Science Chemistry - Bayport-Blue Point School District
advertisement

POINTUNIONFREESCHOOL
BAYPORT-BLUE
DISTRICT
NEWYORK
BAYPORT,
Boardof Education
JamesS.March
President
CarolA. Cinelli
VicePresident
WilliamT. Barry
JeanineBrowning
JaneBurgess
LeonardCamarda
LauraJankowski
AndreaM.O’Neill
Andrew1. Wittman, Jr.
Superintendentof Schools
RichardW.Curtis
AssistantSuperintendentfor Curriculum,Instruction,
and GeneralAdministration
JoanE.Grazda
AssistantSuperintendentfor Business
DorleeseJ.Stewart
)
ChemistryCurriculum(revision
Summer2002
Writers
BrianCunningham
BryanHoran
BryanFranke
MargaretRomaine
Adopted 2004 2005 SchoolYear
BAYPORT-BLUEPOINTUNIONFREESCHOOLDISTRICT
BAYPORT,NEWYORK
Chemistry Curriculum
Labs
Table of Contents
Unit I
Intro to Chemistry
Lab 01:
Lab 02:
Lab 03:
Lab 04:
Lab 05:
Lab 06:
Lab 07:
Lab 08:
Lab 09:
LaboratoryEquipment& Skills
Introductionto Measurement
The PaperAirplane Lab
QualitativeObservationsof a ChemicalReaction
QuantitativeObservationsof a ChemicalReaction
MeasuringMass
Uncertaintyin Measurement
How Much Sugar is in Your BubbleGum?
TemperatureMixing
Unit 2
Physical Behavior of Matter
Lab 10:
Lab 11:
Lab 12:
Lab 13:
Lab 14:
Lab 15:
Lab 16:
Lab 17:
Lab 18:
Lab 19:
Lab 20:
Lab 21:
Lab 22:
PaperClip Formulas
ProblemSolvingto Separatea Mixture(Inquiry)
What Colorsare in Your Candy?
Chemicalor PhysicalChangeLab
ExploringEnergyChanges(Exothermic& Endothermic...)
CaloricContentof Foods
TheThree Phasesof Matter
Molecules& Energy
MeasuringEnergyChanges(Heat of Fusion)
DiscoveringInstantCold Packs(Heat of Solution)
Specific Heat
Why Doesn’tthe BalloonBurst?
Gas Law Lab
Unit 3
Atomic Theory
Lab 23:
Lab 24:
Lab 25:
Lab 26:
Lab 27:
Lab 28:
Who’s Counting?(atoms,mass, & moles)
Atomic Coatings(the size of the atom)
Line EmissionSpectraof Elements
FlameTests (atomicemission& electronenergy levels)
Isotopesof Pennies
Bean Bag Isotopes
Page
--
1
8
11
16
20
27
30
32
Page
36
37
38
41
43
49
51
53
56
62
66
67
68
Page
70
75
79
81
87
92
Unit 4
Periodic Table
Lab 29:
Lab 30:
Lab 31:
Lab 32:
It’s in the Cards (Discoveringthe PeriodicLaw)
Testingfor Metals
Determiningthe Trendsin a Group
DiscoveringPeriodicTrendsin Reactivity
Unit 4
Bonding
Lab 33:
Lab 34:
Lab 35:
Lab 36:
Lab 37:
Propertiesof Ionic& CovalentCompounds
BondmgPractice
MakingModelsof Compounds
CovalentMoleculesfrom a 3-DimensionalView
MaqicSand
Unit 6
Formulas, Equations, Moles, & Solutions
Lab 38:
Lab 39:
Lab 40:
Lab 41:
Lab 42:
Lab 43:
Lab 44:
Lab45:
Lab46:
Formulas& OxidationNumbers
The ChalkLab
Compositionof Hydrates
RelatingMolesto Coefficientsof a ChemicalEquation
PerfectCup of KoolAid Lab
FactorsAffecting SolutionFormation
Solubility& Temperature
A HeatedDiscussion
CommonGases(Physical& ChemicalProperties)
Unit 7
Types of Reactions
Lab 47:
Lab48:
Lab49:
Lab 50:
Lab 51:
Lab 52:
Exploring_Chemical_Changes
Types_ofChemical_Reactions
Preparing& TestingHydrogenGas (A microscaleapproach)
ProducingHydrogenGasfrom CalciumMetal
Reactivityof Metalsin Single-ReplacementReactions
Double ReplacementReactions& SolubilityRules
Unit 8
Kinetics & Equilibrium
Lab 53.
Lab 54:
Lab 55:
Lab 56:
Lab 57:
Lab 58:
Lab 59:
Lab 60:
Introductionto ReactionRates (The Blue Bottle Reaction)
Ratesof Reaction
DeterminingReactionRate
Catalysts,ReactionRates& ActivationEnergy
The Effectof Temperatureon ReactionRate
ExploringEquilibrium(It Works BothWays)
ChemicalEquilibrium& Le Chatelier’sPrinciple
Gas Phase Equilibrium
Page
96
103
108
110
Page
113
115
118
123
125
Paqe
129
134
136
139
144
148
151
162
157
Page
173
176
182
187
189
191
Page
195
201
207
208
211
217
219
223
Unit 9
Redox Reactions
Lab 61:
Lab 62:
Lab 63:
Lab 64:
Lab 65:
Lab 66:
Page
ChangesDuring RedoxReactions
The MirroredFlask
Activitiesof Metals(Redox)
The Gold Penny Lab
Corrosionof Iron
ChemicalHand-Warmer
228
229
230
234
2236
239
Unit 10
Acids & Bases
Page
Lab 67:
Lab 68:
Lab 69:
Lab 70:
Lab 71:
Lab 72:
Lab 73:
Lab 74:
Propertiesof Acids & Bases
Acid, Base IndicatorLab (TableM)
NaturalIndicators(Acids, Base,& the pH Scale)
Acid-BaseTitration
Titrationof VineqarLab
Total Acidity (Titrationof Fruit Juices)
Determinethe PHof an UnknownSolutionLab
ChemMystery(The LimestoneCave)
Unit 11
Organic Chemistry
Lab 75:
Lab 76:
Lab 77:
Modelsof OrqanicCompounds(Hydrocarbons)
Modelsof Other OrganicCompounds(TableR)
Saponification
Unit 12
Nuclear Chemistry
Lab 78:
Lab 79:
Lab 80:
Lab 81:
Half-LifeSimulation(Pennium)
NuclearFissionChainReaction(dominoes)
RadioactiveDecay:The Half-Lifeof an M&M
BubbleGum Half-LifeLab
Unit 13
Additional AP Chemistry Labs
Lab 82:
Lab 83:
Lab 84:
Lab 85:
Lab 86:
Lab 87:
Lab 88:
Lab 89:
Lab 90:
Atomic Spectra(Light,Energy,& ElectronStructure)
QuantumLeap Lab (Probability& ElectronStructure)
MagnesiumOxide (% Composition& EmpiricalForumula)
FreezingPoint Depression(How LowCanYou Go?)
FreezingPoint Depressionwith Antifreeze
ClassicTitration (pH Curves& an Unknown)
MeasuringAcid Strength(Ka valuesof Weak Acids)
Heatsof Reaction& Hess’sLaw (Small-ScaleCalorimetry)
The EquilibriumConstant(ComplexIon Formation)
12/04
241
247
250
255
261
263
267
269
Paqe
273
281
287
Page
291
294
296
298
Page
301
308
317
322
328
331
338
343
347
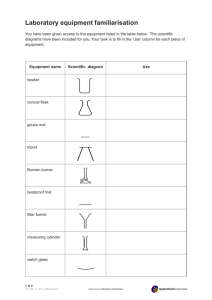
Page1 Introductionto Measurement
-
.
1
Introductionto Measurement
Mass, Length, and Volume
Introduction
Muchofwhat we knowabout the physicalworldhas been obtainedfrom measurements
made in the laboratory.Skill is required to designexperimentsso that careful measurements
can be made. Skillis also neededto use lab equipmentcorrectlyso that errors can be
minimized.Atthe same time, it is important to understand the limitations of scientific
measurements.
I
Concepts
..
rft
Measurem
• Accuracyand precision
Background
•
•
•
Significantfigures
•
Experimentalerror
I
Experimentalobservationsoften include measurementsof mass, length, volume, temperature, and time. There are three parts to any measurement:
•
its numericalvalue
•
the unit of measurement that denotes the scale
•
an estimate ofthe uncertainty of the measurement.
The numericalvalueof a laboratorymeasurementshould alwaysbe
recordedwith the proper number of significantfigures. The number of
significantfiguresdependson the instrument or measuring deviceused
and is equalto the digitsdefinitelyknownfromthe scaledivisionsmarked
on the instrument plus one estimated or “doubtful”digit. The last, esti
mated, digit representsthe uncertainty in the measurement and indicates
the precision ofthe instrument.
Measurementsmadewith rulers and graduated cylindersshould always
be estimatedto one placebeyond the smallest scale divisionthat is
marked.If the smallestscale division on a ruler is centimeters, measure
ments of length should be estimated to the nearest 0.1 cm. If a ruler is
marked in millimeters,readings are usually estimated to the nearest 0.2
or 0.5 mm, dependingon the observer.The same reasoningapplies to vol.
ume measurementsmade using a graduated cylinder.A 1O-mLgraduated
cylinder has major scale divisionsevery I mLand minor scale divisions
every0.1 mL.It is therefore possibleto “read”the volumeof a liquid in a
10-mLgraduatedcylinder to the nearest 0.02 or 0.05 mL.Three observers
might estimatethe volume of liquid in the 10-mLgraduatedcylinder
shown at the right as 8.32, 8.30, or 8.33 mL.These are all valid readings.
It wouldNOTbe correct to record this volume of liquidas simply 8.3 mL.
Likewise,a reading of 8.325 mL would be too precise.
Introductionto Measurement
_____
_____
_____________
T
J
Introductionto Measurement Page2
—
Some instruments, such as electronic balances,give a direct reading—thereare.no obvious
in an electronic
rtainty
or marked scaledivisions.This does NOTmean that there is no unc
balance measurement; it means that the estimation has been carried out internally (by elec
tronic means) and the result is being reporteddigitally.There is still uncertainty in the last
digit. On an electronic centigram balance, for example,the mass of a rubber stopper might
be measured as 5.67 g. If three observersmeasuredthe mass of the same rubber stopper,
they might obtain readings of 5.65,5.67, and 5.68 g. The uncertainty of an electronicbal
ance measurement is usuallyone unit in the smallest scaledivisionthat is reported—-ona
centigram balancethis wouldbe ±0.01 g.
Accuracyand precisionare two differentwaysto describethe error associatedwith measure
ment.Accuracy describeshow “correct”a measured or calculatedvalue is, that is, howclose
the measuredvalue is to an actual or acceptedvalue.The only wayto determine the accuracy
of an experimentalmeasurementis to compare it to a “true”value—ifone is known!
Precisiondescribesthe closenesswith which.severalmeasurementsof the same quantity
agree. The precisionof a measurement is limited by the uncertainty of the measuring device.
Uncertaintyis often representedby the symbol± (“plusor minus”),followedbyan amount..
Thus, if the measured length of an object is 24.72 cm and the estimated uncertainty is 0.05
cm, the length wouldbe reportedas 24.72±0.05 cm.
Variationsamong measured results that do not result from carelessness,mistakes, or
incorrect procedureare called experimental errors.Experimentalerror is unavoidable.The
magnitude and sources of experimental error should alwaysbe consideredwhen &valuating
the results of an experiment.
I
Experiment Overview
The purpose of this activity is to make measurements using the metric system, to learn the
meaning of significantfigures in the measurements, and to compare the accuracy and preci
sion of laboratorymeasurements.
Pre-Lab Questions
1. Howdoesthe concept of significantfigures relate to uncertainty in measurement?
2. A pipet is a type of specializedlab glasswarethat is usedto delivera specifiedvolumeof
liquid.A5-mL pipet has major scale divisionsmarkedfor everymilliliterand minor scale
divisionsmarked for every0.1 mL. Howwould you estimatethe uncertainty in volume
measurementsmadeusing this pipet? Wouldit be proper to report that the pipetwas
used to deliver3.2 mL of liquid?Explain.
3. Astack of ten musical compact disks is 1.15cm tall.Whatis the averagethicknessof one
disk?Tothe nearest whole number, how many diskswillbe in a stack that is 5 cm tall?
Flinn Chemlopic®Labs
—
Introductionto Chemistry
Page3- Introductionto Measurement
Materials
Balance,centigram (0.01g) or milligram(0.001g) precision
Beaker,50-mL
Graduatedcylinders,10-,25-, 100-,500-, and 1000-mL
Metric ruler, markedin millimeters
Nickels,5—6
Pennies, 5—10(at least one ofwhich was minted in 1982 or before)
Pipet, Beral-type
Water
Safety Precautions
The materialsin this lab activity are considerednonhazardous.Always wear chemical
splashgoggleswhen working in the laboratorywith glassware,heat, or chemicals.
Procedure
Part A. Volume Measurements
There are five graduatedcylinders,each labeledand each containing a specifI quantity Of
liquid to which some food coloringhas been added to make the volume easierto read.
1. Recordthe capacityand the major and minor sêaledivisionsof each graduatedcylinderin
DataTableA.
2. Measurethe volumeof liquid in each cylinderand record the results in DataThb
e
A
Rememberto includethe units and the correct number of significantfigures..
3. Estimatethe “uncertainty”involvedin eachvolumemeasurementand enter the valuein
DataTableA.
Part B. Comparing Volume Measurements
4. Usetap water to fill a 50-mLbeakerto the 20-mLmark. Usea Beral-typepipet to adjust
the water leveluntil the bottom ofthe meniscusis lined up as preciselyas possiblewith
the 20-mLline.
5. Pour the waterfrom the beakerinto a clean, 25-mLgraduatedcylinder.Measurethe vol
ume of liquid in the graduatedcylinderand record the result in DataTableB. Remember
to includethe units and the correct number of significantfigures.
6. Transferthe liquid from the 25-mLgraduatedcylinderto a clean, 100-mLgraduated
cylinderand againmeasure its volume.Recordthe result in DataTableB. Discardthe
water into the sink.
7. Repeatsteps 4—6two more times for a total of three independentsets ofvolume measure
ments. Drythe beakerand graduatedcylindersbetweentrials. Recordall results in Data
TableB.
8. Calculatethe average (mean) volumeofwater in both the 25- and 100-mLgraduated
cylindersfor the three trials. Enter the results in DataTableB.
Introductionto Measurement
IVinIIIIIII
!,*J,
,44.t
IIItç
t’ni,,iit
I
7J(
4
*
the Diameter and Thickness of a Coin
i
in Data TableC. Report each measurement or calculationto the proper num
,
i,ii
a/I cIa/
1,u/icant figures.Don’tforgetthe units.
r at .u
Part C. Measuring
¶I•
Usea metric ruler to measure the diameterof a penny.Recdrdthe measurementin both
centimeters,(cm) and millimeters(mm).
10. Estimateand record the uncertainty involvedin this measurementof length.
11. Makea stack of pennies that is as close as possibleto 10millimetersin height. Count the
number of pennies used to makethis 10-mmstack and record the result.
12. Measureand recordthe preciseheight of the stack of pennies in both centimeters and
millimeters.Dividethe measuredheight of the stack of penniesby the number of
to calculatethe “averagethickness”of a penny in millimeters.Recordthe result.
pennies
•
13. Measurethe thickness (in millimeters)of one penny chosenat random from your stack
and enter this valuein DataTableC.
14. Repeatsteps 9—12using nickelsinsteadof pennies.
• Part D. Mass Measurements
15. Obtainfivepenhiesand recordthe year in which each pennywas minted. Measurethe
mass of each penny individuallyusing a centigram or milligrambalance.Recordthe
results in DataTableD.Note: Makesure that at least one of the pennieswas minted in
1982or before.
Flinn Chemlopic®
Labs Introductionto Chemistry
—
Page5-Introduction to Measurement
Name:
_______________________
Class/Lab Period:
____________
Introduction to Measurement
Data Table A. Volume Measurements
Graduated
Cylinder
A
MajorScale
Divisions
Capacity
MinorScale
Divisions
Volume,of
Liquid
Estimated
Uncertainty
S
B
C
,D
.
E
Data Table B. Comparing Volume Measurements
MeasuredVolumeof “20 mL” of Water
Trial
25-mLGraduatedCylinder
100-mL GraduatedCylinder
1
5
2
3
Average
Data Table C. Measuring the Diameter and Thickness of a Coin
Diameterof Coin
Centimeters
Penny
Millimeters
Uncertainty
Centimeters
Nickel
Millimeters
Uncertainty
Introductionto Measurement
Numberof
Coinsin
10-mm Stack
Measured
Thickness
of Stack
Average
Thickness
of Coin
Measured
Thickness
of Single Coin
—
Introduction to Meosurernent
Page6
Data Table D. Mass Measurements
Penny
Mass
YearMinted
1
2
•3
.
4
5
Post-Lab Questions (Usea separatesheetof paper to answer the followingquestions.)
betweenthe scaledivisionsmarked on the graduated cylindersin
hip
1. Whatis the relation
Part A’ançlthe estimateduncertainty in volume measurements?
2. Which graduatedcylinder(s)gave the most precise volumemeasurement?Doesthe
• number bfsignificantfiguresallowedfor each volume measurementin Part Areflectthe
precision of the graduatedcylinders?
3. It is common td get differentvolume readings for each container in Part B.What explana
tion can you offerfor an apparent decreaseor increase in volume?
4. For both the 25- and 100-mLgraduated cylinder measurements in Part B, calculate
the deviation of each measured volume from the average. The deviation is equal to
the absolute value of the difference between each measured volume and the average
as follows:
deviation = Imeasuredvolume
—
averagevolume I
5. Calculatethe averagedeviationfor both the 25- and 100-mLgraduatedcylindermeasure
ments in Part B.The averagedeviationis equal to the sum of the individualdeviations
from the averagedividedby the number of measurements.Reportthe volume measure
ments for both cylindersin the followingform:
average ± average deviation
6. The averagedeviationshowshow precisea series of measurementsare. Comparethe preci
sion of the volumemeasurementsobtained using the 25-and 100-mLgraduatedcylinders.
Dothese values reallyrepresent the precisionof the graduatedcylindersthemselves?
7. Assumethat the averagevolume found using the 25-mLgraduatedcylinder in Part B is
the “true” or acceptedvalueof the volume.Calculatethe percent error for the average
volumeofwater measured using the 100-mLgraduatedcylinder.The percent error is
calculatedas follows:
Imeasuredvalue acceptedvalue I
x 100%
acceptedvalue
—
percent error
=
8. Doespercent error measureaccuracyor precision?Explain.
Flinn Chemlopic®Labs
—
Introductionto Chemistry
Page7- Introduction to Measurement
9. Are the averagethicknessand measurea thickness ofthe coins measuredin Part C the
same?Whatfactorsmight explainthe difference?Whichmethod do you think givesa bet-.
ter estimateofthe true thicknessof a coin?
10. Comparethe massesof the pennies measuredin Part D.Areall of the massesthe same,
within the limits of uncertainty in the balancemeasurement?Arethere apparent differ
ences basedon when the pennieswere minted?
11. Howdoesthe mass of a penny minted in 1982or beforecomparewith the mass of newer
pennies.Tryto explainthe difference.
Introductionto Measurement
ThePaperAirplaneLab
In class,you’velearnedthatthe scientificmethodallowsyou to solveproblems
throughfocusedtrialanderror. Inthis lab, youwillbe usingthescientificmethod
to figureout howto makeanairplanethatfliesfartherthananybodyelse’s.
:
Prelab
Listthe six stepsof the scientificmethodin order:
.
.
:
Procedure
1)
Makea paperairplaneof the type you are mostfamiliarwith. (If you don’t
will showyou.) With
knowhowto makea paperairplane,your instructOr
yourlab partner,measurehowfar the paperairplanegoeseachtime you
throwit.
Distanceof throw1:
Distanceof throw2:
____________
Distanceof throw3:
Distanceof throw4:
Distanceof throw5:
____________
Average distance of all five throws:
© 2000 CavalcadePublishing(teachers visit www.cavalcadepublishing.com,students visit www.chemfiesta.com)
‘2)
Whatyounowwantto do is makea guessaboutwhatmightmakethe
paperairplanesfly better. Thiscouldbe a differentmethodof foldingthe
planes.or someadditionto the planedesign. You maychangewhatev
r
you like, butrememberto ànlychangeone thing.
In the spacesbelow,writedownhOwyou thinkthat you cangeta better
flight distanceusingthe scientificmethod.
.
Purpose:
Hypothesis:
.
.
Materials:
Procedure:
Results:
Conclusion:
© 2000 CavalcadePublishing (teachers visit www.cavalcadepublishing.com,students visit www.chemfiesta.com)
,
ID
Postlab Questions
:
1)
Was yourhypothesisdisprovedor confirmed?Explain,usingthe
informationyouobtainedin yourexperiment.
2)
Do you thinkthis is howscientistsconducttheir own research?Explain
whyor why not.
3)
Explainwhy it wasimportantthat you changeonlyonethingbetweenyour
initialexperimentand yourlatercxperim&nt.
4)
Do you thir
k
thatthescientificmethodis a goodwayto makescientific
discoveries?If so, thenexplainwhy you thinkthisis. If not,explainwhy
you thinkit isn’t.
© 2000 CavalcadePublishing(teachersvisit www.cavalcadepublishing.com,studentsvisit www.chemfiesta.com)
Date
Name
_________________________________
•
___________
of a
QualitativeObservatgons
Chemical
Reaction
Class
___________
Lab
Textreference:Chapter1. pp. 2—4
PreLabDiscussion
.
Scientists rely heavily on experimentation. A good scientist must observe
and interpret what is happening. Observing means using the senses:
seeing, smelling, touching, hearing, and sometimes tasting NEVERTASTE
CHEMICALSUNLESSINSTRUCTEDTO BYYOURTEACHER
When scientists make observations, they try to’ be objective. Being
objective means putting aside any preconceived ‘notions. Scientists are
interested in what really occurs, not in what they wish would. occur.
• After observations are made, scientists must make interpretations.
Interpretations are based on previous knowledge and experience. Because
people have different experiences, one scientist may interpret observa
tions in one’ way while another may interpret the same observations to
mean something else. When we interpret, we attempt to make sense out
of observations. Scientists never assume that their interpretations are cor
rect until they test hem fully and repeatedly. After complete testing, sci
entists then come to their conclusions.
In this investigation, you will make some qualitative observations of
a chemical reaction. That is, no measurements will’be made. During a
chemical reaction, one or more substances change into one or more other
substances. The burning of wood, wax, oil, gasoline, and coal are examples
of a chemical reaction known as combustion. The reactiQn you will study
in this investigation is a combustion reaction.
Purpose
Carefully observe and interpret a chemical reaction.
Equipment
beaker, 250-mL
Erlenmeyer flask, 125-mL
glass square
microspatula
metric ruler
microslide
rubber stopper
safety goggles
lab apron or coat
Materials
candle (2-cm diameter)
matches, 2 or 3
toothpicks, 2
limewatér solution
Safety
W
string
aluminum foil
cobalt chloride paper
fl A
In this experiment, you will be working with an open flame. Tie back
long hair and secure loose clothing. Also, wear safety goggles and a lab
1
apron or coat at all times when working in the lab. Be sure all matches
and burned materials are completely extinguished before they are
discarded.
Procedure
Record observations for each steip.
1. Note appearance, odor, and feel of the unlighted cnd1e.
2. Heat the bottom of the candle and secure it to a glass square
on your lab bench. Light the candle and allow it to burn for
several minutes. Note any changes. Bnefly describe the burning
candle.
3. Blow out the flame and immediately place a lighted match in
the “smoke” about 2 cm above the wick. See Figure 1-1.Note
the result.
I.
Figure1-1
4. Use a microspatula to transfer a small amount of liquid from
the bowl of the candle onto a microslide. Try to light it and note
the result.
5. Place a toothpick into the soft candle next to the unhighted
wick to form a wooden wick. Light the toothpick and note the
result.
6. Place a length of string about 4 cm long on the glass square.
Light it and observe its behavior.
7. Make a slit in a small piece of aluminum foil. See Figure 1-2.
Light the candle. Place the foil between the base of the flame
and the liquid in the candle bowl. Note the behavior of the flame.
aluminumfoil
Figure 1-2
2
Name
.
I
/(3
of a Chemical
Reaction(continued)
QualitativeObservations
Figure 1-3
8. Invert a 250-mL beaker over the lighted candle. See Figure
1-3. Note any substance that collects on the inside of the beaker.
Test the liquid with cobalt chloride paper.
9. Invert a 125-mL Erlenmeyer flask over the lighted candle for
several minutes. Remove the flask, turn it right side up, and add
about 10 mL of the clear limewater solution. Stopper and shake
the flask. Note any change in the limewater solution.
‘I
II
ii
Observationsand Data
.
3
1*
.
:
‘
..I
,
I’
.
and Questions
Condusions
1. What phases (solid, liquid, gas) are present in the unlighted candle? In
the burning candle? Which phase appears to take part in the chemical
reaction?
4
Name
.
I
of a Chemical
Reaction(continued)
QualitativeObservations
.2. What part does the wick play in the burning of.the candle? What prop
erties should the wick have? Explain the result when aluminum is placed
between the liquid and the wick. Is the wick part of the chemical reaction?
I
3. What two substances are indicated by the cobalt chloride and limewater tests? Is it possible that other substances are produced when the
candle burns? Explain.
—_____________________________
.
4. A source of energy is needed to start the burning of the candle. What
energy source is used? Did the reaction give off or absorb heat?
5. Give an example, illustrating the difference between observation and
interpretation.
5
•
•
Name
Date
___________________________________
Class
____________
of a
Observations
Quantitative
Chemical
Reaction
____________
,
Lab2
Textreference:Chapter 1, pp. 2—4
Pre-LabDiscussion
•
•
•
Most experiments require the investigator to. make some quantitative ob
servations, or measurements. The numerical values of these measure
ments are called data. The most frequently measurçd quantities in the
chemistry laboratory are mass, volume, and tempez
ature.
When conducting an experiment of a quantitative ‘nature, the first step
• in the procedure is to ‘nake and record measurements of the materials
that are being investigated. If the materials take part in a chemical reaction
(undergo chemical change), many, if not all, of the initial measured values
probably will change. The nature and extent of these changes often help
the investigator to understand what is taking place. Some of these
‘changes, sudh as temperature change, can be measured and, recorded as
the reaction is taking place. When the reaction is ended, measurements
again are made and recorded. The collected data from all of these mea
surements provide an overall record of what quantitative changes took
place during the reaction.
When making measurements, you should keep in mend that the
’instrilments
numerical values can be only as accurate as the
used to make
the measurements. These values also are affected by the care and skill of
the person using the instruments. As you gain more experience in the
laboratory, you will become more familiar with tbe limitations and accu
racy of the various instruments you use. You also will become more skill
ful in the use of these instruments and in carryng out various
activities that are essential to a successful investigation.
Scientists must be imaginative. In many cases, they must devise their
own experiments and decide what measurements will provide useful infor
mation. In this investigation, you will make measurements to determine
the ‘effects of a chemical reaction (combustion). You then will be asked
to decide how these measurements can be used to extend your under
standing of the reaction.
Purpose
Make a quantitative investigation of a chemical reaction.
Equipment
laboratory balance
ring stand
iron ring
wire gauze
thermometer
beaker, 250-mL
graduated cylinder, lOO-mL
watch or clock with second hand
glass square
safety goggles
lab apron or coat
Materials
candle (2-cm diameter)
matches, 2 or 3
7
/7:
Safety
In this experiment, you ‘v(rillbe working with an open flame! Tie back long
hair and secure loose clothing. Also, wear safety goggles and a lab apron
or coat at all times when working iii the lab. Be sure matches are com
pletely extinguished before they are discarded.
