A Study of Matter
advertisement

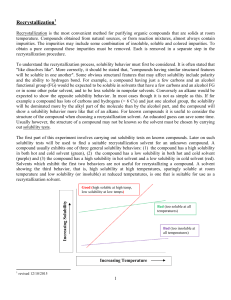
Worksheet: More on Solubility KEY Name______________ 1. Explain what is meant by the expression “like dissolves like”. Substances with “like” polarity will dissolve in each other. 2. An unknown compound is observed to mix with benzene (a nonpolar solvent) but not with water. Is the unknown compound ionic or covalent? covalent If the unknown compound is a liquid, will it be able to dissolve table salt?____________ Explain: no Table salt is an ionic compound. Since the unknown compound is covalently bonded, it will not dissolve an ionic compound. “Like dissolves like.” 3. What are the chemical characteristics of a good dry-cleaning solvent? Because “like dissolves like”, a good-dry cleaning solvent should be nonpolar (covalent) so that it can dissolve oil and grease. 4. Explain why you are more likely to overdose on vitamin A than on vitamin C. Because “like dissolves like”, a good-dry cleaning solvent should be nonpolar (covalent) so that it can dissolve oil and grease. 5. Some industrial plants use water from nearby rivers and streams as a coolant. When the water is returned to the river or stream, the water is warmer than it was originally. This is referred to as “thermal pollution”. Using your knowledge of solubility, why might this thermal pollution be harmful to fish? The warmer the water, the less oxygen can be dissolved. The decrease in dissolved oxygen could cause a fish kill. CHEMISTRY: A Study of Matter © 2004, GPB 10.14a 6. After a bottle of carbonated drink has been open for a while, it tastes “flat”. Explain why. According to Henry’s Law, more CO2 can be dissolved under pressure. When the bottle of carbonated drink is opened, the pressure is decreased and the excess CO2 escapes, causing the soda to taste flat. 7. For most solid solutes, the degree of solubility in a liquid solvent (increases, decreases) with an increase in the temperature of the solvent. 8. Describe what happens to the degree of solubility of a gaseous solute in a liquid: a) with a decrease in the temperature of the solvent. Solubility of a gaseous solute in a liquid solvent increases with a decrease in temperature. Henry’s b) with an increase in pressure ( ____________ Law). Solubility of a gaseous solute in a liquid solvent increases with an increase in pressure. 9. The following statement is false: It is not possible to make a saturated solution from a substance that is described as only slightly soluble. Explain why this statement is false. It is possible to make a saturated solution from a substance that is described as only slightly soluble. It just isn’t possible to make a concentrated solution from a substance that is described as only slightly soluble. CHEMISTRY: A Study of Matter © 2004, GPB 10.14b