells Neuroendocrine cells Alveolar cells Type II cells ?? ?? Ciliated cells Goblet cells Type I cells Maturation of the Lungs • By 20 weeks Type II alveolar cells begin producing surfactant. • Surfactant permits expansion of terminal sacs. • Fetus needs to weigh 1000 gm and be between 26 and 28 weeks before enough surfactant is produced. • Surfactant and enough capillaries are necessary. Maturation of the Lungs • Alveolar Period ( late fetal period to childhood) • Squamous epithelium forms. • During this period respiratory bronchioles end as terminal sacs. • Terminal sacs become alveolar ducts. • Alveoli form after birth. • From 3rd to 8th year alveoli continue to develop. Maturation of the Lungs • Alveolar Period – Now there are: • • • • Type I alveolar cells Type II alveolar cells Macrophages Fibroblasts Lungs at Birth • At birth the lungs are filled with fluid. • Fluid is replaced by air. • Fluid cleared through: • Mouth and nose • Pulmonary capillaries • Pulmonary arteries, veins and lymphatics • After birth most growth is in the number of respiratory bronchioles and alveoli and not an increase in the size of alveoli. Formation of blood vessels • Angiogenesis – Angiogenesis new blood vessels from preexisting blood vessels • Vasculogenesis – angioblasts develop into endothelial cells and new blood vessels form • Vegf helps regulate this along with ephrinB2 and B4 The pulmonary vasculature develops in the absence of lung specification. • Cardiac outflow tract and pulmonary vasculature come from cardiopulmonary mesoderm progenitors that lie in the posterior splanchnic mesoderm. ! • Lung mesenchyme develops separately from these progenitors. T Peng et al. Nature 000, 1-4 (2013) doi:10.1038/nature12358 Clonal analysis reveals that CPPs generate related lineages within the cardiopulmonary system. T Peng et al. Nature 000, 1-4 (2013) doi:10.1038/nature12358 Hedgehog signaling is required in CPPs to coordinate the vascular connection between the heart and lung. T Peng et al. Nature 000, 1-4 (2013) doi:10.1038/nature12358 Development of Body Cavities Development of HorseshoeShaped Pericardial Cavity Lateral body folding occurs as well as head folding. Development of Body Cavities • In the fourth week the embryo has: – large pericardial cavity – left and right pericardioperitoneal canals – large peritoneal cavity Embryonic Circulation Common Cardinal Vein Dorsal Aorta Brain and Spinal Cord Posterior Cardinal Vein Anterior Cardinal Vein Umbilical Artery Umbilical Vein Yolk Sac Aortic Arches Ventricle Atria BODY CAVITY Septum Transversum Vitelline Artery & Vein Division of Body Cavities • Pericardioperitoneal canal is dorsal to septum transversum. • pericardioperitoneal canal is lateral to the foregut. Septum Transversum Division of Body Cavities • As lung bud grows a membrane develops between the lungs and the heart. • Ridge of tissue grows into the pericardioperitoneal canals. • Ridges grow from the lateral walls of each canal. • Ridges called PLEUROPERICARDIAL FOLDS Division of Body Cavities • Pleuropericardial membranes separate pericardial cavity from pleural cavities. • Pleuropericardial membranes contain the common cardinal veins which drain in the primitive heart. • The internal layer of the pleuropericardial membrane becomes the fibrous pericardium. Division of Body Cavities • Pleuropericardial Membranes Division of Body Cavities • Pleuroperitoneal membranes separate the pleural cavity from the peritoneal cavity. • Attachment to the to the dorsolateral abdominal wall. • Project into the pericardioperitoneal canal. • Fuse with the: • dorsal mesentary of the esophagus • septum transversum • Lateral body wall mesoderm Pleuroperitoneal membranes Development of the Diaphragm • The diaphragm develops from: • Septum transversum • Pleuroperitoneal membrane • Dorsal mesentary of the esophagus • Lateral body walls (cervical somite myotomes). Development of the Diaphragm • At week 4 the septum transversum lies opposite the 3rd, 4th, and 5th cervical somites. • Myoblasts from these somites migrate into the diaphragm. • Phrenic nerve comes from cervical nerves 3, 4, and 5. Development of the Diaphragm • As body grows diaphragm appears to migrate caudally. • By 6 weeks diaphragm lies opposite thoracic somites. • Phrenic nerve passes through the pleuropericardial membrane. • Phrenic nerve comes to lie in fibrous pericardium.