Procedure
1. Find the mass of the candle.
2. Measure exactly 100 mL of tap water in a graduated cylinder.
Pour the water into a 250-mL beaker and place the beaker on a
wire gauze as shown. in Figure 2-1. Measure the temperature of
the water
thermometer
containing 100mL
of water
iron
ring
candle
glass
square
ring stand
Figure 2-1
3. Light the candle and place it on the glass square, as shown.
Adjust the height of the ring so that the flame is 2 cm below the
base of the beaker. Using the candle, heat the water for exactly
10 minutes. Extinguish the flame and measure the temperature
of the water and the mass of the candle.
4. Relight the candle and repeat steps 1—3for a second trial.
Observationsand Data
Trial 1
Original mass of candle
Mass of candle after burning
Time candle burned
Original temperature
Final temperature
Time water heated
8
of wat.er
of water
Trial 2
Name
•
2 Quahti:tative Observationsof a ChemicalReaction (continued)
Calculations
For each trial, find:
ThaI 1
Trial 2
1. The change in the mass of the candle
2. The change in the mass of the candle per minute
3. The change in the temperature of the water
4. The change in the temperature othe
water per minute
andQuestions
Conclusions
1. Compare your trial results and calculations
with those of other lab
teams. Are your results exactly the same? How do you account for any dif
ferences in data? If one set of data differs from another in an experiment,
does this mean that one or both sets are wrong? Explain your answer.
.
______________
9
2. What doesthe term tate mean? a. What was the rate
candle? b. What was the rate of heating of the water?
.
bEburning
of the
I
3. Explain how the heat from the combustion reaction iS related to the
temperature change of the water.
4. Outline a laboratory
more heat—a gram of
type of experiment be
better fuel? What other
procedure that would determine which produces
candle wax or a gram of alcohol. How could this
used to decide which substance would make the
I?
factors might enter into choosing a fu
t
.
10
$
Name
•
___________________________________
Date
____________
Mass
Measuring
Class
____________
Lab3
Text reference: Chapter 2, pp. 15—17
Pre-LabDiscussion
For laboratory work in a general chemistry course, three basic types of
measurement using the lab balance should be mastered. These are:
measuring mass directly, “measuring out” a specific nass of a substance,
and determining mass by difference. These three types of measurement
and the techniques for making them are briefly descrihed in the following
paragraphs. “
‘1. Measuring mass directly. Direct measurement is used to deter
mine the mass of a beaker or flask or similar object. This is the simplest
type of measurement made with the balance. In a direct measurement,
the object with a mass to be measured is placed on the balance pan,
riders are moved into positions along the beams
and the appl
opriate
until the pointer is balanced at the zero point. The mass of the object is
read directly from the positions of the riders on the beams.
This technique often is used to
2. “Measuring out” a substance.
obtain a desired mass of a solid chemical, such as table salt, that exists
in a granular or crystalline state. To do this, the balance riders are preset
asured
out then is
to the desired mass reading. The substance being m
added to the balance pan until the pointer is balanced at the zero point.
Keep in mind, however, that chemicals should never be’ placed directly
on the balance, pan. A piece of paper or a contairker of some kind should
be placed on the pan to receive the substance being measured. The
preset mass must then be equal to the mass of the’paper or container
plus the mass to be measured out. This technique can be used for liquids
as well as solids. The liquid is poured into a container resting on the
balance pan with the preset mass equal to the mass of the empty con
tainer plus the mass of the liquid to be measured out.
3. Determining mass by difference.
Finding mass by difference
is an important laboratory technique. As the name suggests, this tech
nique involves subtraction. One common use for this technique is to
determine the mass of a quantity of a substance, such as a liquid, that is
in a container. In a case of this sort, it is necessary to subtract the mass
of the empty container from the combined mass of the container and the
substance. Another important use of this technique is to measure
changes in mass that occur during a chemical reaction.
In this experiment, you will learn how to perform all three types of
measurement described here. In addition, you will gain valuable practice
in using the laboratory balance and in handling different materials and
apparatus.
,
•
‘,
Purpose
Practice the various techniques of measuring masses using the lab bal
ance. Gain experience in the techniques of handling laboratory materials
and equipment.
11
Equipment
lab balance
watch glass
beaker, 150-mL
beaker, 50-mL
microspatula
graduatedcylinder, 100-mL
timer (watch or clock)
safety goggles
lab apron or coat
Materials
sodium chloride (NaCI)
calcium chloride (CaC1
2)
filter paper
toy balloon
coins (1 penny, 1 nickel)
Safety
Do not allow lab chemicals to come in contact with your sdn. Calcium
chloride (CaC
I2)
removes moisture from the skin and can produce irrita
tion and a burning sensation. If contact should occur, immediately flush
the area with cold water. Always. wear safety goggles and a lab apron or
coat when working in the Jab.
Procedure
PARTA MEASURING MASS DIRECTLY
1. Check your alance to make sure that the painter is prop
erly “zeroed.” If an adjustment is necessary, consult your
teacher.
2. Place a penny on the balance pan. Move the rider(s) until
the pointer isbalanced (zeroed). Record the mass of the penny.
3. Repeat step .2 for objects listed below. Record the mass of
each object.
a. a nickel
b. a watch glass
c. a 150-mL beaker
d. a I00-mL graduated cylinder
PARTB MEASURING OUT A SUBSTANCE
4. Place a piece of filter paper on the balance pan. Move the
rider on the front beam until the balance is zeroed. Record this
reading.
5. Move the riders until they read exactly 7.50 g more than the
reading you obtained in step 4. Record this setting.
6. Obtain a quantity of sodium chloride (NaCI) on a piece of
paper. Using your microspatula, add this substance to the filter
paper on the balance pan until the pointer is balanced. (If you
add too much, remove enough with your microspatula to make
the pointer balance.) Discard the NaCI and filter paper as in
structed by your teacher.
7. Place a dry 50-mL beaker on the balance pan. Move the
riders until the pointer is balanced. Record this reading.
8. Move the riders until they read exactly 22.0 g more than the
reading you obtained in step 7. Record this setting.
12
Name
•
3
__________
_______
MeasuringMass (continued)
9. In a 100-mL graduated cylinder, obtain exactly 30.0 mL of
cold tap water. Stowly and carefully pour water from the
graduated cylinder into the beaker on the balance pan until the
pointer is balanced. Avoid splashing water onto the pan. Note
and record the volume of water remaining in the graduated cylinder. Discard the water and dry the beaker.
PARTC DETERMININGMASS BY DIFFERENCE
•
10. Measure and record the mass of a watch glass:
.
Ii
Using the procedure describeçl by your teacher, obtain 20—
25 crystals of calcium chloride (CaCl
2)
on a piece of paper. Care
fully transfer the crystals to the watch glass.
12. Measure and record the combined mass of the watch glass
and calcium chloride. Note and record the time you make this
measurement.
13. Using your microspatula, spread the crystals out on the
watch glass. Study the crystals and record your observations.
Set the watch glass and crystals aside to be reexamined later.
.
14. Measure and record the mass of a piece of filter paper.
15. Obtain a quantity of sodium chloride (NaCl) on a piece of
paper. Using your microspatula, place one heaping scoop of the
NaCI on the filter paper. Measure and record the combined mass
of the filter paper and Na(.l.
16. Add a second heaping scoop of NaCI to the sample on the
filter paper. Measure and record the combined mass of the filter
paper and the two scoops of NaCI.
17. Add a third heaping scoop of NaCI to the sample on the
filter paper. Measure and record the combined mass of the filter
paper and the salt. Discard the salt and the filter paper.
18. Measure and record the mass of a l50-mL beaker.
19. In a I0O-mL graduated cylinder, obtain exactly 30 rnL of
cold tap water. Carefully pour this water into the beaker. Meas
ure and record the combined mass of the beaker and water.
20. Inflate a toy balloon and tie off the open end so that no gas
can escape. Measure and record the mass of the inflated balloon.
21. Puncture the balloon and allow all the gas t.o escape. Meas
ure and record the mass of the deflated balloon. Discard the
balloon.
22. Reexamine the calcium chloride crystals on the watch glass
that you set aside earlier. Record your ol)servat.ions.
•
23. Measure and record the combined mass of the watch glass
and its contents. Not.e and record the time of this measurement.
Discard the calcium chloride and clean and dry the watch glass.
13
Observationsand Data
PARTA
•
1. Mass of a penny
g
2. Mass of a nickel
g
3. Mass of a watch glass
g
4. Mass of I50-mL beaker
g
5. Mass of I00-mL graduated cylinder
g
PARTB
6. Mass of filter paper
g
7. New setting (filter paper
+
7.50g)
g
8. Mass of 50-mL beaker
g
9. New setting (50-mL beaker
+
22.0 g)
g
mL
10. Volume of water remaining in graduated cylinder
PARTC,
11 Mass of watch glass
g
12. Mass of watch glass
±
I2
CaC
g
12
13. Time of measurement
14. Mass of filter paper
g
15. Mass of filter paper
+
I scoop of NaCI
g
16. Mass of filter paper
+
2 scoops of NaCl
g
17. Mass of filter paper
+
3scoopsofNaCl
g
18. Mass of 150-mL beaker
g
19. Mass of 150-mL beaker
+
30 mL of water
20. Mass of inflated balloon
21. Mass of deflated balloon
22. Mass of watch glass
23. Time of measurement
14
-f
(after sitting)
2
CaCl
22
g
_g
g
g
Name
*
•
3
24
Measuring Mass (continued)
Observations
Calcium chloride crystals
a. initial examination (step 12):
b. later examination (step 22):
Calculations
e
of water added to the beaker
1. Calculate the vo1un
9..
in step
volume remaining in
(initial volume
graduated cylinder)
—
• 2. Calculate the mass o1 I mL of water.
(mass of water ÷ volume of water)
g
3. Using the mass-by-difference technique,
calculate the mass of:
2
crystals added to the watch glass
a. CaCI
(step 12— step 11)
g
b. NaCl(1 scoop)(step
15
—
step 14)
g
c. NaCI (2 scoops) (step 16
—
step 14)
g
d. NaCJ(3 scoops) (step 17
—
step 14)
g
19— step 18)
g
p
e. 30 mLof tap water (st
l9
f. moisture absorbed by Ca(.
(step 22 step 12)
crystals
g
—
4. Calculate the difference in mass between the
inflated balloon and the deflated balloon
(step 20— step 21).
5. Calculate how much time, in minutes,
elapsed between the two measurements
of the CaCJ
2
23
step 13).
(step
—
g
_________
m
15
Conclusionsand ‘Questions
1. In steps 15—17of the procedure, you measured the combined masses
of a piece of filter paper plus one, two, and three heaping scoops of NaCI,
respectively.
From Calculation 3(b), what was the mass of the first scoop of
NaCI?
b. From your data, calculate the masses of: the second scoop of NaCI
(step 16 step 15) and the third scoop of NaC1 (step 17 step 16).
c. Compare the masses of each individual scoop of NaCI as deter
mined in (a) and (b) above. Are the three values identical? If not, how
do you account for the differences?
‘
a.
—
—
,.
‘
2. What measurements would you make to determine the average mass of
a heaping microspatula of NaCI? ‘How would you use these measurements
to arrive at an average mass?
3. The difference between the mass of the balloon when inflated and its
mass after being punctured is not an accurate determination of the mass of
the gas in the inflated balloon. Why is this?
16
Name
.
3
,A.
MeasuringMass (continued)
4. Suppose you were asked to measure out 5 grams of calcium ‘chloride.
Briefly describe how you would make this measurement.
5. Suppose that you wanted to know the mass of a quantity of orange
juice that was poured into a drinking glass. Describe how you would
determine this mass.
6. A beaker contains a quantity of a liquid. You want to know the com
bined mass of the beaker and the liquid. Describe how you would go about
making this determination.
7. Suppose you were asked to compare the mass of a nickel and
of NaCI crystals.
a. Which method would you use to determine the mass of the
b. Which method would you use to determine the mass of
crystals?
c. Which of the two measurements is likely to be the more
one? Explain.
a sample
nickel?
the NaCl
accurate
.
17
Name
Date
_________________________________
Class
___________
___________
t27
S
Uncertaintyin Measurement
Lab
Textreference:Chapter3. pp. 50—55
Pre-LabDiscussion
Laboratory measurements are never exact. The uncertainty of a measured
quantity depends on the skill of the researcher and the limitations of the
measuring instrument. These two factors determine the accuracy of a
,measurement. Accuracy reflects the nearness of a measurement to the
actual or accepted value. When reporting experimental data, researchers
record all digits they are certain of plus one digit they are uncertain of.
These are known as significant figures. The uncertainty of the measure
ment too is indicated. This depends on the size of the smallest increment
on the scale of the instrument.
When gathering data to solve a problem, skilled experimenters select
instruments that will produce the desired level of acc
Thus, they
iracy.
must know the level of accuracy possible ,with calibrated instruments
commonly found in laboratories. By studying some simple laboratory
equipment, a better understanding of uncertainty in measurement can be
achieved.
Purpose
Examine some calibrated measuring instruments, make some measure
ments, and record data. Report data using the correct number of signi.fi
cant figures and scientific notation. Indicate the uncertainty of the meas
urements.
Equipment
laboratory balance, four-beam
centimeter ruler
thermometer (O°—120°C)
graduated cylinder, 100-mL
calibrated pipets, burets, l0-mL
graduated cylinder, triple-beam
balance, meter stick
safety goggles
lab apron or coat
Materials
a coin, such as a nickel
piece of paper, less than 10-cm square
water
Safety
.
Handle glassware with care, especially thermometers, pipets, and burets.
They are fragile and easily broken. Always wear safety goggles and a Jab
apron or coat when working in the lab.
19
Procedure
Record all measurements in the data table. Report data to the correct
number of significant figures and using scientific notation. Indicate the
urements.
uncertainty of the mea
1. Using the four-beam balance, measure and record the mass
,1
ofthe coin.
the
2. Using the centimeter ruler, measure
length of on side of
the piece of papers
3. Using the thermometer, measure and record the temperature
of the air in the laboratory.
4. Using the i0O-mLgraduated cylinder, measure and record
the volume of a sample of water between 0 and 100mL.
5. Make additional measutements, as time permits, using cali
brated pipets, bufets, a 10-mLgraduated cylinder, atriple-beam
balance, a meter stick, and other devices provided.
Observationsand Data
.
DATATABLE
Instrument
20
,
.
S
Measurement
four-beambalance
massof coin
centimeterruler
paperedge
thermometer
O°—120°C
temperature of room air
100-migraduatedcylinder
volumeof water
Data
Uncertainty
Name________
•
4
______________________________
Uncertainty in Measurement
(continued)
and QuestiOns
Conclusions
1. What’ is the difference between precisiop and accuracy? Is a precise
measurement always accurate? Explain.
.
T’.
EE
2. A square piece of paper has an edge measuring 7.06 cr (using a centi
meter ruler). How many significant figures should appear in the calculated
Write the result correctly.
)?
surface area (A = S
3. Why do liquid vitamins for babies come wit.h a dropper pipet rather
than a container marked with graduated lines, as on a graduated cylinder?
Explain in terms of accuracy.
.
21
How muchsugar is in yourbubblegum?
A PercentComposition
Lab
t3O
Name_____
bate___________
Thtroductjon: It is sometimes appropriateto analyze mixtures or
to determine the properties of its various components.If
compounds
youread the label on the packageofany food,the ingredients in that
food must be listed in order according to their mass.,The most
abundantcomponent is listed first. In this experiment, you will
determinethe percentageof sugarin a pieceof bubble gum.Basedon
what the label says, you should be able to make an estimate
(hypothesis)as to whatyoushouldexpectto find inthis lab.
1 pieceof bubblegumwithsugar(Bubblicious,,
Materials:
•
Bubble Yum,Bazooka,
etc.)
Balance
Hypothesis:
thCLwhai percent of your gum is made up of sugar?
Onthe bc:ickof this pagedesigna procedure.Thenmakea datachart
of sugarin your
to showdata. Finally,calculatethe percent composition
piece of gum.
Twopointsto consider:(1) The wrapper hasa mass.(2) Howlongwill
youchewyourgum?
*
(31
Groupmembers:
Questions for PercentSu9ar’in BubbleGumLob
1. What was the mass of the 1 pieceof gumbefore chewing?
2. What wasthe massof 1 pieceof gumafter chewing?
grams
grams’
ed
3. What was the massof dissoh
sugar in1 pieceof gum?
grams
4. What isthe percentageof sugarin1 pieceof gum?ShowyourWork
ow.
b
•
5. Howdidthe percent that youcalculatedabovecompareto your
hypothesis?
6. Howlongdidyouchewyourgum?.
.
minutes
7. Why didyoudecideto chewyourgum.for this particular lengthof time?
formula: )?
8. What isthe molecular
C1
2
H
1
0
2
massof sugar (molecular
5howyour workbelow.
Besure to handin:
• Thesequestionsanswered
• The procedure
• Thedatachart
I,
Name_____________________
Bayport-BluePointHS
ChemistiyLab••
RATURE
ThMP
MIXING
MATERiALS:
beakers
2-400rril..
2 5OmLbeakers
beaker
1- lOOrnL
3‘thermometers
hotplate
water
ice
PROCEDURE:
200mL
1. In a lãrgébeaker,boilapproximately
ofwater.
2. In a second
200mLof
beaker,addiceto approximately
water.
3. Foreachofthethreetrials,besureto recordtheexact
ofboththehotandcoldwaterthatyou
temperature
onthedatachartto
use.Also,shadethebeakers
theamountofwaterinthebeaker.
indicate
a. Trial1:
I. Intoa smallbeaker,pour3OmLofhotwater
andrecordthetemperature
ii. Intoa second
smallbeaker,pour3OmLof
coldwaterandrecordthetemperature
iii. Predict
thevolume& temperature
thatyou
willgetwhenyoumixthem
1133k
iv. Mixthe 2 volumesofwater. Immediatel
’
recordthe temperature
andthevolumeih
the“Actual”column.
b.Trial2:
i. Repeatthe aboveprocedureusing2OmLof,
hotwaterand4OmLof coldwater
c. Trial3:
i. Repeatthe aboveprocedureusing4OmLof
hot waterand2OmLofcoldwater
JtJ
DATA:
HOT
COLD
3OmL
3OmL
PREDICThD
ACTUAL
mL
•••;
mL
,
oc
4OmL
mL
oc
mL
oc
4OmL
oc
2OmL
oc
mL
oc
mL
.oc
oc
oc
oc
2OmL
,
(31
QUES1IONS:
Defineheat. Explain
the
1. Definetemperature.
between
& heat.
difference
temperature
2. Howmanykilojoules
(k.J)arein1.0Joules
(3)?
3. Howmany)areinlkJ?
thefollowing
Celsius
to Kelvin.
4. Convert
temperatures
formula
ShOW
& substitution.
ALLwork,including
c. —20°C
a. 24°C.
d. 18°C
b.30°C
e. 141°C
thefollowing
Kelvintemperatures
toCelsius.
5. Convert
formula& substitution.
ShowALLwork,including
a. 370K
c. 418K
b.241K
(33
d. OK
e. 81K
6. Whatdowemeanby“fixedpoints”
ona temperature
scale?Whatarethefixedpoints,indegrees,
onthe
& Celsius
scales?
Kelvin
‘7. If thetemperature
of a’substance
from
wereincreased
32°Cto 48°C,howmuchwouldthetemperature
increase
ontheKelvinscale?EXPLAIN
youranswer!
vit:
Act
•1•
(36
Paper Clip Formulas
formulas and classifying matter
l
Purpose: To give students practice interpreting chernic
Materials: 3 containers .containing three different size paper clips. The paper clips
represent atoms of three different types. The following “atomic” symbols will represent
each type of “atom”.
R = regular plastic coated
Jb =jumbo
Sm = small metal
Procedure:
1. You are to make the substances listed below and place them on a piece of paper
lab&e (“chernica) formula style”).
rh
piv
substance 1:
2
JbSm
substance 5:
R
2
Jb
4R
substance 2:
Sm
2
Jb
+
2
R
substance 6:
3ubstance3:
2 Sm
+
3
R
substance 7:
22
Sm)
3
Jb(R
Substance 8:
RSm
2
Jb
4
‘I.
substance 4:
3 Sm
2
Answer these questions on what you have made.
I. Which of the above substances are elements?
2. which of the above substances are compounds?
3. Which of the above substances are mixtures?
4. How many total atoms are there in each of the above substances? Put the total under
each model.
5. Which represent diatomic molecules? triatomic molecules?
6. How many molecules are there in each of the above substances?
Reference: Adapted from Elna , Woodrow Wilson Chemistry Institute, 1995.
r lem- I.iflgt
• separate a Mixtuj
MISSION POSSIBLE:
to separatethe contentsof mixturein the cupif
Youare challenged
andare now
possible.Eachof the cupsstarted with4 substances
combinedintoone mixture.
The contentsare: Sand
Iron Filings
PoppySeeds(like the roll)
.5alf Crystals
Thekey thing to rememberabout mixtures is that you start with somepieces.
combinethem, andthen you cando somethingto pull those piecesapart again.,
Youwind up with the samemolecules(in the sameamounts)that you started
with.
Write a lab report with the following information:
Objective
Methodsand Materials
•
tesults
Conclusionor Summary
Youand your group write an objective. State howyou are goingto
:
Step ONE
reach your objective (method)andthe materials neededto reach it.
Ask mefor thematerials
:
(f possible).I’ll giveeverything I haveto
Step TWO
you.
:
Howdid your methodswork, (Results)canchangesbe made?
Step ThREE
What did you find? bid you support or disproveyour objective, how
:
Step FOUR
do you know.
Name
Class
Date
What
colors
candli?
areinqour
(-3
Yellow dye #5 is an artificial food coloring approved by the FDA, but some
people are allergic to this dye. Many candies contain Yellow #5 as part of a
mixture to color the candies. Dye mixtures can be extracted from the candy
and separated into their component colors using paper chromatography. The
yellow food coloring that you buy in the grocery store: contains Yellow #5 and
can be used as a reference standard.
Separations by paper chromatography are possible because different sub
stances have different amounts of attraction for the paper. The greater the
attraction the substance has for the paper, the slower it will move up the
paper with the solvent.
PROBLEM
Are,there any colored candies ‘that a person
with an allergy to Yellow #, can safely eat?
OBJECTIVES
C)
C
• Observe separation of colors in dye mix
tures.
• interpret data to determine which can
dies contain Yellow #5.
a,
0.
E
C
C)
I
a,
0
0
U,
:0
HATEAIALS
10 cm X 10 cm piece
of Whatman #1
filter paper.
large jar with lid
colored candy
yellow food coloring
small plastic cup
water
salt
toothpicks
ruler
U,
PROCEDURE
I
,
0
C,
U,
8
0
U
-C
a
0
C)
1. Record your observations in the data
table.
2. Make a fine line with a pencil about
3 cm from one edge of the piece of filter
paper.
3. Place a small amount of water in a plas
tic cup.
4. Dip the tip of a toothpick into the water.
5. Dab the moistened tip of the toothpick
onto a piece of colored candy to dissolve
some of the colored coating.
Chemistry:Concepts and Applications
AND
CHEMLAD
MINILAD
WOANSHE
CHEMLR
.9
6. Place the tip of the toothpick with dye
onto the filter paper to form a spot along
the pencil line.
7. Remoisten the tip of the toothpick, and
dab the same piece of candy to dissolve
additional coating. Place the tip of the
toothpick onto the filter paper on the
same spot made in step 6. Repeat this
step until a concentrated spot is
obtained.
8. Using a new toothpick and fresh water,
repeat steps 4 to 7 with a different col
ored piece of candy. Make a new spot for
each piece of candy, and keep a record in
your data table.
9. Dip a fresh toothpick into a drop of the
yellow food coloring to be used as a refer
ence standard. Make a spot along the
pencil line and mark the location of the
reference spot.
10. Carefully roll the paper into a cylinder.
The spots should be at one end of the
cylinder. Staple the edges. Avoid touch
ing the paper.
11. Add water to the jar to a level of about
1.5 cm from the bottom. Sprinkle in a
pinch of salt. Close the lid and shake.
12. Place the filter-paper cylinder into the
jar so that the end with the spots is clos
est to the bottom of the jar. The water
level must be at least 1 cm below the
pencil line. Adjust the amount of water if
necessary and close the lid.
ChemLab 9’
59
14.
15.
16.
17.
from the top of the filter paper.
Carefully remove the filter paper, open
it flat, and mark the solvent edge (the
farthest point the water traveled) gently
with a pencil. Lay the filter paper on a
paper towel to dry.
For each piece of candy spotted, mea
sure the distance from the original pen
cil line to the center of each separated
spot. Record these data in your data
table. Some candies may have more
than one spot.
Measure and.record the distance from
the original pencil line to the marked
solvent edge.
Record the distance from the original
pencil line to the center of each spot
separated from the reference spot of
Yellow #5.
I
Y
3
HNliI
UNC1UOE
HNU
1. Interpreting Observations Do any of
the candies contain Yellow #5? How can
you tell?
-
.,,
...,-.,.
I.
ODSEAYAT)DNS
DATA
AND
(distance from first pencil mark to solvent edge)
Solvent distance:
.
OriginalSpot
Distance(color1)
Yellow#5 Reference
Distance(color2)
Distance(color3)
w
.
C
(V
0.
E
0
()
I
Candy1
0
(V
Candy2
0
C
0
,
(I
•0
(V
Candy3
I
C)
(V
Candy4
8
C
(V
0
Candy5
>
0.
0
0
Candy6
60
Chemistry:Conceptsand Applications
ChemLab9
2. Comparing and Contrasting Do any
of the candies contain the same dyes?
Explain.
2. Why was it important to use a pencil
instead of a pen to mark the paper?
3. Inferring Which candies would be safe
to eat if you were allergic to Yellow #5?,
4. Designing an Experiment Can you
devise a better way to remove the dye
from the candy and place a spot on the
paper?
3. Why was it important to do the experi
ment in a closed jar?
4. What makes the water move up the
paper?
5. How did the rate of water movement up
the paper. change as the water got high
er on the paper? Suggest reasons why it
changed.
APPLY
AND
ASS[SS
1. On what portion of the paper are the
substances with the greater attraction
for the paper? What conclusions can you
draw about the molecular polarities of
these dyes?
Chemistry:Conceptsand Applications
ChemLab 9
61
Chemicalor PhysicalChangeLab
Usewhatyou’velearnedaboutchemicaland physicalchangesto determineif,
the followingstationsinvolvechemicalor,physicalchanges.Makesure yougive
evidencefor yourdetermination.
Station 1: Heatthe unknownin a crucible
In thisstation,heatthe unknowncompoundin a crucibleuntilyou seea change
take
place.
•
Wasit a.chemicalor physicalchange?
Whatevidencedo you haveto backup your guess?
•Station2: Combinethe two solutions
In thisstation,add ne dropperfulof compoundA intoa 50 mLbeakerfollowed
by onedropperfulof compoundB. Makesureyou usedifferentdroppersfor’
eachsolution.
,
$
,Was it a chemicalor physicalchange?
Whatevidencedo you haveto backup your guess?
Station 3: Heatthe unknown in a crucible
In thisstation,heattwo largepiecesof the unknownin a crucibleuntil you seea
changetakeplace.
Wasit a chemicalor physicalchange?
Whatevidencedo you haveto backup your guess?
© 1999CavalcadePublishing
For chemistryhelp,visitwww.chemfiesfa.com
‘Station 4: Heatthe unknownin a crucible
In thisstation,heatone smallscoopfulof the uhknownin a crucibleuntilyou see
a changetake place.
Wasit a chemicalor physicalchange?
Whatevidencedo you haveto backupyourguess?
Station 5: Combinethe two solutioins
lb thisstation,add onedropperfulof compoundA intoa 50mL beakerfollowed
by onedropperfulof compoundB. Makesureyou usedifferentdroppersfor
eachsolution.
’sicaI
Wasit a chemicalor ph
change?
Whatevidencedo you havetobackup yourguess?
Station6: Heatthe unknown in a crucible
Inthisstation,addten dropsof the unknownto a crucibleand heatovera
Bunsenburner.
Wasit a chemicalor physicalchange?
Whatevidencedo you haveto backup yourguess?
© 1999CavalcadePublishing
For chemistry help, visit www.chemfiesta.com
Page1 ExploringEnergyChanges
-
‘ExploringEnergyChanges
Exothermicand EndothermicReactions
Introduction
The story of chemistry is the story of change—physicalchange, chemical change, and ener
gy change. Energy in the form of heat is exchangedin almost everychemicalreaction or
change in state. Somereactions require,heat in order to proceed. Other reactions release
heat as they take place. In this experiment,we will investigateseveralprocessesin order to
get a feelfor the amount of heat absorbed or releasedin physicaland chemicalreactions.
Concepts
‘
• Thermochemistry
• Heat
‘
,
,
• Exothermicvs. endothermic
•
Energy
•
Temperature
•
Systemvs. surroundings
Background
Thermochemistryis the study ofheat changesthat take place in a change ofstate or a chemical
reaction—heatenergyis eIther absorbedor released.If a processreleasesenergyin the formof
heat, the processis calledexothermic.A prcicèssthat absorbsheat is calledendothermic.How
do we observeor measurethe heat changethat occurs in a physicalor chemicalchange?
from one object to another due to a differencein
Heat is definedas the energy transferr
d
observe
Wedo
not
temperature.
or measure heat directly—wemeasure the temperature
change that accompaniesheat transfer.In a chemicalreaction it is often not possibleto
measure the temperature of the reactants or products themselves.Instead, we measure the
temperature change of their surroundings.
The differencebetweenthe system and the surroundings is a keyconcept in thermochem
istry. The system consists of the reactants and products of the reaction. The solvent, the con
tainer, the atmosphere abovethe reaction (in other words, the rest of the universe)are con
sidered the surroundings. Heat may be transferred from the system to the surroundings (the
temperature of the surroundings will increase)Orfrom the surroundings to the system (the
temperature ofthe surroundings will decrease).
When a system releasesheat to the surroundings during a reaction, the temperature of the
surroundings increasesand the reaction container feelswarm to the touch. This is an
exothermic reaction—the prefixexo- means “out of’ and the root thermic means heat. Heat
flowsout of the sytem. An exampleof an exothermicreaction is the combustionof propane
in a barbecue grill to produce carbon dioxide,water, and heat. Equation 1 givesthe
8)
3H
(C
chemical equation for this reaction; notice that heat appears on the product side in the
equation for an exothermicreaction.
8(g)
3H
C
ExploringEnergyChanges
+
2(g)
50
—
2(g)
3C0
+
20(g)
4H
+
heat
Equation 1
ExploringEnergyChanges Page2
—
4
When a system absorbsheat from the surroundings during a reaction, the temperature of
the surroundings decreasesand the reaction container feelscoldto the touch. This is an
endothermic reaction, where the prefixendo- means “into.”Heat flowsinto the system.A
familiar exampleof an endothermic process is the melting ofice. Solidwater (ice) needs
heat energy to breakthe forces holding the molecules together in the solid state. This physi
cal change is representedbyEquation2; notice that heat appears on the reactant side in the
equation for an endothermic reaction.
20(s)
H
+
heat
Equation 2
20(l)
H
—
Experiment Overview
The purpose of this experiment is to examinethe heat changes in physicaland chemical
processesand to classifythem as exothermic or endothermic.In Part A, three reactions are
carried out in heavy-duty,zipper-lockplasticbags.The bags make it easy to observeand feel
the heat changes that occur. In Part B, the extent of heat transfer in one of these reactions
willbe investigatedby measuring the resulting temperature change. The reactionwill be
carried out in an insulated foam cup and the temperature of the solution will be measured as
a function of time.
Pre-Lab Questions
1. Readthe entire procedureand the recommendedsafetyprecautions.Whathazardsare
associatedwith the use of hydrochloricacidin the lab?Howcan these hazardsbe reduced?
2. Classifyeach of the followingprocessesas a physicalchangeor a chemical change and as
an exothermicor endothermic reaction.
(a) Sugar is dissolvedin water in a test tube and the test tube feelscold.
(b) Gasolineis burned in a car engine.
(c) Wateris convertedto steam accordingto the equation H
20(l)
+
heat
—
20(g).
H
3. Twosolutions,hydrochloricacidand sodium hydroxide,were mixedand the temperature
of the resulting solution was measured as a function of time. The followinggraph was
recorded.Is the reactionbetween hydrochloricacidand sodium hydroxideexothermicor
endothermic?
30
15
0
I
I
I
20
40
60
I
I
80 100 120
Time(sec)
Flinn Chemlopic”Labs
—
Thermochemistry
Page3- ExploringEnergyChanges
Materials
Ammoniumchloride,NH
8—10g
4CI,
Calciumchloride, CaCI
12—14
2,
g
Hydrochloricacid solution, HCI,1 M,40 mL
Sodium bicarbonate,NaHCO
3—5g
3,
Water,distilledor deionized
Temperaturesensor or thermometer
)*
Computer interfacesystem (LabPro”
Computeror calculatorfor data collection*
Datacollectionsoftware(LoggerPro”
)*
*Optional
Balance,centigram (0.0,1g precision).
Beaker,400-mL
Graduatedcylinders,10-and 50-mL
Pen for labeling
Sealabie,zipper-Jockplastic bag, 1
Spatula
Insulated foam (Styrofoam®)cup, 6 oz, 1
Weighingdishes or small beakers, 4
.
I
Safety Precautions
Hydrochloricacidsolution is toxic by ingestion or inhalation and is corrosiveto eyes and
skin. Avoid contact with eyes and skin. Notify the teacher and clean up all spill immediate
ly with largeamounts of water.Ammonium chlorideand calcium chlorideare slightly toxic
by ingestion. Wearchemical splashgoggles and chemical-resistantgloves and apron. Wash
hands thoroughly with soap and water before leaving the laboratory.
Procedure
.
,
I,
Part A. Observing Heat Changes
1. Obtain3 weighingdishes or smallbeakersand labelthem A—C.
2. Weighout the appropriateamount ofsolidinto each weighingdish, accordingto the fol
lowingtable.Recordthe exact mass of each solidin DataTableA.
WeighingDish
Solid
Mass
A
B
C
Ammoniumchloride
2—3g
Calciumchloride
3—4g
Sodiumbicarbonate
1—2g
3. Opena zipper-lockplasticbag and pour the solidfromAinto the bottomofthe bag. Tiltthe
bagso allthe solidfallsinto one corner ofthe bag, then laythe bag flat on the table.
4. Measure10mLof distilledwater in a graduatedcylinderand pour the water into the bag,
trying to pool the water in the upper third ofthe bag.
5. Closethe zipper-lockbag and gently squeeze the bag to mixthe solidand liquid contents.
6. Feelthe sideofthe bag where the reaction is occurring and recordwhether the bag feels
hot or cold to the touch. Observewhat happensas the contents mix.Recordall observa
tions in DataTableA.
7. Washthe contents of the bag downthe drain with excesswater.Rinsethe insideof the
bag with distilledwater and dry it using a paper towel.
8. Repeatsteps 3—6using sample B and 10 mL of distilledwater.
9. Washthe contents of the bag downthe drain with excesswater. Rinsethe inside of the
bag with distilledwater and dry it using a paper towel.
ExploringEnergyChanges
Exploring Energy Changes Page4
-
46
10. Repeatsteps3—6usingsample C and 10rnLof 1Mhydrochloricacid solution.
11. Washthe contents ofthe bag downthe drain with excesswater.Disposeof the bagas
instructed by your teacher.,
Part B. Measuring Temperature vs. Time
Yourteacher will assign you and your group one of the reactionsfrom Part Ato study in
more detail. Recordthe identity of the reaction assigned to you in DataTableB. Usethe fol
lowing table to determine the required amount of solidand liquid for steps 13and 14.
Reaction
Solid (g)
Liquid(mL)
,
A
B
C
,
‘
Ammoniumchloride (6—7g)
Calciumchloride (9-40 )
Soçliumbicarbonate(2—3g)
Distilledwater (30 mL)
Distilledwater (30 mL)
Hydrochloricacid, 1 M (30mL)
12. Set an empty,dry Styrofoamcup into a 400-mLbeaker so that the cup is stable and will
not tip over.
13. Weighout the requiredam9unt of solid in a weighingdish and record the identity and
exact mass of the ‘solidin DataTableB.
0 rnLof the appropriateliquid in agraduatedcylinderand pour the liquid
14. Measure30
into the Styrofoamcup. Recordthe identity and volumeofthe liquid in DataTableB.
15. Plug a temperature sensor into the interface system.
16. Open and format a graph in the data collectionsoftwareso that the y-axisreads tempera
ture in degrees Celsius.Set the minimum and maximumtemperature valuesat 0 and
40 °C,respectively.
17. Thex-axisshouldbe set for time in seconds.Set the minimum and maximumtime values
at 0 and 240 sec, respectively.
18. Set the time intervalto take a temperature reading every10seconds.
19. Placethe temperature probe in the liquid in the Styrofoamcup.
20. Waitone minute (to allowthe temperature sensor to becomeacclimatedto the liquidtem
perature), then press startto begincollectingdata.Immediately add the solidfrom the
weighingdish into the Styrofoam cup and gently mix the contents using a stirring rod.
21. The system willautomaticallyrecord data for the allottedtime (240sec), then stop.
22. Removethe sensor from the Styrofoamcup and rinse it with distilledor deionizedwater.
Washthe contents of the cup downthe drain with excesswater.
23. If possible,obtain a printout of the data table and graph from the computer.
24. CompleteData TableB: Usingthe data from the computer tableand graph of tempera
ture vs. time, recordthe initial temperature of the solution (beforeaddingsolid)and the
maximum or minimum temperature obtained after mixing.
FlinnChemTopicLabs Thermochemistry
—
Page5- ExploringEnergyChanges
•1
.
Name:
Class/Labperiod:
ExploringEnergyChanges.
Data Table A. Observing Heat Changes
Reaction
Solid + Liquid
A
4C1(s)
NH
+
B
2,(s)
CaCI
+
Massof Solid (g)
Observations
.
20(l)
H
.
3(s)
NaHCO
20(l)
H
-
HCI(aq)
,
Data Table B. Measuring Temperature vs. Time
4ssigned Reaction
.
.
Identity of Solid
Massof Solid (g)
Identity of Liquid
.
Volumeof Liquid (mL)
Initial Temperature (°C)
Maximumor Minimum
Temperature (°C)
ExploringEnergyChanges
,
ExploringEnergyChanges Page6
-
48.
Post-Lab Questions
Attach the printout of the data table and graph for Part B to your lab report.
1. Completethe followingResultsTableto indicatewhethereach reaction in Part A repre
sents a physicalor chemicalchange and whether it is exothermicor endothermic.
Reaction
Physicalor ChemicalChange?
Exotherinicor Endothermic?
A
B
c
I
:
:
,
2. Achemicalchange involvesa change in the compositionof matter—the formationof a
new chemicalsubstance (product)with physicaland chemicalproperties differentfrom
those of the reactants. Describethe evidenceused to decideif any of the processesin Part
A were chemicalchanges.
3. Didyou observeany qualitativedifferencesin the amount ofheat generated in the re
c
tions that were characterizedas endothermic in PartA?
4. ConsiderReactionA: Wasenergy releasedor absorbedby the reactants in this system?
When you touched the reactioncontainer (the plasticbag)was energy being rIeased or
absorbedby your hand?
5. Writea balancedequation for each of the processesin Part A.Rememberto includeheat
on the reactant or product side,as appropriate.
6. In Part B,was the temperature that was measured a part of the system or the surround
ings?Howlong did it take for the maximum or minimum temperature to be reached?
7. Describein wordsthe temperature versus time graph that was recorded in Part B. Be as
specificas possible.
8. Completethe followingsentence to summarize the observationsand conclusionsfor the
reactionin Part B:The reaction of ________________________________________
with
is
(exothermic/endothermic)
an
process in which
____________________________________
energywas (absorbed/released)by the system and the temperature of the surroundings
(increased/decreased).
FIinn Chem’I’opic”Labs
—
Thermochemistry
358
Unit Four:Thermodynamics
and Kinetics
4.1.5 CALORICCONTENTOF FOODS
Concepts to Investigate: Calorie (kilocalorie), calorie, food energy, heat.
Materials: Aluminum soft drink can, ring stand, paper clip or twist-tie, test tube clamp, mod
eling clay (or large eraser), straight pin, peanut, marshmallow, match.
Safety: Never eat any food materials used in experiments!
Principles and Procedures: Exothermic chemical reactions are essential for life. As you di
gest food, glucose and other molecules are absorbed into thebloodstream and taken to cells
to be metabolized. ]n the process of metabolism, energy is released and then stored in mole
cules of adenosine tn-phosphate (ATP). ATPmolecules subsequently undergo an exothermic
decomposition, and the energy released is used to power endothermic processes necessary for
growth.
We measure the energy that foods give us in terms of Calories. A Calorie (w
itten
with
since
with
is
kilocalorie
1000
calories
small
(written
cab
a
“c”)
or
a large “C”) actually a
a
ne is the amount of energy required to raise one gram of water one degree ‘Celsius, a kilocalorie or Calorie is the amount of energy required,to raise 1000 grams (1 kg) of waler one
es
degree Celsius. Teenagers generally consume between 1500 and 3000 Cabot
(kibocabories)’
of food energy each day. Those foods which release a large amount of chemIcal energy when
metabolized are frequently referred to as “fattening” foods because people store excess energy as fat. In this activity you will investigate the energy content of some simple foods.
in order to measure the caloric content of foods, it is necessary to construct a simple
calorimeter such as illustrated in Figure H. Place 100 mL (lOOg) of water in an empty alu
minum soft drink can, record the water temperature, and suspend the can from a ring stand
using a twist-tie or paper clip as shown. Weigh a shelled peanut to the nearest tenth of a gram.
Carefully skewer the peanut with a straight pin and embed the other end of the pin in a lump
of clay to make a stand as shown. Light the peanut on fire and lower the can so that it is im
mediately above the flame. The calorimeter will be accurate only if the heat from the burn
ing food is used to heat the water in the can. To reduce heat loss to the surrounding environ
ment, reflect it back to the can by surrounding the flame with a shield made of perforated
aluminum foil. (It is necessary to make holes in the foil to allow oxygen in to sustain the
flame.) When the peanut has been consumed by the flame, determine the increase in temper-
CALCULATION
4.0CaI/g
H
‘
j
f
4 1 Thermochemistry
359
ature of the water in the can and calculatethe amountof energytransferredto the can in terms
of calories by multiplying:
100 g water x
•
_°C
=
calories
Convert this value to Calories (kilocalories)by dividingby 1000:
calories x 1 CaI/1000 calories
=
_____
Cal
Determme the numberof calones per gram by dividingby the‘massof the peanut
—
Cal/_
ut
pe
Cal/g
Repeat the procedure using a miniaturemarshmallow,a cashew,or otherfood of your choice
Which food has the grçatest amount of energyper gram?
Questions
r
(1) Calculate the energy releasedper gram of food.Whichof’thefoodstestedcontainsthe
most energyper gram”
A.
óries
(2)
150-poundperson will burn approximately 750 Ca
per hour playing a full
court game of basketball.How many peanuts would they need to eat to give them
•nough energy to play a one-hour game?
(3) Energy from the combustionof the peanut was used to heat the water in the can. How
is energy from the metabOlismof a peanut used in a human body?
(4) Read the nutritionlabels of three cereals and determine the Caloric content per gram
of each. (A sample calculation is shown in Figure 1.) Which one would you recom
mend fora person on a weight loss diet? Why?
(5) Do you think that this activity will yield a value that is higher or lower than the true
caloric content of the food tested? Explain.
me
N
THETHREE PHASES OF’MATTEk
WARM-UP
Complete the following table to review the properties of liquids and solids. Write “yes”
or “no” in each column of the table.
Solid
Properly
Assumes the shape of its container
.
.
Liquid
Can be formed into a sphere
Flows
.
Crumbles
MATERiALS •& EQUIPMENT
•
•
•
I clear plastic cup
I craft stick
.
corn starch
•
•
water
measuring cups
LAB SAFETY AND PROTOCOL
Do not wash the material you are about to make down the sink. Throw it in the trash.
PROCEDURE
1. Measure 1/4 cup (level) of corn starch into the paper cup.
2. Measure 1/8 cup (level) of water into the paper cup.
3. Stir quickly until the mixture is smooth.
OBSERVATIONS? QUESTIONS
1. Does the material look like a solid or liquid (why
2. Poke your finger into the material. Does it behave like a solid or liquid (why)?
3. Take a spoonful of the material in your hand and roll it into a ball. Then place it
on the desk. How did it behave when you tried to roll it into a ball?
4. Onthedesk?
5. Pick it up again and hold it for a few minutes. Do you observe any changes that
demonstrate the presence of a gas”
CONCLUSION
Is this material a solid, liquid or gas?
‘
Name
Date
Class
Molecvles
ndEner
L
.‘sc3
As you break from a saunter to a full gallop to get to your next class, your
kinetic energy changes. Your energy was increased by the muscles of your
legs propelling you down the hallway. In terms, of energy transfer, the mus
cles of your legs transferred energy obtained from the foods you have eaten.
Can you observe changes in a substance as energy is transferred to it?
PROBLEM
08)ECTIYES
0
.)I
• Observe the temperature changes and
changes of state when a molecular sub
stance is heated and cooled.
• Make and use graphs to analyze tem
perature changes.
• Interpret temperature changes in terms
of the changes in the average kinetic ener
gy of a substance’s molecules.
beaker tongs
timer
test-tube holder
hot plate
20-mm X 150-mm
clamp and ring stand
stearic acid
test tube
400-mL beakers (2)
Celsius thermometers (2)
e
t
S
It
PRECAUTIONS
SAFETY
‘3
e
0.
C.
ii
1. Use the data tables to record your obser
vations. Label one Heating and the other
Cooling.
2. Pour 300 mL of tap water into a 400-mL
beaker and place the beaker bn a hot
plate.
3. Place .a thermometer in the beaker of
water. Thrn on the heat and monitor the
water temperature until it reaches 90°C.
C
Maintain the water temperature at 90
control
of the hot plate
by using the heat
or by adding cold water.
4. Half fill the test tube with stearic acid.
Gently push the bulb of the second ther
mometer down into the substance. After
the temperature of the. thermometer has
adjusted to the stearic acid, record this
temperature in the fi.rst line of the
Heating Data table.
5. Attach the clamp to the test tube and
immerse the tube in the beaker of hot
water as shown. Read and record the
temperature and the physical state or
5S
states of the stearic acid every 3onds until all of the material has melted
and its temperature is about 80°C.
6. Pour 300 mL of cold tap water into the
second 400-mL beaker.
7. Remove the test tube and contents from
the first beaker and immerse it in the
cold water in the second beaker. Read
and record in the Cooling Data table the
temperature and physical state or states
of the stearic acid every 30 seconds until
the material has solidified.
I
MATERIALS
I
CHEMLR
PROCEDURE
How does energy transferred to or from a
molecular substance affect the average kinet
ic energy of its molecules?
)
CREMLRB
RND
MINILAB
WORKSH
_
Use beaker tongs when handling the beaker
of hot water and a test-tube holder when
handling the hot test tube.
)
Chemistry: Concepts and Applications
ChemLab10
67
c-
HEATING:DATA
ElaspedTime (s)
PhysicalState (s, I, or g)
Temperature(c)
0
.
15
30
.
.
45
60
,
,
75
90
,
1 0.5
.
,
‘
135
150
.
‘
165
‘180’
.
‘
.
195
‘
,
.
.
210
,
,
225
240
.255
.
270
285
300
315
330
345
360
375
390
405
,
,
COOLINGDATA
ElaspedTime (s)
0.
15
PhysicalState (s, I, or g)
Temperature (c)
-
•
•
,
30
.
45
,
60
75
.
,
90
,
105
• 120
.
.
135
150
165
,
180
.
‘195
.
210
.
225
240
255
,
.
270
285
.
300
315
330
Part 2:
Construct a graph for your heating & cooling data. Use MS excel
to graph the data. Connect the data points with a straight line
Label the appropriatesegmentswith with solid, solid & liquid, or liquid.
Page1 MeasuringEnergyChanges
-
H
Measuring Energy Changes
Heat of Fusion
Introduction
A physicalchange ofstate or a chemical;reaction may be either exothermic or endothermic.
Anexothermicreactionreleasesheat to its surroundings, while an endothermic reaction•
absorbsheat from its surroundings. What is the source of the heat energy that is releasedin:
an exothermicreaction?What happensto the heat energy that is absorbedin an endother
mic reaction?Canthe amount of heat energy be measured?
Concepts
• Heat vs. temperature
.
Exothermicvs. endothermic
• Heat of fusion
•
Heat of vaporization
• Enthaipychange
Background
Iay
Our every
experiencetells us that energy’inthe form of heat is neededto melt ice or boil
water. Imaginea beakeroiwater at room temperature on a hot plate. Asthe beakeris heated.
the temperature ofthe water will increasesteadilyfrom 25 °C to 100 °C, the boiling point of
water. If the water is heated further, it will begin to boil, but the temperature of the water in
the beakerwill remain constant at 100°C until all of the water has been converted to steam.
Allof this time, of course,while the temperatike has remained constant, heat has been
added to the system.The heat absorbed by.thewater has been used to break apart the forces
(e.g.,hydrogenbonding)betweenwater moleculesin the liquid phase. The amount of heat
that must be absorbedto vaporizea specificquantity of liquid (usuallyone gram or one
mole) is calledthe heat of vaporization. The heat of vaporizationfor water is 2260Joules.per
gram. In a similar manner, heat is also required to melt ice. The amount of heat that must
be absorbedto melt a specificquantity of solid is calledthe heat of fusion.
Vaporizationand fusion are examplesof endothermic physicalchanges.The reversephysical
processes must thereforebe exothermic.Whensteam condensesto water, for example,an
amount of heat energy equal to the heat of vaporizationmust be releasedto the surround
ings. This explainswhysteam burns are more dangerous than hot water burns. Heat is also
releasedwhen water freezesto ice.
The amount of heat transferred in these processes dependson the differencein the energy
stored in each substance.This stored energy is calledthe heat content or enthalpy,and is
representedby the symbolH. The enthalpy change (z
H) for a physicalprocess or a chemical
reaction is definedas the heat change that occurs at constant pressure. This is convenient,
because most of the reactionsthat are carried out in the lab are in flasksor containers that
are open to the atmosphere—thatis, they take place at a constant pressure equal to the
barometric pressure.
MeasuringEnergyChanges
Measuring Energy Changes Page2
-
%57
Equation 1 showsthe equalitybetweenthe change in enthalpy(AI-j)of a system and the
amount of heat transferred,symbolizedby qp, for a reactioncarriedout at constant pressure.
=
Equation 1
qp
The amount of heat (qp)transferred to a substance or objectdependson three factors:the
mass (m) of the object, its specificheat (s), and the resulting temperature change (ST). See
Equation 2.
qp = m x s x AT
Equation 2
The specificheat (s) of a substance reflectsits abilityto absorbheat energy and is defined as
the amount of heat neededto raise the temperature of one gram of a substance by one
degree Celsius.The specificheat ofwater is equal to 4.18 J/g°C. The temperature change
T) is equal to the differencebetweenthe final temperature and the initial temperature
(i.
initiaiL
T
(i.\T = T
nai
—
In most laborato situations, the temperature change is measured not for the system itself
(the reactants and products), but for the surroundings (the solution and the re
ction vessel).
The amount of heat releasedby the system must be equalto the amount of heat absirbed by
in
urs
the surroundings. The sign conventionin Equation 3 revealsthat the heat change oct
the opposite direction.
q(system) = —q(surrouñdings)
Equation 3
For an exothermic reaction, the heat released by the system results in a temperature
increase br the surroundings (ATis positive)and the heat absorbed by the surroundings
will he a fsibive quantity. The heat releasedby the systemmust have the reversesign—it
must he a ne
iiive
quantity. According to this convention, the enthalpy change for an
exotlierinicreaction is alwaysa negativevalue. For an endothermic reaction, in contrast, the
heat absurbed Iw the system results in a temperature decreasefor the surroundings (ATis
negative)and the heat releasedby the surroundings willbe a negativequantity. The heat
absorbed by the system must havethe oppositesign—it must be a positivequantity.
Accordingto this convention, the enthalpy change for an endothermic reaction is alwaysa
positivevalue.
Experiment Overview
The purpose of this experiment is to determine the temperature and heat changes that occur
when ice melts. In Part A a heating curve will be obtained by melting ice in a beakeron a hot
plate and measuring temperature as a function of time. In Part B ice will be placed in a
beakerof warm water and the temperature change that is producedas the ice melts will be
measuredand used to calculate the heat of fusion ofwater (the heat required to melt ice).
FlinnChemlopic”Labs Thermochemistry
—
Page3- MeasuringEnergyChanges
Pre-Lab Questions
A sample of lauricacid—anorganic compound used to make soap—washeated above its,
melting point in a test tube and then cooledin a bath ofwater until it solidified.The
temperature of the lauric acid was measured as a function of time and the followinggraph
was recorded.
________________
-.----—--
.‘,
I
a)
a.
___________
E
20
0
I
2
I
I
I
I
4
6
81012
Time (mm)
e?
1. Whatis the physicalsignificanceof the flat region (plateau)on the cur
2. Usethe graph to estimate the melting point of lauric acid.
3. Is heat being absorbedor releasedby the lauric acid sampleas it solidifies?
Materials
Beakers,400-mL,2
Distilledor deionizedwater
Graduated cylinders,100-and 250-mL
Ice, crushed, about 200 mL volume
Ice cubes, 2
Digitalthermometer
Hot plate
Insulated foam (Styrofoam”’)cup, 6 oz
Stirring rod
Beakertongs or Hothands®
Safety Precautions
Exercisecaution when using the hot plate and handling hot glassware.Remember that a
“hot”hot plate looks exactly the same as a “cold”hot plate. Usebeaker tongs or Hothands®
to handle hot glassware.Wearchemicalsplash goggles wheneverchemicals, heat, or glass
ware are used in the laboratory.
Procedure
Part A. Temperature and Phase Changes
1. Obtain a 400-mLbeakerand fillthe beaker to the 150-mLmarkwith crushed ice.
2. Placethe digitalthermometer in the ice and measure its temperature (it should be
between0 and 2 °C).Recordthe lowesttemperature as the initial temperature (time 0) in
DataTableA.
3. Placethe beaker on the hot plate and adjust the heat setting to the halfwaypoint (for
example,if the heat dial goes from 0—10,adjust the setting to 5). Stir the ice constantly
using a stirring rod.
MeasuringEnergyChanges
MeasuringEnergyChanges Page4
-
4. Measurearid record the temperature of the ice very minute. Notein Data TableAthe
temperature at whichall of the ice has melted.
5. Whenalt of the ice has melted,adjust the hot plate setting to its maximum value!(10 in
the aboveexample)and continue heating and stirring the water.Donot allowth ther
mometer to touch the bottom ofthe beaker.
6. Recordthe temperatureofthewater everyminute.Notein DataThbleA the temperatureat
whichthe waterbeginsto boil.Continueheating until the waterhas boiledforthree minutes.
7. Turnoffthe hot plateand carefullyremovethe beakerusingbeakertongsor Hothand®heat
protectors.
Part B. Energy Needed to Melt Ice,
8. Obtain 100mL of warmwater (about 50 °C) in a graduatedcylinder.Measurethe initial
volumeofwater to the nearest 0.2 mL and record this valuein DataTableB.
9. Place a Styrofoamcup in a 400-mLbeakerfor stabilityand pour the water into the cup.
Measureand record the temperature of the warm waterto the nearest 0.1 °Cin Data
TableB.
10. Obtainseveralice cubes.Shake any excesswater offthe icecubesand carefullyadd the
ice cubesto the warm water bath.
11. Stir the ice/watermixtureuntil the ternperatur is around 0 °C (within 2—4°C).Add
more ice, if necessary,to coolthe water to this temperature.
12. Whenthe temperature has reachedits lowestvalue (again,this should be between0 and
4 °C),record the temperature and immediatelyremoveany unmelted ice from the water
bath using tongs.
13. Carefullypour the water from the Styrofoamcup into a 250-mLgraduated cylinder.
Measureand record the finalvolumeofwater to the nearest 1 mL in DataTableB.
Note: If a 250-rpLgraduatedcylinder is not available,measure the volume ofwater in
two batches using a 100-mLgraduatedcylinder.
FlinnChem’I’opic Labs
—
Thermochemistry
Page5- Measuring Energy Changes
I
Name:
eriod:
Class/Lab F
Measuring Energy Changes
Data Table A. Temperature and Phase Changes
Time
(mm)
Temperature
(°C)
Time
(nun)
Notes
continued
Temperature
(°C)
Notes
continued
‘
0’
•1
(initial)
.
.
.
Data Table B. Energy Needed to Melt Ice
Initial
Volume
(Warm
Water)
MeasuringEnergyChanges
Initial
Temperature
(Warm
Water)
Final Volume
Final Temperature
(Ice Water)
(Ice Water)
MeasuringEnergyChanges Page 6
-
-Lab
Post
Calculationsand Analysis (Usea separatesheetof paperto answerthe /blluivin
j
qiwclit,ns.)
Part A. Temperature and Phase Changes
1. Usingyour data, drawa graph of temperature (y-axis)versus time .(x-axis).
2. Note on the graph the temperature at whichall ofthe icehas meltedand the temperature
at which the water startsto boil.
•
3. Arethereanytemperatureplateaus(flatregionsofthecurve)on the temperatureversus
timegraph?Namethe physicalpropertyofwatercorresponding
to eachtemperature
plateau.
4. Washeat being addedto the systemduring the times that the temperature remainedrela
tivelyconstant?Usethe lawof conservationof energyto describewhat happenedto the
heat energy that was absorbedduring this time.
5. Washeat being addedto the systemduring the times that the temperature was rising?
Usethe lawof conservationof energy to describewhat happenedto the heat energy that
was absorbedduring this time.
Part B. Energy Needed to Melt Ice
6. Usethe appropriatedensityvaluefrom the followingtableof density versus temperature
to calculatethe mass of warm water that was cooledby the ice in Part B.
Temperature
Densityofwater
40 °C
45 °C
50 °C
0.992g/mL
0.990g/mL
0.988g/mL
55 O
0.985g/mL
7. UseEquation 2 in the Background section to calculatethe amount of heat in joules
releasedby the warm water as it cooled.
8. UseEquation3 to calculatethe amount of heat absorbedby the ice as it melted.
9. Determinethe volumeof ice melted in Part B and calculateits mass.Note: The density of
water at 0 °C is 1.00 g/mL. Dividethe result in Question#8 by the mass of ice meltedto
determinethe amount of energy absorbedper gram of ice as it melted.
10. (a) Usethe gram formula weight ofwater to calculatethe heat of fusion ofwater in kilo
joules per mole (Id/mole).
•(b)Writea chemical equation for melting ice and includethe energy term in kJ/mole.
(c) The literature valuefor the heat of fusionof water is 6.02kJ/mole.Calculatethe per
cent error in the experimentallydetermined heat of fusion.
Percent error
I experimental literature I
x 100%
literature
—
=
.
FlinnChemlopic”Labs Thermochemistry
—
Page1 DiscoveringInstantCold Packs
-
Discovering Instant Cold Packs
•
Heat of Soluflàn
Introduction
Instant cold packsare familiarfirst aid devicesused to treat injuries when ice is inavailable.
Mostcommercialcold packs consist of a plasticpackagecontaining a white solid and an
‘inner pouch ofwater.Firmlysqueezing the pack causes the inner pouch to break. The solid
• then dissolvesin the water producinga change in temperature. Can we measure the temper
‘ature change that occurs when the cold pack solid dissolvesin water and determine the heat
change for this process?
Concepts
• Enthalpychange
•
Heat of solution
• Calorimetry
•
Dependentand independent variables
Background
•1
The energyor enthalpychange associatedwith the process of a solute dissolvingin a solvent
is calledthe heat ofsolution ).
In the case of an ionic compounddissolvingin water,
501
H
(i
the overallenergychange is the net result of twoprocesses—theenergyrequired to breakthe
attractiveforces (ionicbonds)betweenthe ions in the crystallattice, and the energy released
when the dissociated(free)ions form ion-dipoleattractiveforceswith the water molecules.
Heats ofsolutionand other enthalpychangesare generallymeasuredin an insulatedvessel
called a calorimeterthat reduces or prevents heat lossto the atmosphereoutsidethe reaction
vessel.The processof a solute dissolvingin water may either releaseheat into the aqueous
solution or absorbheat from the solution, but the amount of heat exchangebetweenthe
calorimeterand the outside surroundings should be minimal.When using a calorimeter,the
reagents being studied are mixed directlyin the calorimeterand the temperature is recorded
both beforeand after the reaction has occurred.The amount of heat changeoccurring in the
calorimeter may be calculatedusing the followingequation: q = m x s x T, where m is
the total mass ofthe solution (soluteplus solvent),s is the specificheat of the solution, and
zT is the observedtemperature change. The specificheat of the solution is generallyassumed
to be the same as that of water,namely,4.18 J/g°C.
Experiment Overview
The purpose ofthis inquiry-basedexperimentis to designand carry out a procedureto deter
mine the enthalpychange that occurs when a “coldpack solid”dissolvesin water.
Pre-Lab Questions
Considerthe followingquestions or guidelines:
1. What information (data)is neededto calculatean enthalpychange for a reaction?
2. Identifythe variablesthat will influencethe experimentaldata.
3. Whatvariablesshould be controlled (keptconstant during the procedure)?
InstantColdPacks
Discovering
Discovering Instant Cold Packs Pacjc 2
f3.
variable iii an experiment
isthe variablethat is changed by the experi
4. The independent
variable respond to (dependson) changes in the ii’idepend
ndciit
menter,’whilethe dep
it.
ent variable.Choosethe ekpendent andindependentvariablesfor this experime
5. Discussthe factors that will ailed: the precisionof the experimentalresults.
Materials
Beaker,400-mL
“Coldpack solid,” 15 g
Distilledor deionizedwater
Graduatedcylinder,100-mL
Insulated foam (Styrofoam
’)
cups, 6 oz
Balance,centigram (0.01 g precision)
Digitalthermometer or temperature sensor
Spatula
Stirring rod
dishe
Iing
Wei
Safety Precautions
The coldpack solid is slightly toxic by ingestion and is a body tissue irritant.Avoidcontact of
all chemicalswith eyes and skin. Wearchemicalsplashgogglesand chemical-resistant
gloves and apron. Washhands thoroughly with soap and waterbeforeleaving the laboratory.
Procedure.
Part A. What Is on Instant Cold Pack?
Completethe followingactivityto becomefamiliarwith the nature and amounts of materials
in a commercialcold pack.
1. Obtaina labelof a commercialcold pack and
write the name of the solidused in the pack.
2. Readthe warning informationon the label
and record any hazardsassociatedwith this
product.
3. Usingthe known charges of ions, write the
formulaof the solid.
4. Calculatethe molar mass of the solid.
5. Determinethe total mass ofthe solid:Tarea
largeweighing dish or ciip on the balance.
Transferthe cold pack solidto the tared
weighingdish. Recordthe mass of the solid
to the nearest 0.01 g.
6. Calculatethe number of molesof solidin
the pack.
7. Measurethe volumeofwater contained in
the inner pouch.
8. Calculatethe mass ofwater in the commer
cialcoldpack (assumethe density ofwater
is 1.0g/mL).
_______________
Nameof solid
Warning
Formulaof solid
_______________
Molarmass
.
_______________
Massof solid
Molesof solid
Volumeof water
_______________
Massof water
Labs Thermochemistry
FlinnChemTopic”
—
Page3- DiscoveringInstantCold Packs
Part B. Measuring the Heat of Solution
that occurs
Designand carry out a procedure to determine the enthalpychange )(t
501
H
when the cold pack soliddissolvesin water.Usea maximumof 5 grams of solid per measure
ment. Write out the procedure in steps and construct a data table that clearly showsthe data
that will be collectedand the measurementsthat will be made.Haveyour teacher check the
procedure and data tablebefore beginning the experiment.
Procedure
1.
2.
3.
4.
5.
6.
Data Table. Enthalpy Change for Dissolving the Cold Pack Solid
DiscoveringInstant ColdPacks
.1
J’
DiscoveringInstant ColdPacks PU
-
N;une:
$
iss/Lab Period:
Cl
_______________
Discovering Instant Cold Packs
Post-Lab Calculations andAnalysis
1. Calculatethe heat energy c/zan.cle injoules when the cold pack soliddissolvedin water in
your experiment.Recall:q = m x s x T, where s (specificheat of water) is equal to
4.18 J/g•°C.
•
2. Calculatethe energy changeinjoules per gram of solid for the cold pack solid dissolving
in water.
• 3. Calculatethe energychange in units of kilo]oules per mole ofsolidfor the cold pack solid
dissolvingin water.Todo this:
(a) Convertthe heat energy change found in Qu
stion
#1 to kilojoules.
(b) Convertthe grams ofsolidused in the experimehttp moles.
(c) Dividethe energychange in kilojoulesby the number of molesof solid to determine
the energy change in units of kJ/mole.If more than one trial was performed,calculate
the averagevalue ofthe heat of solution also.
4. Usingthe result from Question#3c and the informationobtainedin Part A, calculatethe
number of kilojoulesinvolvedwhen the entire cold pack is activated.
5. Circlethe correct choicesin the followingsentence to summarizethe heat change that
occurs when the commercialcold pack is activated:
“When the white solidin the commercialcold pack dissolvesin water, the pack feels
(hot/cold) becausethe temperature of the solution (increases/decreases).Energy is
(absorbed/released)from the surroundings during this reactionand the reaction is
classifiedas (endothermic/exothermic).The sign of iH for the heat of solution is
(positive/negative).”
Flinn ChemTopicLabs Thermochemistry
—
Date
Name
-
Specific Heat
Purpbse:Calculatethe specificheat of glass marbles.
6.
I.
Materials: 6 glassmarbles
laboratorybalance
400-mL beaker
test tube
test tube holder
graduatedcylinder
thermometer
hot plate
calorimeter
Procedure:
1. Preparea hot-waterbath by filling the beakertwo-thirdsfull of water. Heat the
beaker on the hot plate.
2. Using the laboratorybalance,determinethe mass of 6 glass marbles.
3. Placethe marblesinsidethe test tube and place the test tube in the hot-water bath.
4. Measureout 200 mL of roomtemp
water and pour it into a clean, dry
rature
calorimeter.
5. Measure
and
recordthe temperature of the water.
6. To obtainthe temperatureof the glass marbles placethe thermometerinside the
test tube when the water begins to boil. Continueheatingthe marblesin the hotwater bath untilthe temperatureremains constantfor 3 minutes. Recordthe
temperatureof the marbles.
.7. Carefullyremovethe test tube from the hot-waterbath using the test tube holder.
Quickly transferthe marblesto the calorimeterbeing carefulnot to splash any water
out of the calorimeter.
8. Place the thermometerin the calorimeternd recordthe temperatureof the water
and the marblesevery 30 second until the temperatureremains constantfor four
consecutivereadings.
Observations:
Time (mm)
0.0
0.5
1.0
1.5
2.0
.
2.5
Temperature(°C)
________________
________________
_______________
________________
__________
___________
Mass_of_marbles
Initial_Temp_of_marbles
marbles
Final_Temp_of
0
2
Mass_of_H
0
2
Volume_of_H
0
2
Initial_Temp_of_H
0
2
Final_Temp_of_H
1. Calculatethe change in temperaturefor the marblesand for the water.
2. Did the water gain or lose energy? Did the marbles gain or lose energy in the calorimeter?
3. Calculatethe heat transferredby the water, q water.
4. Calculatethe heat transferredby the marbles, q marbles.
5. Calculate the specific heat of the marbles.
Name:
Regents Chemistry: Bayport- Blue Point HS
_________________________________
WlnjDoesn’ttheBalloonHurst?
Balloons are rather fragile things. You know that they must be kept away from sharp
objects. The also need to be kept away from flames. A fire can weaken the balloon and
cause it to burst. However, in this experiment you will find out how you can hold a
balloon directly in a flame without breaking the balloon.
Materiajs
•
•
•
two round balloons, not inflated
several matches
water
Procedure
Inflate one of the balloons and tie it closed.
Place 60 milliliters (¼ cup) of water in the other balloon, and then inflate it and tie it shut.
Light a match and hold it under the first balloon. Allow the flame to touch the balloon.
What happens?
________________________
Light another match. Hold it directly under the water in the second balloon. Allow the
flame to touch the balloon. What happens with this balloon?
_______________________
Why does the balloon with no water break in the flame?
How does the balloon with water in it resist breaking in the flame?
Water is a particularly good absorber of heat. It takes a lot of heat to change the
temperature of water. It takes ten times as much heat to raise the temperature of I gram of
water by 1°C than it does to raise the temperature of I gram of iron by the same amount.
This is why it takes so long to bring a tea kettle of water to the boil. On the other hand,
when water cools, it releases a great deal of heat. This is why areas near oceans or other
large bodies of water do not get as cold in winter as areas at the same latitude further
inland.
Name:
Regents Chemistry: Bayport-Blue Point HS
___________________________________
Directions: Your lab group is to complete stations 1-3., We ‘willdo station 4 as a class.
Station 1: Bell Jar & Vacuum Pump
Materials needed: jar, pump, marshmallow, balloon, tape
Procedure:
ed
1. Blow up the balloon until it has a diameter of about 8cm. Tie the balloon cIo
and tape it to the side of the bell jar.
2. Close the bell jar and turn on the vacuum pump (which removes air from the jar).
Explain what is happening to the balloon.
3. Turn off the pump and allow air back into the bell jar.
4. Place 2 marshmallows inside the bell jar, close the cover and turn on the pump.
Explain what is happening.
5. Stop the pump and switch the hose so that you are allowing air back into the jar
(see your teacher if you need help with this).
Explain what is happening to the marshmallows now.
Observations:
Balloon:
Marshmallows:
What law is this representing?
Station 2: Water Balloon in a Bottle
Materials needed: water balloon (6cm), wide mouth bottle, matches, newspaper
Procedure:
1. Place the filled water balloon on top of the bottle and note if there is a difference
in size.
2. Lift the balloon. Light a SMALL piece of newspaper and drop it into the bottle.
IATELY
IMMED
place the balloon over the mouth of the bottle.
what
is
happening.
Explain
Observations:
Size difference?
How did the balloon go into the bottle?
What law is this representing?
v
Station 3: Do Gases Occu
?
Space
Materials: small beaker, large beaker, water, newspaper
Procedure:
1. Place a small wad of paper into the bottom of the smallbeaker. Be sure that the
paper will not fall out when the beaker is upside down.
2. Fill the larger beaker half-fill with Water.
3. Invert the beaker with the paper and immerse it into the container with water so
that no bubbles escape. You want to make sure that you do NOT tilt the beaker as
you place it into the water.
4. remove the small beaker with the paper and observe the paper.
Is the paper wet? Why or why not?
Was there any air in the small beaker? If so, did it occupy space? How did you come to
this conclusion?
Station 4: 1101Air Balloons & Charles’s Law
Materials: tissue paper, scissors, glue, heavy paper, hair drier
Procedure:
See attached sheets
Page1 -Who’s Counting?
.
1
H
Who’s Counting?
Atoms, Mass, and Moles
Introduction
Wecannot see atoms or molecules.Wecannot tOuchthem or feelthem to determine their
size. But we can count them! How can that be?
Concepts
• Averageatomic mass
•
Molarmass
• Moleconcept
•
Avogadro’snumber
Background
“Whenwe attempt to conceivethe number of particles in an atmo
spherefgasJ,it is somewhat like attempting to conceivethe number of
stars in the universe;we are confoundedwith the thought. But if we
limit the subject, by taking a given volume of any gas, we seem
persuadedthat.. the number of particlesmust be finite.”
.
Thesewords were written by John Dalton in 1808to describehis approachto the theory of
atoms. Atoms may be invisible,immeasurablysmall; infinitesimalin size—but they are
finite. Furthermore, as finite particles, atoms must have real properties.
Allatoms of a given element have the same atorhic nass. Atomsof differentelements have
differentatomic masses.Thesestatements—the cornerstone of Dalton’satomic theory—
provideda starting point for calculating relativeatomic masses of atoms based on the mass
percentage of elementsin compounds. There was still a long way to go, however,before
Dalton’ssuggestionof actuallycounting atoms wouldbe possible.
In 1811,ArnedeoAvogadro,an obscure Italian chemistryprofessor,publisheda stunning
hypothesis—thatequal volumes of gases at equal pressures and temperatures contain equal
numbers of atoms or molecules.This hypothesiswasso stunning that it was completely
ignored for more than 50 years. It was not until manyyears after Avogadro’sdeath that his
hypothesisbecameenshrined as a lawand acquireda numerical valueto go along with it.
Avogadro’snumber is definedas the number of representativeparticles (atomsor molecules)
containedin a specifiedmass, calledthe molar mass, of any pure substance.
The averageatomic mass of atoms and Avogadro’snumber are routinely used todayby
chemists all over the worldto count both atoms and molecules—chemistscount atoms by
weighing them. “Countingbyweighing” makes it possibleto analyzecompoundsfound in
nature and synthesizenew compounds in the lab.
Who’sCounting?
Who’sCounting? Page2
-
/,
7
Experiment Overview
The purpose of this activityis to discover,byanalogy,howchemists count atoms and mole
cules. Knownquantities of rice and beans willbe counted out and their masses measured.
The averagemass of a single rice grain or beanwill then he calculatedand used topredict
the expectedmass of a much larger number of particles.t’inally, the accuracy of this “count
ing byweighing”methodwill also be determined.
Pre-Lab Questions
“Countingbyweighing”is used in other applicationsbesideschemistry.A hardware store,
for example,may sell nails in packagesof 500. Similarly,the officesupplystore probablycar
ries boxesthat contain 100 paper clips. Do you think someone counts out,by hand everynail
or paper clip in these products?
,
,
1. The averagemass of one paper clip is 0.39 g. Whatis the expectedmass of 100 paper
clips?
2. A paper clip manufacturerfindsit more efficientto packagepaper clips in 100-gramlots.
Howmanypaper clips wouldbe containedin a 100-gpackage?
3. In designinga labelfor this packageof paper clips,how manypaper clips would you’rec
ommend the labeladvertise?
Materials
Navybeans, dried, about 75 g
Wholerice, about 10g
Weighingdishes, 4
Balance,‘centigram(0.01 g preciskn)
Markingpen
Safety Precautions
Although the materials used in this activity are considerednonhazardous,followall normal
laboratorysafety guidelines. The food-gradeitems that have been brought into the lab are
consideredlaboratory chemicals and are for lab use only. Do not taste or ingest any materi
als in the chemistry laboratory. Washhands thoroughly with soap and water before leaving
the laboratory.
Procedure
1. ReadParts Aand B in the procedure below.Predictwhether rice or beanswill givemore
accurate results in the “counting-by-weighing”method. Developa hypothesisthat justi
fiesyour prediction. Recordyour hypothesis,includinga brief,one-sentenceexplanation,
in DataTableA.
Part A. Average Mass of Rice and Beans
All mass measurements should be made to the nearest 0.01g. Recordall mass measure
ments and the results of all calculations in Data TableA.
2. Labeltwo weighing dishes“10”and “20”,respectively.
3. Neasure and recOrdthe mass of each weighing dish.
Flinn ChemTopic’Labs
—
MolarRelationships& Stoichiometry
Page3- Who’s Counting?
7d..
4. Count out the appropriatenumber of individualrice grains into each weighingdish.
5. Measureand recordthe combinedmass of each weighingdish and rice sample.
6. Calculatethe averagemass of one rice grain in each sample “10”and “20.”Note: Report
the results to the nearest 0.001 g.
7. Calculatethe “averageof the averages”to determinethe averagemass of a singlegrain of
rice.Note: Reportthe result to the nearest 0.001 g.
8. Discardthe rice grainsas directed by your instructor.
9. Repeatsteps 4—8using navybeans insteadof rice.
Part B. Counting by Weighing
All mass measurements should be made to the nearest 0.01g. Recordall mass measure
ments and the results of all calculationsin Data TableB.
10. Labeltwo weighingdishes“A’and “B”and measure and record the mass of each weigh
ing dish.
11. Usethe averagemass of a singlegrain of rice to calculatethe predicted nass of 100rice
grains.
12. Measureout two separatesamples,eachwith this predictedmass of rice grains, into
weighingdishesAand B, respectively.Note: It may not be possibleto obtain the exact
predicted mass. Get as close as possible—whetheraboveor belowthe predictedvalue:
Rememberto take into account the mass of the weighing dish.
13. Count the actual number of rice grains in each sampleAand B.
14. Discardthe rice grains as directed by your instructor.
15. Repeatsteps 10—14
using navybeans instead of rice.
Who’sCounting?
Who’sCounting? Page4
-
yl
7
Name:
__________________________________
Class/Lab
Period:
_______________________________
Who’s Counting?
Data TableA. Average Mass of Riceand Beans
Will
or beans
rice
give
more
accurate
results
in the
method?
“counting-by-weighing”
Hypothesis:
Sample
Rice
NavyBeans
Rice
NavyBeans
,Mass
Weighingdish
.
Weighingdish and sample
.
Saniple
•
Average
•
.
of one
mass
“particle”
Weighingdish
Weighingdish and sample
“20”
‘
-
Sample
•
,.
Averagemass of one “particle”
.
“Average-of-Averages”
Mass of one particle
Data Table B. Counting by Weighing
Mass
•
Sample
Predicted mass of 100 particles
‘
Weighingdish
Weighingdish and sample
A
•
Sample
Number of particles
Weighingdish
B
Weighingdish and sample
Sample
Number of particles
Flinn Chemlopic” Labs
—
MolarRelationships& Stoichiometry
Page5-Who’s Counting
?
Post-LabQuestions (Useaseparatesheetof paperto answerthe follou
’ing
questions.)
1. In Part A, doesthe averagemass dependon the number of particles in the sample?What
f-averages”mass to calcu-,
are the advantagesand disadvantagesof using the “average
o
late the expectedmass of 100 particles?
2. In Part B, what is the averagenumberof particlescontainedin samples.Aand B for both
rice and beans?
3. Usethe followingequationto calculatethe percenterror in the “counting-by-weighing”
‘methodforboth rice and beans.Note:The number 100in the numerator and denomina
tor is the predictedvaluefor the number of particles.
Percent error
=
I(averagenumber of particles)
100
—
100I
x 100%
4. The percent error describesthe accuracyof the “counting-by-weighing”method. Is this
method more accuratefor rice or beans?Give possibleexplanationfor any differencein
the accuracyofthe method for rice and beans.
5. Referbackto DataTableA: Doesthe accuracyof the results for rice versus beans agreeS
with the hypothesisyou made prior to doing the experiment?Explain.
6. The mass of a mixture containing both rice and navybeans was foundto be 143.85g. The
rice grainswereseparated from the navybeans by putting the mixture through a large
strainer (the smallrice grains fell through the holes, the larger navybeans did not). The
mass of the rice that separatedout was 4.65 g. Usethe results of the abóvèexperiment to’
estimate the number of rice grains and navybeans in the mixture.
7. Expressthe ratio of rice grains and navybeans in this mixture to the nearest whole num
ber (e.g.,1:2,2:1, 1:3,etc.).
8. The mass of a single rice grain is extremelylarge comparedto the mass of a single atom.
(Atypicalhydrogenatom has a mass of 1.66 x 10-24g—too small to evenimagine!)
Chemiststherefore count atoms in large groups, calledmoles,where one mole contains
6.02 x 1023(Avogadro’s
number) of atoms. Let’sdefinea food-moleas containing 602
particles(rice,beans, peas, etc.) Calculatethe mass of one food-moleof rice. This is the
food-molarmass of rice.
9. Calculatethe mass of one food-moleof navybeans.This is the food-molarmass of navy
beans:
10. (Optional)Writea short paragraph explaininghow a chemist would count the number
ofatoms in a piece of aluminum wire. Includethe followingterms in the explanation:
molar mass, mole, Avogadro’snumber.
Who’sCounting?
Page1 AtomicCoatings
-
Atomic Coatings
The Size of an AtOm
Introducton
The size of an atom is too small to imagine.Counting the number of atoms in eve a micro
scopic amount ofmaterial is an impossibletask—itwouldtake a billion years! It turns out,
however,that it is possibleto applya thin surface coating of metal atoms onto another
metal. This is done to change the properties of the underlyingor base metal. In this experi
ment, the thicknessof a zinc metal coatingon galvanizediron will be determined and used
to “count”the number of layers ofatoms in the coating.
S
Concepts
• Atomicsize
•
Density,mass, and volume
Background
Galvanizediron is producedby coating iron with a thin layer of metalliczinc. Thd zinc
coating protects the underlying iron metal against rusting or corrosion. Zinc is more reac
tive than iron and thus reacts with oxygenin the air and with water beforethe iron does. In
er
this way,the zinc coatingprevents oxygenfrom reaching the iron. The grea
reactivityof
zinc continues to protect the iron even after the surfaceof the zinc has been broken or
breached. Galvanizediron has many applications,including rain gutters, heating ducts, nails
and screws,etc.
The amount of zinc depositedon the surfaceof galvanizediron can be determined by react
ing the zinc with hydrochloricacid,accordingto the followingequation.
Zn(s)
+
2HCI(aq)
—‘
2(aq)
ZnCI
+
2(g)
H
Equation 1
The products of the reaction are zinc chloride, which dissolvesin the hydrochloric acid solu
tion, and hydrogengas, which bubblesout of the solution. Bymeasuring the mass of a piece
of galvanizediron beforeand after its reaction with hydrochloricacid, the mass of zinc that
reacted can be calculated.The mass of zinc can be related, in turn, to the number of layers of
zinc atoms in the zinc coating by considering the density of the metal, the surface area of
the galvanizediron, and the size of a zinc atom.
Experiment Overview
The purpose of this experiment is to determine the number of layers ofzinc atoms in the
protectivecoating on a sheet of galvanizediron.
Pre-Lab Questions
1. Readthe Procedureand the Safety Precautions.Whathazards are associatedwith the use
of hydrochloricacid?What safetyprecautionsmust be followedto protect against these
hazards?
2. The reactionrepresentedby Equation 1 must be carried out until all ofthe zinc has
reacted.What visiblesign of reaction can be followedto determinewhen all of the zinc
has reacted?
AtomicCoatings
AtomicCoatings Page2
-
7a
Materials
Balance,centigram (0.01-gprecision)
Beakers,250-and 400-mL,1 each
Forceps
Galvanizediron sheets, about 3 cm x 3 cm square, 2
Hydrochloricacid solution, HCI,6 M, 50 mL
Metricruler, marked in mm
Paper towels
Tapwater
Safety Precautions
•
Procedure
Hydrochloricacidsolution is toxic by ingestion or inhalationand is severely corrosiveto
skin and eyes. Avoid contact with skinand eyes. The piecesof galvanized iron may have
sharp edges that can cut skin. Handle the metal pieces with forceps. Wearchemical splash
goggles and chemical-resistantgloves and apron. Washhands thoroughly with soap and
water beforeleavingthe laboratory.
,
1. Obtain a piece of galvanizediron and measure and recordits mass to the nearest 0.01 g
using the centigram balance.,
2. Measurethe length andwidth of the piece of galvanizediron using a metric ruler. Record
the length’andwidth ofthe iron to the nearest 0.1 cm.
3. Placethe metal piece in a 400-mLbeakerand add enough 6 M hydrochloricacid to cover
the metal (about 25 mL).
4. Letthebeakerstanduntil the rapidbubblingstops.Note:Whenthe reaction is complete,
the piece of galvanizediron will begin to discolorand the solutionwill turn a pale green
color.
5. Whenthe signs of reactionindicatethat all of the zinc has reacted, add about 200 mL of
tap water to the reactionbeaker.This willdilute the hydrochloricacid solution and stop
the reaction.
6. Pour offthe diluted acidinto a waste beaker as directedby your instructor.
7. Removethe metal with a forceps.Holdingthe metal with the forceps,rinse the metal
thoroughly with tap water.
8. Dry the metal on a pieceof paper toweling.When the metal is completelydry,measure its
massagain and recordthe value to the nearest 0.01 g in the data table.
9. If time permits, repeatsteps 1—8with a second piece of galvanizediron.
10. Return the used metal piecesto the instructor for disposal.
FlinnChemlopic’Labs AtomicandElectronStructure
—
Page3—AtomicCoatings
7?,
Name:
Class/LabPeriod:’
Atomic Coatings
Data and ResuJtsTable
Thall
Tha12
,
Massof galvanizediron, initial
.
‘Lengthof galvanizediron
,
.
Widthof galvanizediron
,
Massof galvanizediron, final
.‘
Massofzinc removed
Volumeofzinc coating
.
‘
‘
Volumeofzinc coating per side
Thicknessof coating per side
‘
.
,
‘
Numberof layersofzinc atoms per side
,
Post-Lab Calculations and Analysis
(Show all work.Enter the results of the calculationsin the Data and Results Table.)
1. Subtract the final mass of galvanizediron fromthe initial mass of galvanizediron to cal
culate the mass of the zinc coating on the piece of galvanizediron.
2. The density ofzinc is equal to 7.13 g/cm
Calculatethe volumeof zinc metal correspon
3.
ding to the mass of the zinc coating on the piece of galvanizediron.Hint: Rearrangethe
formulafor densityto solvefor the unknownvolume.
Density =
AtomicCoatings
Mass
Volume
AtomicCoatings Page4
-
3. The zinc’coatingwas present on both sidesof the piece of galvanizediron. Dividethe total
volumeof the zinc coating(Calculation#2) by two to determinethe volumeof the zinc
coating per sideof the galvanizediron.
4. What is the formulafor the volumeof a rectangular solid?Rearrangethis formulato
solvefor the unknown height (thickness)of a rectangular solidif the volume,length, and
width of the solid are known.Checkwith the teacher beforesolving.
*
5. Solvethe aboveequationfor the thicknessof the zinc coating per side ofthe galvanized
iron: Substitute the knownvaluesfor the volume (perside) and the length and width of
the galvanizediron into the formula.
6. The thicknessof a ream (500sheets) of piper is 5.0 cn. Comparethe thicknessof a piece
of paper to the thicknessof the zinc coating.
7. The diameter of a single zinc atom is 2.7 x 1O cm. Dividethe thickness of the zinc
coating per sideof the galvanizediron by the diameterof a single zinc atom to calculate
the number of layersofatoms in the zinc coating.
Numberof layersof zinc atoms
Thicknessof zinc coating per side (cm
)
Diameterof zinc atom (cm)
.
Flinn ChemTopic’Labs AtomicandElectronStructure
—
Name
Date
Class
it
Line
Emission
Spectro
ofElements
Emission spectra of elements are the result of electron transitions within
atoms and provide information about the arran
ements
of electrons in the
atoms. Observe and compare a spectrum from white light with the emission
spectra of several elements.
CHEMLAD
AND
MINILAII
SHEETS
WUR
MINILAD
2.2
PROCEOUNE
1. Obtain a diffraction grating from the
teacher. Hold it only by the cardboard
edge and avoid touching the transparent
material that encloses the diffraction
,grating.
2. Observe the emitted light from an incan
descent bulb through the grating as you
hold it close to your eye. Record ‘your
observations.
3. Next, observe the light produced by the
spectrum tube containing hydrogen gas’
and record your observations. It may be
necessary for you tomove to within a
few feet of tlie spectrum tube in order to
effectively observe the emission spec
trum. CAUTION: The spectrum tube
operates at a high voltage. Under no cir
cumstances should you touch the spec
trum tube or any part of the transformer.
4. Repeat procedure 3 with other spectrum
tubes as your teacher designates.
=
C,
U
0
U
2. If each hydrogen atom contains only one
electron, how are several emission spec
tral lines possible?
3. How do you interpet the fact that other
elements emit many more spectral lines
than hydrogen atoms?
ANALYSIS
1. How do you explain why only certain col
ors appear in the emission spectra of the
elements?
C,
0
C
0
Chemistry: Concepts and Applications
MiniLab 2.2
19
Blank Visible Spectra
This page has been included for your convenience. You may photocopy or duplicate this page for use in recording
observations and answering questions in this experiment.
I
IlillIllIll
i
ii
i
i
I
I
Ii
I
I
I
600
650
yellow orange
I
uuI
I
I
IIl
600
i
i
III
i
I
I
I
I
I
I
I
I
600
650
yellowccange
I
I
I
I
I
I
I
I
I
I
I
II
I
I
I
III
I
I.
I
i,
700 750,
red
I’J
II
1I
I
I
liii
•.
II
II
11111111
650
1•III
I
II
II
700 750
red
yelloworange
II
II
.111
II
III1I
III
I
II
111111
I
I
i
i
i
I
I
,
I
I
I
I
I
I
I
I
I
I
I
i
i
i
i
700 750
red
I
I
I
I
I
I
I
I
I
Ii
i
iii
GOD 650
yellow orange
II
700 750
red
Page1 FlameTests
—
Flame Tests
Atomic Emissionand ElectronEnergy Levels
Introduction
nent
Just as a fingerprint is unique to each person, the color of light emitted by an ele
heated in a flameis also unique to each element. In this experiment, the characteristic color
of light emitted bycalcium, copper,lithium, potassium,sodium, and strontium ionswill be
observed.
Concepts
• Atomicemission
•
Wavelengthand energy of light
• Excitedvs.ground states
•
Flametests
Background
When a substanceis heated in a flame, the
atoms absorb energy from the flame.This
absorbed energyallowsthe electrons to be
promoted to excitedenergy levels.From
these excitedenergy levels,there is a natu
ral tendency for the electrons to make a
transition or drop backdown to the ground
state. When an electronmakes a transition
from a higher energy levelto a lower energy
level,a particle of light called a photon is
emitted (see Figure 1).Both the absorption
and emissionof energyare quantized—only
certain energy levelsare allowed.
$
Excited Energy State
—
Emissionof Light (Photon)
•
—
____________
Ground State
Figure 1. Absorptionand
emissionof energy.
Anelectron may drop all the way backdownto the ground state in a singlestep, emitting a
photon in the process.Alternatively,an electron may drop backdown to the ground state in a
series of smaller steps,emitting a photon with each step. In either case, the energy of each
emitted photon is equalto the differencein energy betweenthe excitedstate and the state to
which the electronrelaxes.The energy of the emitted photon determinesthe color of light
observedin the flame.The flamecolor may be describedin terms of its wavelength,and
Equation I may be used to calculate the energy ofthe emitted photon.
AE
=
—s-A
Equation 1
E is the differencein energy betweenthe two energy levelsin joules (J),
h is Planck’sconstant (h = 6.626 x 10 Jsec),
c is the speed of light (c 2.998 x 108mlsec), and
A(lambda)is the wavelength of light in meters. The wavelengthsofvisible light are given in
units of nanometers (1 m
1 x iO nm). See Table1 on the followingpage.
FlameTests
Flame Tests Page2
—
The colorof light observedwhen a substanceis heatedin a flamevaries from one substance
t
,
the
to another. Becauseeach elementhas a differer
spacingof electron energy 1evel
in
possibleelectron transitions for,agiven substance are unique.Therefore,the diff
rence
ofthe emitted photon, and the cdrresponding
y
energy betweenenergy levels,the exactener
and
color
substance.As
to
wavelength
a result, the colors observedwhen a
are unique eacI
substanceis heated in a flame may be used as a means ofidentification.
The Visible Portion of the Electromagnetic Spectrum
Visiblelight is a form of electromagneticradiation. Other familiarforms of electromagnetic
radiation include ‘j-rays,X-rays,ultravidlet (UV)radiation, infrared (IR).radiation, microwave
radiation, and radio waves.Together,all forms of electromagneticradiation make up the
electromagneticspectrum. The visibleportion of the electromagneticspectrum is the only
ion
other formsof electromagnetic radia
portion that can be detected by the human ey,e—’-—all
are invisible.
Visiblespectrum
Violet
400
Blue
Green
Yellow
500
Orange
600
Red
700
Wavelength,?. (nm)
Figure 2. The visiblespectrum.
The visiblespectrum spans the wavelengthregion fromabout 400 to 700 nm (Figure2).
Light of 400 nm is seen as violet and light of 760 nm is seen as red. Accordingto Equation 1,
wavelengthis inverselyproportionalto energy. Therefore,violet light is higher energy light
than red light. Asthe color of light changes, so dogs the amount of energy it possesses.
Table1 lists the wavelengthsassociatedwith each of the colorsin the visible spectrum. The
representativewavelengthsmay be used as a benchmarkfor each color.For example,instead
of referring to green as light in the wavelength range 500—560
nm, we may approximatethe
of
An
infinite
of
shades
of each color may be
520
number
wavelength a green light as
nm.
observed.
Table 1.
Representative.
Wavelength,nm
WavelengthRegion,
nm
Color
410
400—425
Violet
470
425—480
Blue
490
480—500
Blue-green
520
500—560
Green
565
560—580
580
580—585
Yellow-green
‘Yellow
600.
585—650
Orange
650
650—700
Red
Flinn Chemlopic”Labs
—
Atomic and Electron Structure
Page3—Flame Tests
Experiment Overview
The purpose of this experiment is to observethe characteristicflametest cOlorsof different
metal compoundsand to use this informationto identifyan unknown metal ion.
Pre-Lab Questions
1. Fill in the blanks:When an atom absorbsenergy,the electrons move from their
state to an
state. When an atom emits energy,the elec
from
state and give off
trons move
state to their
a(n)
______________
______________
_______________
_______________
2. Is aflametest a qualitativeor quantitát
ve
test for the identity of an unknown?Explain.
Materials
Calcium chloride, 2O
0.5 g
CaCl
H,
0.5 g
Copper(II)chloride,2O
CuCl
H
,
Lithium chloride,LiCI,0.5 g
Potassiumchloride,KCI,0.5 g
Sodium áhloride,NaCI,0.5 g
Strontium chloride, 2O
0.5 g
6H,
SrCl
Unknownmetal chloride,0.5 g
Safety Precautions
Beakers,250-mL,2
Laboratoryburner
Scoopor spatula
Weighingdishes, 7
Woodensplints soakedin water, 7
Water,distilled or deionized
•0•
Copper(II)chlorideis highly toxic by ingestion;,avoidcontactwith eyes, skin, and mucous
membranes.Lithium chlorideis moder
toxic by ingestionand is a body tissue irritant.
itely
the
wooden
Fullyextinguish
splints by immersing them in a beakerof waterbeforediscarding
them in the trash to avoidtrash can tires. Wearchemicalsplashgoggles,chemical-resistant
gloves,and a chemical-resistantapron. Washhands thoroughly with soap and water before
leavingthe laboratory.
Procedure
1. Fill a 250-mLbeakerabout half-fullwith distilledor deionizedwater.Obtainsevenwooden
splints that havebeen soakedin distilledor deionizedwater.Placethem in this beakerof
waterto continue soakingat your labstation.
2. Fill a second250-mLbeakerabout half-fullwith tap water.Labelthis beaker“rinsewater”.
3. Labelsevenweighingdishes Ca,Cu,Li,Na,K,Sr,and unknown.Placeonescoopful(about
0.5 g) ofeachsolidmetalchlorideinto the correspondingweighingdish.
4. Light the laboratoryburner.
5. Dip the soakedend of one of the woodensplints in one ofthe metal chlorides,then place
it in the flame.Observethe color ofthe flame.Allowthe splint to burn until the color
fades.Trynot to allowany of the solidto fallinto the barrel ofthe laboratoryburner. If
necessary,repeat the test with the same splint and additionalsalt.
FlameTests
Flame Tests—Page4
L
6
6. Immerse the woodensplint in the “rinsewater”to extinguishit, then discardit in the
trash.
7. Recordyour observationsfor the flamecolor producedby the metal chloride in the Data
Table.
8. RepeatSteps 5—7for the other fivemetal chlorides.Recordyour observationsfor the
flamecolor producedby each metal ion in the DataTable.
9. Perform a flametest on an unknownmetal chlorideand record its characteristic color(s)
an’dthe probableidentity of the unknown in the DataTable.
Flinn ChemTopic
Labs
”
—
AtomicandElectronStructure
— Flame Tests
Page5
Name:
___________
iod:
Class/LabPer
I
Flame Tests
Data Table
MetalIon
Colorof Flame
Calcium
Cop,per
Lithium
Sodium
Potassium
Strontium
Unknown
ResultsTable
Metal/FlameColor
FlameTests
A.(nm)
A(m)
tIE (J)
FlameTests Page6
—
Post-Lab Questions
rd
the approximatewavelengtho light
1. UseTable1 in the Background section to rec
emitted for each knownmetal ion in the ResultsTable.
2. Converteachwavelengthin the ResultsTablefrom nanometersto meters. Show one
sample calculationin the spacebelowand record all valuesin the ResultsTablet
3. The characteristiccolorof the sodiumflame‘isdue to tWoclosely-spacedenergy transi
tion to calculatethe averageenergy(4E)
tions. UseEquation 1 from the Background se
correspondingtothe observedflamecolor for each metal.Show one samplecalculation
in the space belowand recordall valuesin joules in the ResultsTable.
4. Whatevidenceis there from your results that the characteristiccolor observedfor each
compound is due to the metal ion in each casç? Describean additionaltest that couldbe
done to confirmthat the color is due to the metal ion.
5. A glass rod was heated in a burner flameand gave offa bright yellowflame.What metal
ion predominatesin the glass rod?
6. The alkalimetals cesium (Cs)and rubidium (Rb)were discovered‘basedon their charac
teristic flamecolors.Cesiumis named after the sky and rubidium after the gem color.
Whatcolorsof light do you think these metals give offwhen heated in a flame?
Flinn ChemTopic’Labs AtomicandElectronStructure
—
Isotopes of Pennies
LabSheet
•I
Youwilldo a lab that willdealwith isotopes,mass number,and atomicmass.Before
you begin your workIn the lab, try to explaintheseterms in yourown words.Afteryou
havefinished the lab, you willhavea chanceto reviseyourexplanationsbasedon what
you haveiearfledin the activity.
Isotope
Is
Massnumber
Atomicmass
Nkir.s
Scáen L
mae
M 1s
L
Sheet- lsc*cpeeol P
vies
iotmaJ
ScienceNe1LIr*sSluderdSheenmaybe,uçxoóicedlot edt
.
pwpce
Isotopesof Pennies
LabSheet
In 1982, the UnitedStates governmentchangedthe wa It mintedpennies. Before1982,
pennies were madeof 95%copper and 5%tin. Now they are madeof zinc Coatedwith
copper.Becausetheyweighdifferentamounts(havedifferentmasses),we can call
them Isotopes of pennies.
inthisexercise?
• Whatdothetwokindsof penniesrepresent
•
Howdothe penniesdiffer? Howdo isotopes
differ?
haveincommon?
• Whatdothe pennieshaveincommon?Whatdo isotopes
Scien
Afl n1s
NetLiiks Lab Sheet - Isciopes ot Pemies
iuserved. Scien NetLinks Student Sheet
may be ,prodi.ced
tat educatiemi pwpoees.
Isotopes of Pennies
LabSheet
PartA
1. Obtaina sampleoftenpennies.
2. Weighseveralpre-1982(old)penniesandrecordtheiraveragemass
3. Weighseveralpost-i982 (new)penniesandrecordtheiraveragemass.
_______
howmuchthreeoldpenniesplussevennewpenniesshouldweigh.
4. Calculate
410 findtheweighted
5. Divideyou,ranswerfornumber
averagemassofthe penniesinthe
samplecontainingthreeold plussevennewpennies.
6. Nowweighyoursampleofthreeoldandsevennewpennies.Recordthe mass.
_______
7. ‘Divide
youranswerfornumbersixbytentofindthe averagemassof a pennyinyour
sample.
’
g
fiveto yourarwwerfornumberseven.Isthe weighted
• Compareyouranswerfornumber
averagemassclosertothemassofan old pennyora newpenny?Why?
•
Howisthisweighted
averagemassrelatedtoatomicmass?
Science Ne(Li*s Lab Sheer - Is
1opes of Pem.es
SWtlied Sheets may be rexod
s
NeILM
xed
An ñ
veserved. ScBi
’ls
Ici ethicatior
t
pwposes.
Isotopesof Pennies
LabSheet
Part B
sixoldpenniesandfournewpennies.
1. Obtaina samplecontaining
I
*
2. Usingthe massof an old pennyand a new pennyfrom partA above,calculate
a weighted
ofallten
forthis
Youneedtofindthe
of
mass
penniesand
sample pennies.
averagemass
‘dMdebytentofindtheweighted
averagemass.
_______
3. Nowweighyoursampleof pennies.Recordthemass
_______
4. Dividethemassofyoursampleoftenpenniesbytentofind theactualaveragemassof a
pennyinthis sample.
Compareyouranswerfromnumbertwoto youranswerfornumberfour. Is.theweighted
averagemassclosertothemassofan old pennyora newpenny?Why?
e
Scierx
LiiI
sN
.
reserv
Afl ñ
1s
Lab Sheef - Is*opes of Perwies
S*udenfSheet may be re
Scierv NetLief
s
ed
xoths
lot educabonal purposes.
Isotopesof Pennies
LabSheet
PartC:The MysterySample
frompartBto yourteacher.Geta canisterof pennies.
1. Returnyoursampleoftenpennies
Don’t open It. Recordits identifyingnumberorletter_____
2. Recordthe massoftheemptyfilmcanister,
whichisonthe labeloftypecanister.
_______
3. Weighthe sealedfilmcanister
tenmixedpennies.
containing
4. Returnthecanister
to yourteacher.
Calculations:
• Calculatethe numberofoldandnewpenniesin yourcanister.
• Sincethetotalnumberof penniesisten,wecan saythattherearex oldpenniesp
Ius 10—
Thetotal
ofthe
with
áss
minus
therr
ofthe
x newpennies.
mass
pennies(canister pennies
isusefulhere.
canister)
• X timestheaveragemassofan old pennyplus(10 x) timesthe averagemassof a new
Set upanequationandsolvefor
pennyequalsthetotalmassofthepenniesinthecanister.
Subtract
thatnumber
x. Thenyouwillknowhowmanyoldpenniesare inyourcanister.
fromtento findthenumberof newpenniesthat are inyourcanister.
—,
• Showyourmathhere:
• Howmanyoldpenniesdoyouhave?
• Howmanynewpenniesdoyou have?
• Whatpercentageofoldandnewpenniesdo youhave?
______
______
cience NelLh*s Lab Sheel - l1opes oI Pemies
All rr
reserved. Scierv NeUJrlcs Student Sh
1s
J
may be reprodtxed for edurabor
purposes.
Page1 Bean Bag Isotopes
-
Bea,nBag Isotopes
RelativeAbundanceand Atomic Mass
Introduction
At the beginningofthe 1gtb century, John Daltonproposeda new atomic theory—allatoms of
the same element are identicalto one another and equal in mass. It wasa simpleyet revolu
tionary theory. It was alsonot quite right. The discoveryof radioactivityat the beginning of
th century madeit possibleto study the açtüal structure and mass of atoms. Gradually,
the 20
evidencebeganto buildthat atoms of the saine element couldhavedifferentmasses.These
atoms were calledisotopes.Howare ,isotope!sdistinguishedfrom one another?What i the
relationshipbetweenthe atomic mass of an elementand the mass of each isotope?
Concepts
• Isotope
•
Percent abundance
• MassnUmber
•
Atomicmass
Background
Twolinesofevidencein the early2Øth centurysuggestedthe possibleexistenceof isotopes.The
first came fromwork byJ. J. Thomsonwith ‘positiverays,”positivelychargedstreams of atoms
generated in gas dischargetubes.Whenthese positiverays were bent or deflectedin the pres
ence of electricand magneticfieldsand then allowedto strike a photographicfilm,they left
curved“spots”on the film at an angle that dependedon the mass and charge ofthe atoms. In
1912,Thomsonfoundthat when the gas in the tub was neon, he obtainedtwo curvesor
spots. The majorspotcorrespondedto neon atomswith a mass ofabout 20 atomic mass units
(amu).There wasalso a much fainter spot, however,correspondingto atomswith a mass of
about 22 amu. Althoughthese results were consistentwith the existenceoftwo typesof neon
atoms having differentmasses,theywerenot preciseor accurateenoughto be conclusive.
The second line of evidencesuggesting the existenceof isotopescame from studies of
radioactivity.Oneofthe products obtainedfrom the radioactivedecayof uranium is lead.
When the atomic mass of lead deposits in radioactiveuranium minerals wasanalyzed,it was
found to be significantlydifferentfrom the atomic mass of lead in lead ore. The actual com
position of the leadatoms seemed to be different,dependingon their origin.
In 1913, FrederickSoddy,professor of chemistryat the Universityof Glasgow,coinedthe
term isotope to defineatoms of the same element that have the same chemical properties
but differentatomic masses. The word isotopewas derivedfrom Greekwords meaning “same
place”to denote the fact that isotopes occupy the same place in the periodic table (they are
the same element) eventhough they havedifferent masses. Soddyreceivedthe NobelPrize
in Chemistry in 1921for his investigationsinto the nature and origin of isotopes.
Conclusiveproofforthe existenceof isotopescame fromthe workofFrancisW.Astonat
CambridgeUniversity.Astonbuilt a modified,more accurateversionofthe “positiveray”appa
ratus that Thomsonhad earlierused to studyions. In 1919,Astonobtainedprecisemeasure
ments ofthe majorand minor isotopesof neon, correspondingto mass numbersof20 and 22,
respectively.Astonreceivedthe NobelPrizein Chemistryin 1922forhis discoveryof isotopes.
BeanBagIsotopes
sean ScigIsotopes Page2
-
575
The moderndefinitionof isotopesis basedon knowledgeof the subatomic particlestructure
of atoms. Isotopeshavethe same number of protons but differentnumbers of neutrons. Since
the identity ,f an element dependsonlyon the number of protons (the atomic number),
isotopeshavethe same chemical properties.Isotopesare thus chemicallyindistinguishable
from one another—theyundergo the same reactions, formthe same compounds,etc.
Isotopesare distinguishedfrom one another based on their mass number, definedas the sum
of the number of protons and neutrons in the nucleus of the atom.
Chlorine, for example,occurs naturally in the form of two isotopes,chlorine-35and
chlorine-37, where35 and 37 represent the mass numbers of the isotopes. Each isotope of
chlorine has a characteristicpercent abundance in nature. Thus, whether it is analyzedfrom
underground salt depositsor from seawater,the element chlorine alwayscontains 75.8%
chlorine-35atoms and 24.2%chlorine-37atoms. The atomic mass of an element represents
the weighted averageof the masses of the isotopes in a naturally occurring sampleof the
element. Equation 1 showsthe atomic mass calculationfor the element chlorine. The mass
of each isotope is equal to its mass number,to one decimal placeprecision.
Atomicmass (chlorine) = (0.758)(35.0amu)
+
(0.242)(37.0amu)
=
35.5 amu
Equation 1
Experiment Overview
The purpose of this experiment is to investigatethe massproperties and relativeabundance
of isotopesfor the “bean bag” element (symbol,Bg) and to calculatethe atomic mass of this
element.
Pre-Lab Questions
1. Neutrons were discoveredin 1932,more than 10 years after the existenceof isotopes
was confirmed.What property of electronsand protons ledto their discovery?Suggesta
possiblereasonwhyneutrons were the last of the three classicsubatomic particlesto be
discovered.
2. Siliconoccurs in nature in the form of three isotopes,Si-28,Si-29,and Si-30.Determine
the number of protons, neutrons, and electrons in each isotopeof silicon.
3. “Theatomic mass of chlorine represents the mass of the most common naturally occur
ring isotope of chlorine.”Decidewhether this statement is true or falseand explainwhy.
Materials
Balance,centigram (0.01-gprecision)
“Beanbag”element, symbolBg,approximately50 g
Weighingdishes or small cups, 4
Labelingpen or marker
Safety Precautions
Although the materials used in this activity are considerednonhazardous,please observeall
normal laboratorysafety guidelines. The food-gradeitems that have been brought into the
lab are consideredlaboratory chemicals and are for lab use only. Do not taste or ingest any
materials in the chemistry laboratory. Washhands thoroughly with soap and water before
leaving the laboratory.
Flinn ChemTopic’”Labs Atomicand ElectronStructure
—
Page 3- Bean Bag Isotopes
Procedure
1. Sort the atoms in the “beanbag” element sample(Bg)into three isotoperoups (1, 2,
and 3) accordingto the type ofbean. (Assumethat each type ofbean represents a different
isotopeand that eachbean representsa separate atom.) Placeeach isotope group into a
separateweighingdish or small cup.
I
2. Countand recordthe number of Bgatoms in each isotope group.
3..Measurethe total mass of Bgatomsbelongingto each isotopegroup. Recordeach mass to
the nearest0.01g in the datatable.Note: Zero,(tare) the balancewith an emptyweighing
dish on the balancepan, then addallofthe Bgatoms of one typeto the weighingdish and
recordthe mass.Dothis foreach isotopegroup.
Data Table
ResultsTable
“BeanBag”Isotope
(Bg)
.
AverageMass
PercentAbundance
I
,
2
3
Post-Lab Questions (Usea separatesheet of paper to answer the followingquestions.)
1. Determinethe averagemass of each Bg isotopeto three significantfigures. Enter the
results in the ResultsTable.
2. What is the total number of “bean bag”(Bg)atoms in the originalsample?Calculatethe
percent abundanceof each isotope:Dividethe number of atoms of each isotopeby the
total number ofatoms and multiply the result by 100.Enter the results to one decimal
place in the ResultsTable.
BeanBagIsotopes
Bean Bag Isotopes Page4
-
•
3. The atomic mass ofthe “beanbag” element (Bg)representsaweighted average of the
mass of each isotopeand its relativeabundance.Usethe followingequation to calculate
the atomic mass of Bg.Note: Dividethe percent abundanceof each isotope bi 100to
obtain its relativeabundance.Relativeabundance = Percentabundance
100
Atomiciriass = (rel. abundanceisotopex mass
i) + (rel. abundancejsotope
x
2
0,
+
2)
(ret.
3)
x
abundancesotope
massisotope
3
4.
How many Bgatoms in the originalsample wouldbe expectedto havethe same mass as
the calculatedatomic mass of the element? Explain.
5. The isotopesof magnesium(and their percent abundance)are Mg-24(79.0%);Mg-25
(10.0%),and Mg-26(11.0%).Calculatetheatomic mass of magnesium.Note: Toone
decimal place, the mass of each isotope is equal to the mass number. Thus, the mass of an
atom of Mg-24s 24.0 amu.
6. Copper(atomicmass63.5)occurs in nature in the form of two isotopes,Cu-63and
Cu-65.Usethis informationto calculatethe percent abundanceof each copper isotope.
7. Explainwhy the atomic mass of copper is not exactlyequalto 64, midwaybetweenthe
mass numbers of copper-63ançlcopper-65.
8. Radioactiveisotopes(radioisotopes)are widelyused in medicine.Becauseisotopeshave
identicalchemicalproperties,the reaction and distributionof radioisotopesin the body is
e-131,
similarto that of their natural isotopes.Iodii
for example,is an artificialradioiso
tope thatis used to diagnosethyroid disorders.Whenadministered to a patient, the
radioisotopeis taken up by the thyroid gland, wliereit is incorporatedinto the thyroid
hormones,just as iodinein the diet wouldbe. Basedon wherethe followingelements are
nd in the body,match each radioisotopewith its medical use.
likelyto be foi
9.
Sodium-24
a. studies of bone formation
Phosphorus-32
b. red bloodcellstudies
Calcium-47’
c. tracing bloodcirculation
Iron-55
d. genetics (DNA)research
(Optional)Astoncalledthe instrument he designedto measure the masses of atoms the
mass spectrograph.Modernversionsof Aston’s massspectrograph,called massspectrom
eters, are workhorseinstruments in chemical analysis,including forensics.Lookup mass
spectrometry on the Internet and brieflydescribe twoapplicationsof this technologyin
forensicanalysis.
Flinn Chemlopic” Labs
—
AtomicandElectronStructure
Page1 It’s n the Cards
—
It’s in.the Cards
Discoveringthe PeriOdic”Law,
Introduction
S
evements
DmitriMendeleev’sdiscoveryof the PeriodicLawranks ‘asone of the greatest ac1i
in the history of science.It has survivedthe test of time and stands to this dayas the single
most important tool to understand the chemistry of the elements.Aswe try to understand
‘the essence ofthis great discovery,it is worthwhileto go back in time and look at how it was
‘achieved.Whatdid Mendeleevknowand when did he knowit?
Concepts
Periodiclaw
•
Periodictable
• Atomicmass
•
Ionization energy
Background
Dmitri Nendeleev,a professorof chemistry at the Universityof St.
In the years 1868—1870,
of
Petersburg in Russia,was intent on writing a new textbookof chemistry, called”Principles
than
Nore
60
individualelements
a
nd
facts
had
been
Chemistry.
were ‘known, a great many
discoveredabout their properties and compounds.Mendeleevknew the atomic masses of the
the formulas of their compounds”
elements, their densities, boiling points, melting
points,
with hydrogen, oxygen,and chlorine, and many other isolatedfacts.What was missing was a
wayto organize these facts, a way to understand how individualfacts related to each other—
in short, a way to classifythe elements. The followingquote is translated from Mendeleev’s
first publishedreport of the periodic law:
“1wished to establish some sort of system of elements in which their distribution
is not guided by chance. but by some sort of definite and exact principle.”
.
.
Mendeleevdecidedto arrange the elements systematicallyaccording to their atomic mass.
Hewrote out the exact atomic masses (as they were knownat the time) in the margin of a
list of the elements, then wrote out separate cards for each of the elements, with their atom
ic mass and important chemical and physicalproperties. Using these cards, Mendeleev
played“chemicalsolitaire” for several hours, finallycopyingto a sheet of paper the arrange
ment he had worked out with the cards. With slight modification, this became Mendeleev’s
firstPeriodicTableof the Elements.
Experiment Overview
The purpose of this activity is to re-create Nendeleev’sdiscoveryof the classificationof the
elements and the periodic law using a special deck of element cards. The real properties of
the elements, but not their names or symbols,are written on these cards.Asthe cards are
arranged and rearranged based on logical trends in some of these properties, the nature of
the periodic lawshould reveal itself.
It’sin the Cards
• It’sin the Cards Page2
-
Pre-LabQuestions
•
I
.•
The element cards list all of the followingproperties for each element. Useyour textbook to
write a clear definitionof each property.Givethe typicalunits for each numerical property,if
appropriate.
• ionization energy
•
Atomictnass
• Atomicradius
•
Density
• Meltingpoint
•
Electronegativity
Materials
Spèciàldeck of 31 element cards
Procedure
1. Form a research group with two or three other students. Obtain a deck of element cards
and spread the cards out on the lab table.
2. Each cd
lists the properties of a single element (X),as shown below:
Ionizationenergy
Atomk radius
Atomicmass
Formulaof its oxide XbOC
Formula’ofits chlorideXCIa
Meltingpoint
Density*
!ormula of Its hydrideXHd
Electronegativity
for solidsand liquids,g/L for gases.
3
*Densjtyvalues are in units of g/cm
3. Workingin a collaborativemanner, discuss the possibilitiesfor arrangement of the ele
ment cards with all members of the group, and lookfor a logicalarrangement of the
cards.Considerthe followingquestions:
• What are the similaritiesand differencesamong the elements?
• Arethere any numerical or logical trends in their properties?
4. It is NOTwithin the rules of this game of chemical solitaire to look up information in a
textbookor to use a modern periodic table as a guide!
Mendeleev’sgreatest insight in creating the periodic table was in recognizing there were
some gaps when the elements were arranged in logical order.He had the courage not onl(,
to leaveblanks in his table for the missing elements, but also to predict their properties.
Flinn ChemTopic Labs
—
The PeriodicTable
I
Poge3-It’sintheCarcls
••
.
•
re.the
5. One ofthe element cardsis alsOmissingin your deckof cards. Decidewh
missing
ofthe
elementsand
element belongs in the arrangement
rds if necessary
rearrange the ca
to accommodatethe missing element.
6. On the Data Sheet, fill in the Tableof the Elements to illustrate a logicalarrangement of
the element cards.Todo this, write down onlythe atomic mass of each element, as
shown on its card. Leavea blank space for the missing element.Note: The table is 8 x 8
and contains 32 more squares than are needed—plentyof room to arrange the elements
many differentways.
7. Predict the properties of the missingelementby averagingthe properties Ofits nearest
neighbors (in any direction).On the Da1
complete the card for the missing;
aSheet;
element by entering its predictedpropertiesalongsidethe name of each property.
It’s in
the Cards
Name:
_____________
Period: _L
Class/Lab
•
It’s in the Cards
Data Sheet
Tableof the Elements
Properties of the Missing Element
.
Ionization energy
Atomic radius
Atomic mass
Formula of its oxide
Formula of its chloride
Melting point
Density
Formula of its hydride
.
.
Electronegativity
Flinn ChemTopicLabs
—
ThePeriodicTable
Page 5—It’sin the Cards
/.
•
Post-LabQuestions (Useaseparate sheet of paper to answer the following questurns.)
1. Mendeleev’sPeriodicLawcan be stated: “Thephysical and chemical properties of ele
ments areperiodic functions of their atomic masses’ Lookingat your arrangement of
the element cards, describein your own wordswhat the term “periodi function” means.
2. Someofthe the properties listed on each card are periodicproperties, others are not.
Name one property that is periodicand one that is not.
3. The elements in the modern periodictable are arranged in order of increasing atomic
number (insteadof increasing atomic mass).Why didn’tMendeleevuse atomic number to
arrange the elements?
4. From your instructor, obtain a handout showingone possiblearrangement of the ele
ment cards. Identifyeach of the elernehts on the handout with its atomic number and
chemicalsymbol.
.5. Usingthe possiblearrangement of the element cards obtained from your instructor, pick
two of the numerical properties of the elements that are periodic and plot their values on
the graphs below.Giveeach graph a descriptivetitle and labelthe axes.
0
I
-
---
-
-
•
-
-
.
3•
34
It’s
in the Cards
4
5:
6 78
5
67
-
--
t
-
-
-
-
,
,
9 10 11 12 13 14 15 16 17 18 19 20
8
9 10 Ii 12 13 14 15 16 17 18 19 20
Atomic Number
It’sin the Cards-Page .6
/2/
I
t-Lab Questions(continued)
i
6. There are certain trends in the propertiesofthe elements,both within a column (fror
top
to bottom) and acrossa row (fromleftto right) in the periodictable.On the arrow for each
property,write the wordincredsesor decreasesto describehowthat property changes.
,-
Atomic Radius
—
Electronegativity
—
Ionization Energy
H
U
Be
II
Ca Sc
Ti
V
20
21
22
23
24
Sr
38
Y
39
Zr
40
Nb Mo Tc
Cs
55
Ba
56
Rn
88
Fr
87
42
W
Hf
72
41
Ta
73
104
lOS 106
74
43
Re
75
76
7
Br
K
19
Rb
37
27
Rh
45
Ic
‘Ti
N
6
CuT Zn Ga Ge As Sc
31
28
32 33
34
29130
Pd Ag TCd ID Sn Sb Ic
Cr Mn Fe Co Ni
26
Ru
44
Os
C
F
9
I
C
17
12
25
B
5
Al
13
0
8
S
16
3
4,
Na Mg
47I4
I Hg
Si
P
14
15
He
2
Ne
10
Ar
18
Kr
35
36
I
Xe
46
Pt
Au
49
11
50
Pb
51
81
52
Po
53
At
54
Rn
78
79] 80
81
82
83
84
85
86
,
j
La Ce
59
57}58
Ac Tb
I I
107 108 109
Nd Pm
Pa
91
60
U
92
61
Np
9j
Sm
62
Pu
94
•
Eu Gd
63
64
Am Cm
95 96
Tb
65
Bk
97
Dy Ho
66
Cf
98
67
s
E
99
Er Tml Yb
68
69
70171
Pm MO No Lr
tOO 101 102 jtO3
I
7. ,On the outline of the periodic table shownbelow,locate the following:metals, nonmetals,
and metalloids (or sernimetals).Useyour textbook to definethese terms, if necessary.
*
1
Flinn ChemTopic® Labs
—
The PeriodicTable
(
Page7-It’
in the Cards
/
Post-LabQuestions(continued)
8. On the outline of the periodictable shown below,locatethe following:groups or families
of elements, periodsor series of elements, noble gases, alkali metals, a/kaline earth
metals, and halogens. Useyour textbookto definethese terms, if necessary.
*
1
9. On the outline of the PeriodicTableshownbelow,locate the following:transition
elements, inner transition elements, representativeelements. Useyour textbookto
definethese terms, if necessary.
*
t
*
1•
It’s in the Cards
EE
Name
Testing for Metals
Can we determine jf something is a metal?
Materials
•
•
•
•
•
D-size battery
1.5 volt light bulb
Electrical tape
Copper wire
Wire cutter
Different objects to test
•
Hammer
White paper
NQ’L’E: Water is not on the list
of materials needed
!
Procedure to test.for CONDUCTIVITY
1. Cut a piece of copper wire, tape bne end to the,positive end of the battery and
wrap the other end around the bottom of the light bulb.
2. Cut another piece of copper wire and tape one end to the negative end of the
battery.
3. Cut another piece of copper wire and tape one end to the bottom of the light bulb.
4. Take one of the objects you are testing, place the wire that is connected to the
negative side of the battery to one end of the object and place the wire that is
connected to the bottom of the light bulb to the other end of the object.
5. Repeat the last step until you have tested all the objects.
.
.
,
DO NOT USE ELECTRICITY FROM A WALL OUTLET
THIS EXPERIMENT!!
!
The experiment set-up should be as follows:
FOR
/2
Procedure to test for COLOR & LUSTER
1. Put on your safety goggles!
2. Take a piece of white paper, fold it in half, open it, and place it on the lab top.
3. Open vial I and shake about a pea-sized portion of the sample onto the white
paper.
4. Observe the appearance of the sample and record your observations in the “color”
and “luster” columns of the data sheet.
5. Do this for each of the samples you have.
Procedure to test for MALLEABILITY
I Put on your safet goggles!
2. Place a second piece of paper over the top of the sample you prepared in the
section above.
3. Crush the sample with the hammer.
4. Remove the top piece of paper.
5. Observe the sample and record your observations in the “malleability” column of
the data sheet.
6. Do this for each of the samples you have.
iine:
N
OB.JECT
COTNDUC’FIVITY
COLOR
LUSTER
MALLEABILITY
Name:
Discussion:
What properties of elements were you observing in this lab”
What can you infer from your observations? Which objects had characteristics of
metals? Is this true according to the Periodic Table?
Why can metals conduct electricity? Explain in detail (at least 4 sentences).
S
List the properties of metals:
1.
4.
List the properties of metals:
I
4.
S
I
Name:
_________________
Definethe followingterms:
Malleable:
Ductile:
•
Conductivity:
,
Luster:
•
Which element displays the most metallic character? ___________________
Which element displays the least metallic character? ___________________
Which element has the highest electronegativity?Wbat does this tell you
about the element?
4-
Name_____________________________ Date____________ Class
__________
‘5 Laboratory Investigation
Determining Trends in a Group
Problem
How does density change going down a group in the periodic table?
Materials (per group)
balance
mL graduated cylinder
1O
plastic weighing dishes
leadshot(Pb)
silicon pieces (Si)
tin pieces (Sn)
graph paper
metric ruler
-
•
Procedure
O
•
•0
I
-
1. Use the data table on the next page to record all of the measurements
investigation.
you obtain in this
2. Start with any element sample. Check to make sure that the pieces of the sample will fit inside
the graduated cylinder.
3. Using a plastic weighing dish, measure outa sample of the element. The samples should mass
at least 2.0 grams for silicon and tin and at least 5.0 grams for lead. Be sure to account forthe
mass of the dish when measuring your element sample
4. Record the mass of the element sample in the appropriate place in your data table.
5. Pour about 6 mL of water into the graduated cylinder. Record the actual initial volume in your
data table.
6. Add the element srnple to the water in the cylinder. Gently tap the side of the cylinder to
release any trapped air bubbles. Record the final volume in your data table.
7. Calculate the volume of the element sample by subtracting the initial volume from the final
volume. Record this number in your data table.
8. Carefully pour off the water, collect the wet pieces of the element sample, and dry them.
9. Repeat steps 2 through 8 for the remaining two elements.
10. Calculate the density of each element by’ dividing the mass of each element sample by its
volume. Record these numbers in your data table.
I
Prentice-Hall,
chapter 5
23
Date
Name.
5 Laboratory Investigation
Class
__________
(continued)
4
Observations
Measurement
Silicon(SI)
Massoftheelementsample(g)
Initialvolumeof water(mL)
Finalvolumeof waterandelementsample(mL)
Volumeof elementsample(cm3)
Densityof element(g/cm3)
.
Tin (Sn)
4
Lead(Pb)
.
.‘
•
.
.
,
1. Why can you assume that the change in volume in the graduated cylinder is the same as the
volume of the element arnple?
2. Which sample had the greatest volume?
3.
On a separate sheet of graph paper, prepare a graph of density vs. period number for
silicon, tin, and lead.
4
Analysis and Conclusions
i.
How does density change going down a group in the periodic table?
2.
Use your graph to estimate the density of germanium
(C, atomic number 6).
(Ge, atomic number 32) and carbon
3. Calculate the percent error of the densities you determined as compared with accepted values.
4.
What are some possible sources of error in your procedure?
5.
On Yo;r Own Choose another group in the periodic table and look up the densities of the
Do you observe the same periodic trend as you found in this experiment?
elements in that
roup.
.4
24
Chapter5
C Prentice
.Hafl.
Inc.
Name:
___________________________
Chemistry PeriodicTrends
-
Lab: DiscoveringPeriodicTrendsin
Reactivity
Objective:
To comparethereactivitiesof differentmetals..
Materials:
1.“Catciummetal,Ca
2. Magnesiummetal,Mg
3.Aluminummetal,Al
acidsolution,HCI(aq).Caution:HCI(aq)Iscorrosiv
4. Hydrochloric
solution
5. Phenolphthalein
6. Distilledwater
Sixtesttubesanda rack.
Procedure:
1. Puton yoursafetygogglesand apron
2. Comparethereactionsof waterwith Ca,Mg andAl
Ineachof threeof the giventesttubes,addaround10mlof water
thenadd a fewflakesof
anda coupleof dropsof phenolphthalein,
calcium(tube1) magnesium(tube2) andaluminum(tube3).
Recordyour observations;you shouldcheckfor the vigorof the
reaction,andthe degreeof changein the colorof the reaction
solution.
3. Repeatstep2, thistime comparingthe reactionsof the metalswith
HCI(aq).Recordyour observations
TeacherDemonstration: React;vityof GroupI Mt
tals
withwater.
Analysis:
Whatis thetrendin reactivityof the elementsin relationto their
positionon the periodictable? Howwouldyouexpectbarium,
cesiumandsiliconto react?
Lab; DiscoveringPeriodicTrendsin Reactivity
Metal
Calcium
Group Period Reactionin
1120
Color of
SOlutionin
20
H
Reactionin
‘IC’
Aluminum
Solutionin
HCI
-
.
-__-
Magnesium
Color of
1
/,
//
:,
NAME:
Post-Lab Questions:Discovering Periodic
TrendsinReactivity
‘I
1. List the metalsin orderof reactivity(accordingto your observat
ons)
in:
20:
a.H
__________________________
b. HCI:
2. Explainwhy the metalshave a varyingdegree of reactivity.
I
I
*
,
I
I
3. Why did the metalsreact differentlyin water than in acid? Explain.
4. Would you expect these trends with other groups of elements?
Explain.
Properties of Ionic & Covalent Compounds
Problem
How does the properties of ionic compounds differ from the properties of
covalent compounds?
Materials
Salt
Sugar
4 test tubes
test tube rack
Bunsen burner
water
2 100 ml beakers
stopwatch
ters
conductwity te
vegetableoil
Prelab Questions
1. What type of bonding is found in NaCI? Explain your answer.
2. Sugar has the chemical formula 06.C
12
H
What ‘pe of chemical bondng is
found in sugar? Explain your answer.
Procedure
1. Label4testtubes
1—4.
2. Place a small sample of salt (about 1/5 of the test tube) in test tube #1.
3. Place an equal amount of sugar in test tube #2.
4. Using tongs, heat test tube #1 (salt) over the hottest part of the flame of a
bunsen burner. Use the stopwatch to time how long it takes to melt the salt.
5. Repeat step 4 using test tube #2 (sugar).
6.. Fill test tube #3 halfway with vegetable oil. Place a small sample of salt in
the test tube. Shake the test tube for about 10 seconds. Observe the results.
7. Repeat step 6 using test tube #4 with sugar.
8. Pour 50 ml of water into a 100 ml beaker. Pour some salt in & stir until
dissolved. Repeat this step with using a 2 beaker & sugar.
9. Using the conductivity tester, test the electrical conductivity of both the salt&
sugar solutions.
/1*,
$
Observations
I. How long does it take for the salt to melt?
2. How long does it take for the sugar to melt?
3. Does the salt dissolve in the oil?
4. Does the sugar dissolve in the oil?
5. Which compound is a better conductor of electncity?
Postlab Questions
I. What does the time it took for the salt & sugar tell you about their boiling
point?
I
2. How do the properties of the alt & the sugar relate to their bonding?
$
3. Is sugar considered to be a molecular covalent solid or a network covalent
solid? Justify your answer.
4. Would the sait conduct electricity in the solid phase? Explain how this differs
from salt in an aqueous solution.
5. Suppose you were to accidentally mix the sugar & salt samples together. How
would you be able to separate this mixture?
$
e.
Regents Chemistry
Date:
Unit 4
________
-
-
-
Cheniical
Formula
Lewis Structure
-
Type orlionding within
the “molecule” (ionic,
nonpolar covatent,
polar covalent). Why?
Ball & Stick 3
-I)
Representation
is the overall
“molecule” ionic,
nonpolaror polar?
Why?
-
Type of Intermolecular
Force present within
this substance
-
2
F
1111,
13
P
:-
--
-
-
4
CF
.
KI
-
-
Os’,.
Clieniicnl
Formula
-
Lewis Structure
Type ol Bondingwithin
the“molecule”(ionic,
Bali & Stick3-D
s theoverall
“molecule”ionic,
nonpolar covalent,
Representation
nonpolar or polar?
Why?
polarcovalent).Why?
l’4
C
ilL
-
.
11113
3
Nil
Il
-
.
.
.
Type of Intermolecular
Forcepresentwithin
this substance
4.
Chemical
Formula
1
1I
.
lewis Structure
Typeof Bondingwithin
the“molecule”(ionic,
nonpoijir-covalent,
polarcovalent).Why?
Is theoverall
“molecule”ionic,
nonpolaror polar?
Why?
Ball& Stick3-D
Representation
Type f Intermolecular
Forcepresentwithin
this substance
.
c1
3
C11
.
.
-
()
II
2
N
MgO
2
Co
••
0
-
-
0
-
Name
Date
//
Class
‘.
Models
Mdkin
ofCompounds
At first glance, chemical formulas may look complicated. Once you know the
rules for writing them, th
look much more simple. Atoms or ions combine in
y
small whole number ratios. By this we mean such combinations as 1 N with
1 0 (NO) or 1 N with 2 0’s (NO
Not every combi
2)
or 2 N’s with 4 0’s (N
4).
2O
nation will do, however. For ionic compounds, there is just one important
rule: the total positive charge must equal the total negative charge. For
example, in sodium carbonate the charge of the sodium ion is 1+ (Nat) and
the charge of the carbonate ion is 2—(CO
It takes, two sodiwn ions with a
321.
total positive charge of 2+ to neutralize the 2—charge qf just one carbonate
In this laboratory activity, you will practice
ion, giving the formula 3.Na
2CO
monatom
combining
e or polyatomic ions, to give the formulas for ionic
compounds.
negative ion formulas for the vertical
003
I YES
[C
columns and the positive ion formulas
for the rows.
• Use models of ions to assemble formulas.
5. Prepare a table for chemical names by
• List correct formulas foi ionic compounds.
repeating the labels described in step 4.
• Name ionic compounds. from their
Label it Table 2.
formulas.
6. Using the ion models, assemble formulas
for the compounds formed by each posi
MATERIALS
—
tive ion with each negative ion.
Remember: positive charges must equal
negative charges.
7. Count the number of ions of each charge
sheets of ion models (3)
in each formula and write the formula in
scissors
the appropriate space in Table 1.
8.
Write
the name for each compound in
PROCEDURE
Table 2.
1. Cut up the ion model sheets into individ
AND
DATA
OOSEAYATIONS
ual ions.
models
the
ion
into
2.; Separate
two piles:
Negative ion formulas: Use these formulas
positively charged ions and negatively
for the vertical columns, left to right.
charged ions.
F
C1
Br
42
S0
30
2H
C
3. Note that positive ion models have -4and
ion
the
2
3
CO
4
P0
7
0
2
Cr
3
N0
negative
charges on
right
models have charges on the left. This
Positive ion formulas: Use these formulas for
arrangement allows you to match the
the horizontal rows, top to bottom.
charges so that they are neutralized.
K
4
Li
4
Na
4
NH
4
Ag
Note also that when the +‘s and —‘sare
2
Ba
24
Ca
24
Fe
i
24
Mg
lined up, the symbols are in the correct
order to write chemical formulas.
F
Br
cr
4. Prepare a table for formulas similar to
the one under Data and Observations.
4
NH
Labe1 it Table 1 and include all the ion
4
K
formulas listed under Data and
Observations. In the order given, use the
LAB
MANUH
a
a.
0
C)
I
a
C
I
I
8
0
0
)
0
,
4
Li
.....
*
-
Ad
ANALYSIS
1. Which of your positive ions is not a
metal ion?
2. What is a binary compound? Which of
your negative ions always form binary
compounds with metal ions?
CONCLUSIONS
1. Even experienced chemists always
double-check formulas after they have
written them. Check all of your formu
las for the correct numbers of positive
and negative ions so that the charge
equals zero. Also check to see that poly
atomic ions have parentheses around
them if there is more than one in a
formula. Correct any errors.
2. Check the names in Table 2. In which of
these names must a Romart numeral be
used? Why?
3. What kinds of positive and negative
ions always combine in a one-to-one
ratio? Give two examples of compounds
with ions in a one-to-one ratio.
AND
EXTENSION
APPLICATION
1. All the compounds in this activity are
composed of charged ions. What class of
compounds is composed of ions? What
class of compounds is composed of
atoms, not ions?
4. What is the largest number of ions in
any of your formulas? What is the total
positive charge and the total negative
charge in each formula with this many
ions? Give two examples of these
formulas.
2. Two ionic compounds often react so that
the positive and negative ions change
and NaCl
places. For example, AgNO
3
Name
react to form AgCI and NaNO
3.
the two compounds formed by the reac
tion between barium nitrate and potas
sium sulfate. What are the formulas for
these compounds?
3. Find a bottle of vitamins or soft drink
or a package of cookies. Check the
ingredient names on the label and make
a list of any that you think are ionic
compounds. How could you tell?
1.IonModels
fortorq
bor
L
Ion Models—p. 1
S-i
/
I
Lab5
-1
a
E
0
C)
I
0
0
C
0
0
I
U
0
8
C
0
C,
0.
a
110
Chemistry: Concepts and Applications
Lab Manual
S-i
IoriMo forL
1.eIs
boi
torL
/?,‘
I I
ion Models
—
Chemistry: Concepts and Applications
p. 2
Lab 5
-1
Lab Manual
Ti 1
1.IonMo
eIs forLahoratort
S-i
/‘
$
Ion
Lab
3
Models—p.
,+
—
—
’
2
Ca
2
3
CO
2
4
S0
+
—
÷
—
-1
5
—
Ba
ci-
Na’
Cl
Na
+
K’
+
.
‘
‘
Br
—
7
0
2
Cr
.
I
S
+
K’
Br
—
O
3
H
2
C
—
’
2
Mg
—
‘
.
+
+
0
3
H
2
C
I
1
C
—
0
3
H
2
.C
Br
—
Na’
+
a
E
0
C.)
’
2
Fe
.
.4
Li
+
+
I
I
+
—
—
’
3
Al
3
4
P0
+
,J
S
-
3
N0
UI4
S
’
3
‘Al
—
—1
Ti 2
2
3
C0
Chemistry: Concepts and Applications
+
NH
+.
+
—
0
I+
L
.
.5
+
-
+
—
+
—
3
4
P0
+
0
C
COVALENTMOLECULESFROMA 3 DIMENSIONALVIEW
ENGAGEMENT
When two atoms share a pair of electrons,,a single covalentbond is formed. Each atom provides
one of the electronsof the pair. If thetwo atomsare alike,the bondis said.to be nonpolar covalent. If
the atoms are unlike,with one exertinga greaterattractiveforce on the electrons,and.the bond is polar
covalent,then more than one pair of electronscanbe shared.This results in a double or triple bond.
Moleculescanbe either polar
A group of atoms heldtogetherby covalentbondsis called a molecule
.
or nonpolar.If bonds are nonpolar,the moleculeis nonpolar.If bonds are polar, moleculescan still be
nonpolar if the charge distributionthroughoutthe moleculeis symmetrical.A molecules symmetry
depends on its shape, that is, the positions in space of the ‘atoms making up the molecule. Some
possibleshapesare linear, angular(bent),pyramidal,andtetrahedral.
Although we represent moleculeson paper as, being two-dimensionalfor convenience,,hey are
actually three-dimensional.By building molecularmodels,chemists come to understandthe bonding,
shapes,and polarityof even the mostcomplexmolecules.
PURPOSE
Buildthree-dimensionalmodelsof somesimplecovalentmolecules.
Predicttheir shapesand polaritiesfrom knowledgeàf bondsand moleculepolarityrules.
EQUIPMENT
molecularmodel buildingset
‘,,
$I
SAFETY:
Follow all safety procedures as reviewed by your instructor
science agreement.
as well as reviewyour
EXPLORATION
1. Obtain a molecularmodel buildingset. Studythe colorcode identifyingthe differentkindsof atoms.
2. Observethat the followingatoms have one hole which represents1 bonding site: hydr.ogen,fluorine,
chlorine, bromine,and iodine. The atoms with two holes are oxygen and sulfur. A nitrogenatom has
three holes,and a carbonatom hasfour holes.
3. Constructmodelsof the followingmolecules:
2
H
HF
OH
3
CH
0
2
.H
H
2
C
0
2
H
4
CH
CI
CH
2
02
2
Cl
2
N
S
2
H
3
NH
2
CO
•
0•
•
S
•
S
..
EXPLANATION
1. Which moleculeswere nonpolarbecauseall bonds were nonpolar?Be sureto citeevidence.
$
•
•
.
,
I’
-.
I
2. Which moleculeshad polarcovalentbondsbut were nonpolarbecauseof symmetry?Be sureto cite
evidence
3. Which two shapesappearedto producepolar molecules?Besureto citeevidence
EXTENSION
I. Nametwo types of substancesthat do not containmoleculeswithcovalentbonds.
/‘
W
Insidèsand
henlithekidsplayatthe
beach,
theylearnhowto build
and
modelponds,
canals,
sandcastles.
learnabout
Theyalso
theproperties
ofwaterandsand:
Waterwetssaidandflowsfreely
Sandisdenser
between
thegrains.
If a lotofwater
andsinke.
thanwater,
is mixed
withsand,itactsmorelike
a fluidthanasolid:Thewetmixture
thekids’
andresists
slides,
slumps,
sand
to construct
aperfect
attempts
Themoat,buifttorotéct the
castle.
ittocol
sand
castle,
causes
eventually
fromthe
whenwetsandflows
lapse
Butthere
castle’s
foundation.
is
another
typeof sand,caliedmagic
thisway.
behave
sand,thatdpesn
’t
sand
Years
ago,magic wassoldas
suggested
atoy.Theinstructions
waterinalargeglassbowland
putting
otmagic
ina smallamount
sprinkling
likebeach
ofsinking,
sand.Instead
sandwouldfloat!By
sand.themagic
spunkling
moreandmore
ontothesandraft.ftcouldbe
tothebottom.
macetoplunge
It youweretoholdbeach
sanoinonehandandmagic
saneintheotherandlower
the
bothhanasintothewater,
beachsanewould
clearly
showindividual
grains.The
would
sand,however,
magic
tobesurrounded
bya
appear
likeplas
layerlooking
silvery
ncfilm Whenyoultheoyour
iianasouiofthewaler,the
beachsancwouldbewet.
wrrnesgrains
clumping
Beach
sandismostlymineral
intotinypieces.
quartzbroken
Itschemicalnameis siIi
a
and
thegrainsconsist
ofsilicon
and
bonded
ina
covalently
oxygen,
three-dimensional
network
of
billionsofatoms.
Theinterior
01theparficlecontains
twiceas
byDavidRRobson
I
atomsassilicon
manyoxygen
andcanberepresented
atoms,
bythe
formula2
of
thesurface
K) However,
.5
theparticle
containsoxygen
a1onthai
bonded
arecovalently
tohydrogen
atoms(seeFigure
1).These
arepolar
covalentbonds,likethe0—H
bondsin
Thismeans
watermolecules.
thatboth
thesurface
ofthesandaridthewater
molecules
havepositiveandnëgativè
electncai
and,consequently,
charges
waterisattiactedlothesand.Water
‘wets sand(thatis,adropatwater
willspread
outonasandgrain
instead
intoa bead.)Inthejargon.
olgathering
ofchemistry,
thesand
grains
arehydrophilic—water
loving.
01magic
togetherThegrains
sanewouldnotbeclumpec
tact,theywould
IoQemef—tn
bepertectlycry! Thisis
sandwas
memagic
oecause
surrounoed
byalargeairbub
bie:theSilverylayerwasthe
curvecsurtaceotthebubble. This
experiment begins with a layer of motoroil floating on water
Whatismawcsaneandwhy (top photo). When Magic Sand is added (bottom photo), it sinks and
oo C ac sostrangetv?
carries a glob of oil with it to the bottoni.
B ChEMMAflERS.
JiL
19
Where’sthe magic?
sandisbeach
sand
Magic
withminute
coated
particles.
ot
treated
silica.
chemically
Researchers
at theCabot
Corporation
perfected
a
forexposing
glass,
process
andothermaterials
silica,
to
ofasilicon
compound
vapors
called
thmettiy$chlorosilane,
inFigure
Asshown
SiCl.
)3
(CH
molecules
attach
2,these
to
themicrospheres
andgiveita
ofnonpolar
newsurtace
cova
These
lentbonds.
bonds
do
notattract
waterandthus
make
theparticles
hydropho
bic—water
hating.
of
Whena fewgrains
sandaresprinkled
on
magic
thepolarwatermole
water,
otherpolarwater
culesattract
molecules
sostronglythat
/2g.
i3
• Ho—
3
e--cH
I
+
D
3)10,5C_/
t
.
4
.
-
r”
—
1
’i)4
4
Figure1: Mostsandis impure
silica, whichhas a networkof
oxygen and silicon atoms. At
the surface,the oxygen forms
poLarcovalent bonds wilhi
hydrogenatoms.These0—H
groupsCartypartialelectrical
chargesthat attractsimilar
partialchargesin water mole
cules.Theawaction of oppo
site chargesmakeswater
adhereto eachgrain of sand.
Figure2: Magicsandconsistsof ordinarysand
grains coated with tiny particles of pure silica which
have received a Wecial chemicaltreabneriLWhen
the particles are exposedto trimethylhydroxys
llane,
a reactiontakes place betweentwo —OHgroups.
on
nat
This resuits in the for
of water, and the
bondingof the silanecompound to the silica
particles. Foilowingthis treatment, the exterior of
the particle contains -CH
3
groups that are soluble in
oil but are insoluble in water.
thesur
through
tt preventthegrainsofmagicsandfrombreaking
ey
ratherthick.Whenthemagic
faceuntilThelayerofsandbecomes
thesamesurlace
lension
effectalsokeepsit dry.
sandfinallysinks,
Theair between
cannotbeforcedoutbecause
thewater
thegrains
willnotflowbetween
Thehydrophobic
oil
molecules
grains.However,
willreadily
flowbetween
thegrains,andmagicsandcanabsorba
of oil.
surprisingquantity
Magic at wi’t
Figure 3: DiggingthroughfrozenArctic
soil normallyrequireshoursof work with
power tools. To speedunderground
repairs, utility companies can cover
electrical junctionboxeswithmagicsand
and cap the sandwith just a fewinches
of soil Rainwater flows around,not
through, the magicsand (color)and,
whenthe soil freezes solid, the magic
sand remains dry and Ipose. It is easy to
breakthroughthefrozencap,then
shovel awaytheloose magicsand.
Theutilitiesprefer
wires
toburyelectricandtelephone
toprotect
themfromtheharshweather
boxneeds,
but,if awireorjunction
it isnearlyimpossible
todigupThedeeply
duringthe winter,
repair
If the unctionboxiscovered
withmagic
frozen
earth.
sand,however,
ii canbeserviced
themagicsand
remains
easilybecause
dry and
looseyearround
3).
(seeFigure
44
The future of magicsandis in yourhands
all been
Theapplications
described
above
have
tested
successfully
usedtoday.The manufac
WhentheCabotCorporation
w
ork
of
notbeing
originallydeveloped
are
but,foravariety reasons,
magicsand,
it mighibeuseful
for cleansing
W.Estes
issearchingfor
sand,the Cidlord
waterof oilycorflarnina
turerofmagic
Company,
ersexpected
f
or
sand?It so.
o
f
beuseful
f
or
C
an
think
thatmagic
sand
application
magic
I
t
non. wasalsosuggested
newuses. you
a new
might
the Chern
fromoil
in
coastal
When
i
dea.
then
tankers
Waters.
experiments
totest
enter
petroleum
s
pilled
s
imple
trapping
trysome
your
Mattersmagicsandcontest.
spnnkledonfloatingpetroleum,
magicsandwouldmixwiththeoily
ke it sink.Thiswouldpreventthepetro
material.addweight,andrr
REFERENCES
marshesandbeaches
leumfromcornarninaling
and,theoretically,
VIlz, E. MagicSand:Morielrigthe Hydrophobic
EfieciandRevec5eoTested Dernonstrabons,
Phase Liquid Cl
Journal ot
matography,
to beter dredgedfromThebottom.
permitthepetroleum
1990,67
6). 512.
II youhaveeverraisedpottedplants,youknowhowdifflailt it ls
Educatia, 1982, 59,155.
of Cflem,r
,a!
al
Hcthnan, & B.Journ
theplants,theywilt;overto waterthemcorrectly.If youunder-waler
waler,andtherootsrot.Theproblemis thattherootsofmostplants
needtobeexposed
to bothwaterandair. If yougivethe planttoo
muchwaler,youwill displace
thevitalair.WhenmagicsandIsadded
topothngsoil,thehydrophobic
grainspermitairto flowbetween
them,butnotwater,andthusmairttain
to thesur
openair channels
face.
testedbyutilitycompanies
intheArctic.
Magicsandhasalsobeen
CHEM MATFERS.APRIL 1994 9
-4
Name
______________________
.,
11/07/01
MagicSandLab..
Directions: Please answer the following questions based on the article
entitled, Magic Sand and your chemistry knowledge. Be sure to use
complete sentences. Please use a separate piece of paper. When you’r
fmished, your lab group may try the experiment listed that follows.
.
Questions:
1. What mineral is the main component of beach.sand?
2. What type of bonding does the mineral of beach sand contain?
3. What feature of the surface of a grain of sand makes water attracted to it
so that the water wets the sand?
4. What are polar covalent bonds?
5. What do the terms hydrophobic and hydrophilic mean?
6. in what way is magic sand different from ordinary beach sand?
7. How does this difference affect the attraction o.fwater for the surface of
the grains of sand? In other words, why does the magic sand float in
water when the beach sand sinks?
8. The Esies Company had two uses in mind when they developed magic
sand. Name one.
9. Why can magic sand absorb oil but not water?
IO.The article suggested thai magic sand might improve soil conditions for
plants.How can magic sand improve the soil in which plants are raised?
I l.How is magic sand useful in the Arctic?
12.Can you think of a new application for magic sand?
Experimenton the back
*
a
I
Eiperiment:.
S
.
1. Add waler to a large test tube untilit is about two-thirds full. Place the
test tube in a test tube rack.
2. Using a metric ruler and a marker, make a mark on the test tube 5mm
above the top of the waterlevel line.
3. Use a graduated cylinderto measure 3m1of vegetable oil.or motoroil.
4. Using a plasticpipette,.add the vegetableoil to the test tube to form a
layerabout5mm deep.
ml
5. Measurethevolumeof vegetable oil used.
6. Using theelectronicbalanceand a plastic weigh boat,measure two :graims
of magic sand.
7. Sprinkle just enoughmagic sand on the oil to cause it to sink. Continueto
addmagic.sandto the test tubeuntilall theoilhas sunk to thebottom.
8. Weighthe amount of magic sandthatremains(unused).
9. ,Use yourdatato answer the followingquestion.
S
.
.
__________
On January 7, 1994, a barge carrying 1.5 million gallons of heating oil
strucka reef just 300 meters from San Juan’smain touristbeach. About
750,000 gallons (about2,840,000 liters) of oil spilled into the surrounding
water.
S
1. Accordmg to your experiment, calculate how much magic sandwouldbe
requiredto sink the oil that was spilled.
2.. If magic sand costs 90 cents per pound, whatwould be the cost of this
muchsand?
Name
’
/2
Class
Date
Oxidation
Formulas
and
Nvmhers
Oxidation numbers and the charges of ions give the information needed to
write the formulas of many chemical compounds. Only a few guidelines
are needed.
1. In a neutral compound, the charges on ions, or the oxidation numbers,
balance out to zero.
2. One positive charge balances one negative charge.
3. Ions with positive charges or positive oxidation numbers are written first.
4. Subscripts show the relative numbers of atoms or ions in a compound.
5. To show more than one of a polyatomic ion, the symbol is enclosed in
parentheses and the subscript follows; for example, 4
2(S0
)3.A1
In this activity, you will use paper models to show how chemical formulas are
derived from oxidation numbers.
OAJEC11YES
MATERIALS
a
C
0)
C
-
C,
E
0
()
1
C,
C.)
-
)
0
I
V.
0
V.
V-V.
-
-
paper
sheet of ion models
PRO
CEO
HHE
1. Cut out each of the ion squares on your
sheet of ion models.
2. Assemble the ions for a compound con
taining nickel(II) and iodide ions. To do
2
ion on a piece of
this, place the Ni
DATA
AND
DOSED
VHTIONS
C,
C,
Combining Ions
C,
nickel(II) and iodide
0)
0
0
C
0)
C
-V.
B-2
paper. Place enough 1 ions alongside the
2
Ni
ion to balance the dharges.
3. Record the formula and nam of the com
pound of nickel(II) and iodine in a data
table similar to the one shown.
4. Use the rules listed in te introduction
and in your textbook for writing forn
ulas
and naming compounds. Assemble the
ions for five compounds from the follow
ing list and record their formulas and
names in your data table. Nickel(III) and
chlorite; zinc and oxide; copper(I) and
sulfide; nickel(IH) and chlorate; tin(II)
and sulfite; copper(lI) and iodide; tin(IV)
and sulfide; nickel(III) and oxide;
copper(II) and sulfite; zinc and hydrogen
sulfite.
• Write formulas of chemical compounds.
• Name chemical compounds.
scissors
pencil
I MANU
L1U
0
C.)
Chemical Formula
2
Ni!
Name of Compound
nickel(II) iodide
515
NRLY
1. Some oompounds are’described as “bina
ry compounds.” Whet does this mean?
List theformulas and names of any
binary compounds you have constructed
2. Which elements on your list form ions
with two different oxidation numbers?
3. Parentheses must be used to show more
than one of a polyatomic ion. List the
formulas of any cOmpounds on your list
where this was necessary:
CONCLUSIONS
Should the formulas you have written be
described as molecular formulas? Explain.
EXTENSION
QNII
QPPLICHTIQN
/3
1. Some elements have more than one
oxidation number. To show the oidation
number of such elements in a coi
ipound,
a Roman numeral is given in the name
of the compound. Give names for :the
• following compounds.
a. UF
5
c. PbCI
2
b. :UF
d. PbC1
6
4
2. Manganese has an oxidation number of
4÷ in a number of compounds. Write the
formulas and names of compounds of
manganese (IV) with oxygen and
ne.
brorn
•
3. Which of the following are molecular
formulas?
20
H
NaBr
4
CH
80C
• 4H
4
MnSO
4. Hydrogen peroxide and water both con
tain the same two elements. Using
reference materials, write the chemical
formulas for these two substances and
describe their properties and uses.
C
a’
a’
C
a’
a
E
0
0
I
a’
C
a’
a
0
.5
0
a’
a’
a’
0
C
C
C)
0
•
>
a.
0
C)
46
Chemistry:Concepts and Applications
Lab Manual, Chapter 5
Is for or
1.IonMod
tor S-i
Ion Models
—
p. 1
/j/
5-1
Lab
d
E
0
0
I
0
0
C
110
Chemistry: Concepts and Applications
Lab Manual
eIs
L Ion Mo
bordtorq
for L
Ion Models
—
H
Lab 5-1
p. 2
+
—
2
Ca
—
2
3
CO
S042
+
—
+
4
NH
+
Na
+
.
+
2
Ba
Na”
+
+
+
+
+
2
Ba
+
Li
+
—
Li
+
F
—
’
2
Mg
+
Li
d
Li’”
+
+
C
C
a
C
0
+
-
+
-
w
(0
-
2
Fe
I
C
C
0
3
N0
+
-
+
—
01
3
H
2
C
3
NO
Br
-
ci
2
Fe
(0
Br
—
Br
I
(0
C,
-
Ci-
-
Ci
0)
8
C
0)
Ag
0
a
0
0
—
+
—
2
4
S0
Chemistry: Concepts and Applications
Ag
+
2
4
S0
Lab Manual
Ti 1
S-i
1.IOn
Models
Ior
forLdbor
/J3
/s
Ion Models
—
p. 3
•
Lab 5-1
+
—
+
—
+
—
—
—
CI.
Na’
Cu-
Na
+
—
Br-
K’
+
—
Br-
K
+
Ba?
•
2
3
co
2
so
2
Ca
.
+
—
,
7
0
2
Cr
.
+
—
O
3
H
2
C
—
0
3
H
2
C
,
.
2
Mg
I
:
+
—
ci
—
0
3
H
2
C
0
0
+
•
—
Br
Na’
+
0
’
2
Fe
•
+
Li
“4
+
3
AI
4
PO
+
K
+
1.11.1
—
—
a.
E
4
NH
+
0
I
0
+
+
0
C
0
•0
0
I
+
—
+
,J
•
+
0U
0
8
C
0
3
N0
3
AI
—
•
2
3
C0
.
112
+
Chemistry: Concepts and Applications
0
0
-
+
—
+
—
3
4
P0
0)
>
a
0
0
/J,
Lah
’1
tha
i,,
To determinethe numberof chalk particles needed to
Purpose:
write your full name.
“
Materials: Chalk, balance, blackboardor construction, paper, paper towel, metric ruler
Procedure:
I. Wipe a pièce chalk free of dust’particleswith a paper towel.
2. Using the electronic balance determinethe mass of the chalk to the nearest 0.01
de of calcium carbonateonly.)
gram. (Assume’tlat the chalk is n
-
3. Using the chalk, write your full name •on the,,blackboard (or dark colored
constructon paper). The letters should be no bigger than 5 cm eack
’of
4. Wipe the chalk fre
dust again.
5. Reweigh the chalk to the nearest 0.01-grain.
Observations:
Mass of chalk prior to writing
_________________
Mass of chalk after writing
__________________
MassOfchalk used
______________
Questions: (Be sure to show all”of your calculauons.)
I. Whatis the chemical formulafor calcium carbonate?
2. What type of bonding does calcium carbonatehave?
3. Whatis the grain formula mass of calcium carbonate?
/Js
4. Flow many moles of calcium carbonatewere used towrite yur
of chalk?
name with a piece
5. How many molecules of calcium carbonatewere used to write your name with a
piece of chalk?
6. What percentby mass of calcium carbonate is made up of calcium?
7. How many grams of calcium are in used in writing your name wth a piece of
chalk?
8. Calciumcarbonate is found in limestone eggshells and seashells, it is also used in
certain brands of antacids to help neutralize stomach acid and prevent heaitburn.
Write a balanced equation for the following reaction: Aqueous hydrochloric acid
reacts with solid calcium carbonate to yield (produce) liquid water, carbon
dioxide gas and aqueous calcium chloride. Be sure to use Reference Tables E, K
and the Periodic Table of Elements to help.
9. Whatis the coefficient of hydrochloric acid when the reaction is balanced?
10. How many moles of calcium carbonatewould be needed to neutralize 10 moles of
hydrochloricacid?
Date
Name__
S
Class _________
__________
Lab
of Hydrates
• composition
Textreference:Chapter7. pp. 154—1S
pre-Lab Discussion
•
+
Hydrates are ionic compounds isalts) that have a definite amount of water
(water of hydration) as part of their structure. The water is chemically
combined with the salt in a definite ratio. Ratios vary in different hydrates
but are specific for any given hydrate.
The formula of a hydrate is represented in a special manner. The
2O.
xH
hydrate of.copper sulfate in this experiment has the formula CuSO
4
The unit formula for the salt appears first, and the water formula is last.
The raised dot means that the water is loosely bonded to the salt. The
coefficient x stands for the number of molecules of water bonded to one
unit of salt. This special ‘ormula. like all other formulas, illustrates the
law of definite composition.
When hydrates are heated, the “water of hydration” is released as
vapor. The remaining solid is known as the anhydrous salt. The general
reaction for heating a hydrate is:
--e
—
.-
hydrate
•
anhydrous salt
+
water
The percent of water in a hydrate can be found experimentally by
accurately determining the mass of the hydrate and the mass of the
anhydrous salt. The difference in mass is due to the water lost by the
hydrate. The percentage of water in the original hydrate can easily be
calculated:
mass H.,O
x 100
O =
percent H
mass hydrate
-
In this experiment, as was mentioned, a hydrate of copper sulfate will
xH,O). The change froTTT
hydrate to anhydrous salt is
be studied (CuSO
4
accompanied by a change in color:
4CuSO ‘rH,()
blue
-‘
—
4CuSO
white
+
0O
xH
Tlus investiganon should aid in the understanding of the formulas and
composition of hydrates and the law of definite composition.
Purpose
Determine the percentage Of water in a hydrate.
Equipment
evaporating dish, porcelain
crucible tongs
microspatula
laboratory balance
ring stand
iron ring
wire gauze
laboratory burner
safety go,ggles
lab apron or coat
57
Materials
4
SO
copper sulfate hydrate. Ct
Safety
,O
xH
fl
Do not touch a hot evaporating dish with your hands. Te hack long hair
and secure loose clothing when working around anopen ftámv. Now the’
caution alert symbols here and beside certain st.eps in the edure.’
‘Pro
Refer to page xi to review the special precautions associated with each
symbol.
Be sure to wear a lab apron or coat and safety goggles when working
in the lab.
Procedure
4
1. Prepare the setup shown in Figure 11-1.
Figure 11-1
2. Heat the dish with the hottest part of the flame for 3 minutes.
3. Using crucible tongs, remove the evaporating dish from the
apparatus. Place it on an insulated pad and allow it to cool for
several minutes.
4. Find the mass of the evaporating dish to 0.Ol g. Record the
mass in the Observations and Data section.
5. With the evaporating dish on the balance, measure into it
exactly 2.00 g of copper sulfate hydrate. Record the data below.
6. Place the evaporating dish + hydrate on the wire gauze.
Gently heat the dish by moving the burner back and forth around
the base. Increase the heat gradually. Avoid any popping and
spattering.
7. Heat strongly for 5 minutes or until the blue color has disap
peared. During heating, a microspatula may be used to spread”
the solid and break up any “caked” portions of the hydrate. Be
careful not to pick up any of the solid on the microspatula. If
the edges of the solid appear to be turning brown, remove the
heat momentarily and resume heating at a gentler rate.
58
(
Name
—
1
7
icic;
11 Compositionof Hydrate (continued)
$
S. Allow the evaporating dish to co
for about a minute. im
I,
mediately fInd the mass of the dish arthydi-ous sail. and record
the data below.
—
Observationsand Data
a. Mass of evaporating dish
b. Mass of evaporating dish
c. Mass of evaporating dish
+
-
hydrate
anhydrous salt
___________
g
___________
g
___________
g
Calculations
1. Find the mass of the hydrate used (b —a).
2. Findthemassofthewatèrlost(b—c).
3. Find the percentage of water in the hydrate:
•
percent H.,O =
-
mass water
x 100
mass hydrate
Conclusions
and Questions
1. The true value for the percentage of water i.his
is your experimental error?
hydrate is 36.0%.What
2.- Why must you allow the evaporating dish to cool before measuring its
mass?
3. Why must you measure the mass of the anhydrous salt immediately
upon cooling?
59
Name
Date
_________________________________
___________
Class
___________
Lab
RelatingMolesto
of a
Coefficients
ChemicalEquation
Text reference: Chapter 10. pp. 233—238
Pre-Lab.Discussion
The mole is defined as Avogadro’s number (6.02 x 1023)Of particles. These
particles may be atoms, molecules, formula units, ions, electrons, etc. The
concept of the mole is very important, especially wheh dealing with quan
titative studies of chemical reactions. When alculating quantities of solids
or liquids, molar masses are used. The molar mass of a substance is the
mass, in grams, of 1 mole of particles of that substance. When calculating
quantities of gases, molar volumes are used. The molar, volume is the
volume occupied by 1 mole of a gas at STP.
Chemical reactions are represented by balanced chemical equations.
Proper interpretation of ap. equation provides a great deal of information
about the reaction it represents and abput the substances involved in the
reaction. For example, the coefficients in a balanced equation indicate the
number of moles of each substance. Thus, the ratio of moles of a substance to moles of any other substance in the reaction can b determined
I
at a glance.
In this experiment, iron filings will be added to an aqueous solution
of copper(1l) sulfate. A single replacement reaction will take place, the
products being iron(ll) sulfate and copper metal. The balanced equation
for this reaction is:
Fe(s)
+
4(aq)
CuSO
—
4(aq)
FeSO
+
Cu(s).
The quantities of iron and copper sulfate used as reactants will be
such that the copper sulfate will be in excess. Thus, the iron will be the
limiting factor in determining the number of moles (gram-atoms) of prod
ucts that will be formed. As the equation shows, the number of moles of
copper produced should be equal to the number of moles of iron reacted.
This experiment should aid in the understanding of balanced equa
tions and single replacement reactions.
Purpose
Find the ratio of moles of a reactant to moles of a product of a chemical
reaction. Relate this ratio to the coefficients of these substances in the
balanced equation for the reaction.
Equipment
S
balance
burner
beaker, I00-niL
beaker, 250-mL
graduated cylinder, 100-niL
ring stand
iron ring
wire gauze
glass stirring rod
safety goggles
lab apron or coat
75
14o
Materials
4)
copper sulfate crystals (CuSO
iron filings (Fe)
H
Safety
Tie back long hair and secure loose clothing when working with an open
flame. Note the caution alert symbol under “Procedure” and follow the
precautions indicated. Always wear safety goggles and a lá.b apron or coat
when working in the lab.
Procedure
1. Find the mass of a clean, dry 100-mL,beaker. Record this as
(a) in your data. table:
2. Measure o%It8.0 grams of copper sulfate crystals (CuSO
4)
and add these to the beaker.
3. Measure 50.0 mL of water in a graduated cylinder and add
it to the crystals in the beaker.
While one lab partner continues with steps 4 and 5, the other
partner should carry out the instructons in stèp6.
4. Set up the apparatus as shown in Figure 15-1. Heat the mix
w
ture in the beaker to j’ust bek
boiling. DO NOT ALLOWTHE
LIQUID TO BOIL.
’
‘,I
100-mL
beaker
Figure 15-1
5. Continue heating and stir the mixture until the crystals are
completely dissolved. Turn off the gas and remove the burner.
6. Using the balance, measure precisely 2.24 grams of iron
filings. (Remember: do not place any reagent directly on the
balance pan.) Record this mass as (b) in the data table.
7. Add the iron filings, smaLl. amounts at a time, to the hot
copper sulfate solution. Stir continuously. After all the iron has
been added and the mixture stirred, allow the beaker to sit for
10 minutes while the reaction proceeds. Record your observa
tions as (d) in the data table.
76
Name
74’.
‘15 Relating Moles to Coefficients ofa
Chemical Equation
(continued)
8. Decant the liquid into a 250-mL beaker as shown in Figure
15-2. Do not disturb the solid at the bottom of the beaker.
settled
precipitate
Figure 15-2
9. Add about 10 mL of water to the solid in the 100-mL beaker.
Stir vigorously in order to wash off the solid. Let the solid settle
and decant the liquid. Repeat the washing.
10. Spread the solid out on the bottom of the beaker and place
the beaker in a drawer or oven to dry. Complete step 11 and the
rest of this experiment at the beginning of the next lab period.
11. Find the mass of the beaker and the dry copper metal. Re
cord this as (c) in the data table.
andData
Observations
a. Mass of empty beaker
_g
b. Mass of iron filings
c. Mass of beaker
+ copper
g
_g
d. Visual observations:
77
Calculations
Use the following information, as. needed, to carry out the calculations:
mass(g)
no. of gram-atoms
g-atomic mass of Fe
gatomic mass of Cu
$
g-atomic mass
=
=
56 g Fe/g-atom Fe
64 g CuJg-atom Cu
1. Find the mass of the copper produced:
c
—
a
_____________
g
2. Find the number of g-atoms of copper
produced:
3. Find the nupiber of g-atoms of iron reacted:
4. Find the whole number ratio of g-atoms of iron to
g-atoms of copper
Conclusionsand Questions
1. ‘How,does ‘the ratio found in calculation 4 compare with the ratio 9f
the coefficients of the same two metals in the balanced equation for the
reaction?
2. How many moles (g-atoms) of copper sulfate are used to produce the
solution in this experiment? Why is this amount of copper sulfate said to
be “in excess”?
3. Explain why the iron is the limiting factor in this experiment.
78
Name
‘
15 RelatingMolesto Coefficientsofa
td.
ChemicalEquation(continued)
ment
4. A general description of the single replac
reaction in this exper
+
+
salt
in
solution
“new”
metal
ixnent is:
metal “new” salt solution.
another
for
Give a balanced equation
example of this type of single re
placement reaction.
—
5. Give general descriptions of two other types of single replacement
of each type.
reactions. Using balanced equations, give a specific exa
nple
$1
:
6. Consider’&he reaction: Cu(s) -I- 2AgNO
3(aq)
2Ag(s) + Cu(N0
3)2(aq).
‘If 3 moles of copper metal reacts, how many moles of silver metal will be
s produced?
—
•,,
.
t,
--.
—
--.
‘
79
Name
Perfect Cup of Kool Aid
I
Pcirtl: YourPerfectCup
Goal: Makea cupof KoolAid that you betievetastesperfect.
1. Youhaveto makesure that you weighthe amountof fJavoredmix
that you add to yourcup.
Massadded(g)
(Besure to weighyour mix on TAREbweighing“paper)
2. Nowaddthe water. Makesure that you record the amountof
water youare addingusinga VERYCLEANgraduatedcylinder.
We havenewcylinders that are to be usedfor this lab. bo NOT
useoneoff the shelf!
Volumeused(mL)
,
‘
_______
3. betermine the MOLARITYof your KoolAid usingthe steps found
on the questionsheet.
Part II: Dilutions
**Assumethat your perfect cup was1OM.
Howmanymolesare in 1L of 1OMKoolAid?
1. Measureout 5 ml of perfect KoolAid anddilute with water to 20
ml. Observations- what did you notice aboutthe color?
2. Measureout 5 ml of perfect KoolAid anddilute with water to 50
ml. Observations- what did you notice about the color?
3. Measureout 2 ml of perfect KoolAid anddilute with water to 50
mI.Observations-)what did you notice about the color?
/4
Calculations:
1. What is the molarity of eachof your diluted solutions?
Remember,we assumedthat everyone’sperfect KoolAid is
1OM.
V
1
M
V
2
M
2. Howmanymolesare there in eachof your diluted solutions?
Molarity molesof solute/Liters of solution
Name
PerfectCupof KoolAidLabQuestions
1. befinethefo
llowjngterms:
•
Solution-
•
Solute-
•
Solvent
• Molarity
-
—
2. What were the solutes in the Kool Aid Lab?
3. What was the solvent in the Kool Aid Lab?
______
___
4. What is the Molarity (M)equation?
5. How many grams of sugar (crose)
Perfect Cup of Kool Aid?
did you use to make the
_________________
6. How many grams of Kool Aid powder did you useto make the
Perfect Cup of Kool Ad?
__________________
7. How many milliliters of water did you use to make the Perfect
Cupof Kool Aid?
________________
Flip the page over.
/47,.
Use the following information aboutKickin’Kiwi—LimeKoolAid to
answerthe questiOnsthat follow. Be sure to show your
calcukrtions.
Ingredients/Directions to mcikea singlepackageof Kickin’Kiwi
Lime KoolAid:
1 package: 4.8 gramsof KoolAid pOwder
1 cup of sugar (sucrose) 190 grams
2 quarts of water: 1892milliliters (1.892Liters
)
A student wants to makeoneglassofKoOlAid. The gloss holds
200 ml of water.
8. How manygrams of KoolAid will the student need?
-
___
_________
9. How manygrams of sugar will the student need?
10.
What is the formula mass of sucrose )?
21
H
12
(C
1
0
2
11. How many moles is 1.90grarrts of sucrose?
_____________
12. How many grams of sugar are needed to make a 200-mJof a
2.78 M solution of Kool Aid?
______________
Page 1 FactorsAffectingSolutionFormation
-
FactorsAffecting Solution Formation
An Inquiry-BasedApproach
IntroductiOn
Coppersulfate,a crystallineblue solidthat readilydissolvesin water, is an important agri
cultural chemical.In solidform the compound is mixedinto animal feedsto prevent copper
deficiencyin farm animals.Solutions of copper sulfate are sprayedon plants, including
wheat, potatoes,tomatoes, grapes, and citrus fruits, to control fungus diseases.Although
copper sulfateis solublein water,the rate at which it dissolvescan be fast or slow,depending
on conditions.Whatfactorsaffectthe rate a which an ionic soliddissolvesin water?
‘
Concepts
• Solution
•
Solubility
• Solute
•
Solvent
Background
Asolution is a mixtureof two tr more pure substancesthat is homogeneousor uniform
throughout. Thesubstancethat is being dissolvedis calledthe solute, and the substance that
does the dissolvingis called the solvent. Solubility,definedas the amount of solute that will
dissolvein a givenamount of solventat a particular temperature, depends on the nature of
the solute and the solvent,and how they interact. Althoughthe solubilityof a compound
governs howmuch solute may dissolve,it does not predict how fast the solute will dissolve.
’hich a solidwill dissolvein a liquid are the
Some of the factorsthat may affectthe rate at
particle size ofthe solid,the temperature of the solvent, the amount of stirring or agitation
of the mixture, howmuch solute is already dissolvedin the solvent, and the presence of
other dissolvedsolutes.
The process of an ionicsolute dissolvingin water is a surface phenomenon. Free-moving
water moleculesrandomlycollidewith ions on the surfaceof the solid.The water molecules
interact with the ions bymeans of ion-dipoleattractive forcesand gradually“chip”awayat
the surface ions,helpingto separate them from the bulk crystal.Asthe surfaceions dissolve,
the next layer of ions becomesthe new surface layer.This interaction at the surface of a crys
tal continues until the crystal is completelydissolvedor until the solution can accept no
more solute.
Understandingthe way in which an ionic solute dissolvescan help us design conditions to
optimize the rate at which the solid dissolves.
Experiment Overview
The purpose of this inquiry-basedactivity is to investigatethe effectsof crystalsize, degree
of mixing, and temperature on the rate at which copper(1J)sulfate pentahydrate
dissolvesin water.
45H)
(CuSO
2O
FactorsAffectingSolutionFormation
FactorsAffectingSolutionFormation Page2
-
/4’Z
Pre-Lab Questions
•
1. Use the “surfacemodel”describedin the Backgroundto predict liow changing each of
the followingvariableswill change howfast a crystallineionic compounddissolvesin
water: (a)amount of stirring or agitation;(b) temperature of water; (c) size of th crystals.
2. Outline a seriesof tests to determine howeach variablewillaffectthe rate at which cop
per(II) sulfate pentahydratedissolvesin water. Each test should lookat the effectof
changing onlyone variableat a time—allof the other variablesmust be controlled or
held constant for comparison.
3. Readthe Materialssectionand the recommendedSafety Precautions.Write a step-bystep procedure for the experiment,including any safetyprecautions that must be fol
lowed.Note: There are two crystal sizesof copper sulfate.Useabout 0.2 g of the fine
crystal gradeas the control variable.Fill the test tubesabout one-third fullwith distilled
or deionizedwater.
Materials
fine crystals, 1—2g
Copper(II)sulfate, 2O
45H,
CuSO
crystal lumps, 0.2 g
Copper(II)sulfate, 2O
45H,
CuSO
Distilledor deionizedwater
Ice
Balance,centigram (0.01g) precision
Beakers,100-mL,2
Graduatedcylinder,10- or 25-mL
Hot plate or hot water
Mortarand pestle
Spatula
Stirring rod (optional)
nal)
ti
Stoppersto fit test tubes, 3 (op
Testtubes, large, 3
Testtube clamp
‘Testtube rack
Thermometer
Timer
Weighingpaper or dishes
Safety Precautions
Copper(II)sulfate is moderately toxic by ingestion and is a skin and respiratorytract
irritant.Avoid contact with skin and eyes. Wearchemicalsplashgoggles and chemicalresistantgloves and apron. Washhands thoroughly with soap and water before leaving the
laboratory.
Procedure
the procedurewith your instructor and reviewall safetyprecautions.
1. Veri1
’
2. Carryout the procedure and record all data in a suitabledata table.
3. Answerthe followingPost-Lab Questions.
Flinn ChemTopic’Labs SolubilityandSolutions
—
Page3- FactorsAffectingSolutionFormation
/
Post-Lab Questions
1. Whateffectdid mixingor shaking have on the rate at which the solute dissolvedin water?
Givespecificevidencefrom your tests and explainin terms of the “surfacemodel”for
solutionformation.
2 Howdid increasingthe temperature of the solvent change the rate at which copper
sulfatedissolvedin water?Wasthe oppositeeffectobservedwhen coppersulfate was
dissolvedin coldwater?
3. Usethe definitionof temperature and the kinetic theory to explainthe effectof tempera
ture on the rate of solution formationfor coppersulfate.
4. In some cases,increasing the temperature may be a disadvantagein preparinga solutioh.
Suggesta possiblecase where heating the solution might be a problem,
5. What was the effectof crystalsize on the rate of dissolving?Howdo these results support
the surfacemodelfor solution formation?
6. Usethe results of this experimentto predict howthe rate of dissolvingwouldbe affectedif
you tried to dissolvemore copper sulfatein a solution that alreadycontained some copper
sulfaterather than in distilledwater.Note:Assumethat the total (combined)amount of
solute is lessthan the solubilitylimit of copper sulfate.
7. Write a short paragraphdescribing an optimum procedure for preparinga solution.
FactorsAffectingSolutionFormation
Page1 Solubilityand Temperatu.e
-
Solubilityand Temperature
A Solubility Curve
Introduction
Solubility,definedas the amount ofsolute that willdissolvein a givenamount of solvent,
dependson temperature. The solubilityof potassiumnitrate, for example,increasesfrom 14 g
in 100 g of waterat 0 °C to about 247 g in 100 g ofwater at 100°C—a1700%increase!While
these solubilityfactsare interesting,they do not allowus to predict the solubilityof potassi
urn nitrate at any other temperature. The temperature dependencefor the solubilityof a sub
stance can only be determinedby experim&nt,byconstructing a solubilitycurve.
Concepts
• Solubility
•
Saturated solution
• Saturation temperature
•
Solubilitycurve
Background
A solution that containsthe rnaximumamount ofsolutethat willdissolveat a particular tem
perature is calleda saturated solution.The onlypracticalwayto knowfor sure that a solution
is saturated iswhether or not there is undissolvedsolid present. Undissolvedsolute playsan
activerole in the saturated solution. For ar ionic compound, ions continuallybreak apart
from the undissolvedcrystal and enter the solution.At the same time, dissolvedions from the
solution also recombineto form new crystals.Whenthe solution is saturated, the rate at
which the soliddissolvesis exactlyequalto the rate at which solid recrystalizesfrom the solu
tion. As a result, the mass of dissolvedsolute in solution remains constant once the solution
is saturated—aslongas the temperature doesnot change. Sincethe solubilityof a substance
dependson temperature, the amount of dissolvedsolute present in a saturated solutionalso
dependson temperature. The solubilityof a solute is usuallyreported as the mass ofsolute in
grams that willdissolvein 100 grams ofsolventat a specifiedtemperature. The temperature
at which a saturatedsolution is prepared is calledthe saturation temperature.
In this experimentwe will prepare a series of solutions, each containing a premeasured
amount of potassium nitrate in a knownamount of water. The mixtureswillbe heated to
80—90°C until all the solid has dissolved.The solutions will then be cooleduntil the first
signs of crystal formation are observed.The temperature at which crystals first appear is the
saturation temperature for that concentration of potassium nitrate. The solubilitycurve for
potassium nitrate willbe generated by graphing the solubility of potassium nitrate versus
the saturation temperature for each solution.
Experiment Overview
The purpose of this experiment is to construct a solubilitycurve for potassium nitrate in
water by measuring saturation temperatures for six differentsolution concentrations.
Workingin groups of four, each pair of students will prepare three differentsolutions and
measure their corresponding saturation temperatures. The solubilityof potassium nitrate in
each solution willbe calculatedand plotted against the saturation temperature to construct
the solubilitycurve for potassium nitrate in waler.
Solubilityand Temperature
Solubilityand Temperature Page2
—
Pre-Lab Questions
1. Manysolutes, includingpotassiumnitrate, have a tendencyto remain in solution even
after it has been cooledto belowthe saturation point This phenomenon is known as
supersaturation. Readthe entireProceduresectioncarefully.What measure is taken to
prevent supersaturationduring this experiment?
in 5.0 g ofwater was heated
2. Amixture containing 2.75 g ofammonium chloride (NH
4CI)
to dissolvethe solidand then allowedto coolin air.At61 °C, the first crystalsappeared in
solution.What is the solubilityof ammonium chloride (in g of NH
4CI
per 100 g of water)
61
°C?
at
3. The solubilityof ammoniumchloridein water was measuredas describedin this experi
ment and graphedas follows.Usethe solubilitycurve to predict the solubilityof ammoni
urn chloride in water at 40 °C.
CI
4
SolubilityCurvefor NH
90.--p
60 ____________________
(50
20
0
10
0
0
10
20
30
40
50
60
70
80
90
100
°C
Temperature,
4. One of the students doing the experimentonly had time to measure the saturation tem
peratures for three solutionsat 51 °C, 61 °C, and 80 °C,respectively.Lookingat the graph
above,do you think the student wouldhavebeen ableto accuratelypredict the shape of
the solubilitycurve based on these three points?Explain.
Materials
about 3 g
Potassiumnitrate, KNO
3,
Distilledor deionizedwater
Balance,centigram precision
Gloves,heat-resistant, or Heat protector
Hot plate
Spatula
Beakers,50- and 250-rnL
Beral-typepipet, thin stem
Test tubes, small, 3
Testtube clampor holder
Thermometer,digital
Waxpencil or labeling pen
Flinn Chemlopic’Labs
—
SolubilityandSolutions
Page3- Solubilityand Temperature
•/Sj
Safety Precautions
Potassium nitrate in solidform is a strong oxidant and a lire and explosionrisk when heat
ed or in contact with organicmaterials.It is also a skin irritant.AvoidcontacZwith skin
and eyes. Wearchemicalsplashgogglesand chemical-resistantglovesand apron. Usecau
tion when working with a hot water bath and a hot plate. Wearheat-resistant gloves or a
d water
heat protector when handling hot glassware. Washhands thoroughly with soap th
before leaving the laboratory.
Procedure
1. Form a workinggroup with three other students and divideinto two pairs. Each pair of
and measure their
students will preparethree differentsolution concentrations (A—C)
saturation temperatures.One pair ofstudents will use the SeriesI massesshown in the
reagents tablein step 4, the other pair of students will use the SeriesII masses. Both pairs
of students may share the same hot water bath (step2).
2. Preparea hot waterbath (80—90
°C)for use in step 10:Fill a 250-mLbeakerabout twothirds fullwith hot tap water and place it ona hot plate at a medium-highsetting.
. record the
3. Obtainthree cleanand dry test tubes and labelthem A,B, and C.Measureand
of
each
table.
tube
in
the
data
test
mass
empty
4. Usingthe followingtable as a guide, add the recommendedamount of potassium nitrate
Notethat the massesgivenare ranges—anywherein this mass
to each test tubeA—C.
range is fine,as long the exact mass used in step 5 is recorded.
A
B
C
Mass of KNO
g (Series 1)
,
3
0.45—0.50
g
0.70—0.80
g
1.20—1.30
g
Mass of KNO
,
3
g (Series 11)
0.35—0.40
g
0.90—1.00
g
1.50—1.60g
Test tube
5. Measureand recordthe combinedmass of each test tube and.potassiumnitrate.
6. Usea clean, thin-stem pipet to add20 drops ofdistilledwater to each test tube A—C.
7. Place a 50-mLbeaker on the balance pan to support the test tubes. Zero (tare) the balance
with the emptybeakerin place,then placetest tube Ain the beakerand measure the
mass of the test tube and its contents. Recordthe total mass ofthe test tube plus solid
pluswater in the data table.Note: The mass ofwater in each test tube shouldbe at least
0.90 g. If not, add one or two more drops ofwater to the tube and measure the total mass
again.
8. Repeatstep 7 fortest tubes B and C.
9. Placeall ofthe labeledtest tubes in the hot water bath.
10. Immerse the thermometer in test tube C. Gentlystir the mixture in test tube C using an
up-and-downmotion of the thermometer until the soliddissolvescompletely.Note: At
this point the solidsin the other test tubes should also have dissolved,evenwithout stir
ring, since they contain lesssolid.
Solubilityand Temperature
Solubilityand Temperature Page4
-
/j
11. Wearingg!ovesor usinga heat protector, rernpvethe hot waterbath from the leat
source.
12. Usea test tube clampto removetest tube C irom the hot water bath and allowthe tube
and contents to cool slowlyin air. Observethe solution closelyto watch for the first signs
of crystallization.
13. Movethe thermometer gently up and downto stir the solution, make sure it is homoge
neous, and ensure constant, evencooling.Stirring the solutionwill also encourage crys
tal formation and preventsupersaturation.
14. Measurearidrecord the temperature the instant crystallizationbegins in the test tube
(this is the saturation temperature).Note: Th crystalswillbe colorlessin a colorless
solution and willriotbe easyto see. Watch closely—itwilllooklikesnow!
15. Removethe thermometer fromthe test tube and wipe it oncewith a clean paper towelto
removepotassium nitrate crystals.Warmthe thermometer brieflyin the hot water bath,
dry it with a paper towel,and placeit in test tube B.
• 16. Repeatsteps 12—15
with test tubes B and A,in that order.Rememberto record the satu
ration temperature or eachsolution in the data tableand to clean the thermometer
before transferringit to a new solution.
17. Share the data for the SeriesI and SeriesII solutions betweenboth pairs of students in
your working group. Completethe data table for all solutions (Series I and Series II).
18. Disposeof the potassiumnitrate solutions as directed by your instructor.
Flinn ChemTopicLabs SolubilityandSolutions
—
Page5- Solubilityand Temperature
I
ne:
Na
Class/LabPeriod:
•Solubilityand Temperaturf
Data Table
Massof test
tubeplus KNO
3
pluswater
.
Massof test tube
plus KNO
3
Massof empty
test tube
Sample
,
A
Series!
Saturation
temperature
‘,
,
B
•c
A
Series!!
.
•
‘
,
B
C
,
Post-Lab Calculations and Analysis
(Usea separate sheet of paper to answer the followingquestions.)
Construct a Results Tableto summarize the results of the followingcalculationsfor all solu
tions (SeriesI and Series II).
1. Calculatethe mass of potassium nitrate and the mass ofwater in each solution.
2. Calculatethe ratioof the massopotassium nitrate to the mass ofwaterfor each solution.
and 1.06 g H
Example:For 0.47g KNO
2
20, the mass ratio is equalto 0.47 g/1.06 g or 0.44.
3. Multiplythe mass ratio by 100to determine the concentrationof each saturated solution
in grams of potassium nitrate per 100 grams ofwater.
4. Plot a graph of solubilityof potassiumnitrate (in g ofsolute/100g ofwater)on the y-axis
versustemperatureon the x-axis.Scaleeachaxisas necessary.Drawa smooth, best-fit
curvedline though the data points. Don’tforgetto labeleachaxisand givethe graph a title!
5. Usingyour graph,estimate the solubilityof potassium nitrate in water at (a) 0 °C;
(b) 50 °C;and (c) 100 °C.
6. Usingyour graph,predict the temperature at which each of the followingmixtures of
in 25 g H
potassium nitrate in water wouldform a saturated solution: (a)25 g KNO
3
20;
the
in
250
Convertthe
concentrationsto
units
for
Hint:
(b) 100 g KNO
gH
3
20.
proper
solubilitybeforereferring to the graph.
7. Definethe terms saturated, unsaturated, and supersaturated as they applyto solutions.
Usecompletesentences.
Solubilityand Temperature
Solubility and Temperature
-
Page6
/5g.
8. Basedon your graph, classifyeach of the followingsolutionsas either unsaturated or
supersaturated at the indicatedtemperature. Assumethat the solutions do not contain
in 100g H
in 50 g H
20 at 40 °C;(b) 60 g of KNO
3
3
20
any undissolvedsolid.(a)75 g KNO
°C.
at 80
Explainyour reasoning.
9. Some of the water may have evaporatedfromthe test tubes beforetheir saturation tern
peratures were measured.Whateffectwouldthis error have on the solubilityof potassi
urn nitrate for a solution?Wouldthe correspondingsaturation temperature be too high
or too lowas a result of this error?
10. Allthermometers have a lag time—it takes a little whileto register or report a tempera
ture change. Whateffectwouldthis error have on the solubilityof potassium nitrate for a
solution?Wouldthe correspondingsaturation temperature be too high or too low as a
result of this error?
Flinn ChemTopic’Labs SolubilityandSolutions
—
Page1 CommonGases
-
Common Gases
Physicaland ChemicalProperties
Introduction
id odorless,
It’s easyto overlookthe chemistry of gases—becausemany gases are colorlessai
we may not noticethem. If we reflect on the environmental impact of differentgases in the
atmosphere,however,we realizethat not all gasesare the same. Just likesolidsand liquids,
all gases havecharacteristic physicaland chemical properties. Let’slook at the properties of
some commongases.
Concepts
Physicalproperty
•
Chemicalproperty
Background
Pure substances,whether solid or liquidor gas, have a constant compositionor chemical
and gaseous carbon dioxide(C0
makeup. Solidcoppermetal (Cu),liquidwater (H
are
20),
2)
examplesof pure substances.Pure substanceshavecharacteristicphysicaland chemical prop
erties that can be usedto describeand analyzethem. A physicalproperty is a characteristicof
matter that can be observedor measuredwithout changing the chemical composition.
Examplesof physicalproperties include color,odor;physicalstate (at rqom ‘temperature),
conductivity,meltingpoint, boilingpoint, and solubility.A chemical property describesthe
abilityof a substanceto undergo changes in its chemical composition.Examplesof chemical
properties includeflammability,acidity,and corrosion.
Experiment Overview
The purpose of this experiment is to prepare fivecommon gases and observetheir physical
and chemical properties.
Pre-Lab Questions
1. Readthe entireProcedureand the accompanyingSafety Precautions.Whathazards are
associatedwith the use of nitric acid?
2. Whatis the proper procedure for smellinga chemicalin the lab?
Enter the
3. Completethe followingbalancedequations for the reactions in test tubes A—E.
name and formulaof each gas in the data table.
(a) NaHCO
3(aq)
+
HCI (aq)
(b) Cu(s) + 4HN0
3(aq)
(c) NH
4CI(aq)
+
—‘
NaOH(aq)
NaCl(aq) + H
20(l)
—
3)2(aq)
Cu(N0
+
20(l)
2H
NaCl(aq) + H
20(l)
—
(g)
+
+
+
2
(g)
(g)
+
(d) 2H
(g)
20(aq)
20(l)
2H
Note: The catalyst (Mn0
is not represented in the balancedchemical equation.
2)
—
______
(e) Mg(s)+ 2HCI(aq)
CommonGases
—‘
2(aq)
MgCI
+
(g)
CommonGases Page2
-
/54
Materials
:
0.1 g
Ammoniurnchloride, NH
4CI,
•Copper foil,Cu, 5-mm square H
Hydrogenperoxide,l12O2, 3%, 5 mL
• Hydrochloricacid, HCI,3 M, 7 mL
Magnesiumribbon,Mg,2-cm strip
0.1 g
Manganesedioxide,Mn0
2,
Nitric acid, HNO
6 M, 1 mL
3,
Sodium bicarbonate solution, Nal-1C0
0.1 M, 2 mL
3,
Sodium hydroxidesolution, NaOH,3 M, 1 mL
Water,distilled or deionized
Beaker,150-mL
Forceps
Graduatedcylinder, 10-h
L
Litmus paper, 1 piece
Matches
Parafilm®,3-cm square piece
Spatula
Stirring rod
Test tubes, medium, 5
Testtube rack
Washbottle
Woodsplints, 3
Safety Precautions
Nitric acid is severely corrosiveand a strong oxidizing agent. Workwith nitric acid in a
fume hood only and do not remove from the hood. Hydrochloricacid is toxic by ingestion
or inhalation and is cbrrosiveto skin and eyes. Sodium hydroxide is a corrosiveliquid and
is especiallydangerousto the eyei Notify your teacher and clean up all spills immediately.
Hydrogenperoxide is a skin and eye irritant.Avoid contact of all chemicals with eyes and
skin. Copperfoil edgesare sharp and may cause cuts; handle with care or use forceps. Wear
chemical splash goggles and chemical-resistan/gloves and apron. Washhands thoroughly
with soap and water before leaving the lab.Neverspiff any substance in the chemical labo
ratory—to detect the odor of a substance,place th open container about 6 inches away
vard the nose.
from the nose and use your hand to waft the vapors to
Procedure
and place them in a test tube rack.
1. Labelfivemedium test tubes A—E
2. Pour 2 mL of 0.1 Msodium bicarbonateinto test tube A.
3. Add2 mL of 3 M hydrochloricacid into test tube Aand observethe color and odor of the
gas. Recordthe observationsin the data table.Note: If the gas has no color or odor,write
colorlessor odorless,respectively.
4. Lighta wooden splint and insert the burning splint welldowninto the test tube (but NOT
into the liquid). Recordthe observationsin the data table.
• 5. Bringtest tube B to the fume hood and carefullyadd 1 mL of nitric acid to the test tube.
6. Placetest tube B in a rack in the fume hood and add one small piece of copper foil.
Observeand record the color of the gas in the data table. Caution: DoNOTattempt to
smellthe odor of the gas! Leavethe test tube in the fume hood.
7. Adda small amount (abouta spatula-full,approximately0.1.) of solid ammonium chlo
ride to test tube C, followedby 5 mL ofwater.Stir to dissolve the solid.
8. Placethe test tube in a 150-mLbeaker containiii ahout 75 niL of hot tap water.
Ftinn CIiemTopic Labs
—
ChemistryofGases
Page3- CommonGases
9. Add10 drops of3 Msodium hydroxideto test tube C. Gentlyswirlthe test tube to mixthe
contents and cautiouslyobservethe odor. Recordthe colorand odor ofthe gas in the data
table.Caution:Toobservethe odor,hold the test tube about 6—8inches awayfrom the
nose and use your hand to waftthe vapors towardyou. DoNOT“sniff”the odor of any
chemicalin the laboratory.
10. Moistena strip of litmus paperwith a drop ofdistilledwater and place the moistenedlit
mus paper on top oftest tube C.Recordthe colorof the litmus paper in the data table.
11. Pour 5 mLof3% hydrogenperoxidesolution into test tube D.
12. Adda small amount (about the sizeof a grain of rice) of manganesedioxideto test tube D
and gently swirlthe test tube to initiate the reaction.Observeand recordthe color and
odor ofthe gas in the data table.
13. Sealthe mouth oftest tube D bystretching a piece of Parafilm®over the top ofthe tube.
Allowthe test tube to sit undisturbedfor 1—2minutes.
14. Preparea burning wood splint.
15. Removethe Parafilmfrom the test tube. Quicklyblowout the splint so that’it is glowing
rather than burning. Insert the glowingsplint downinto the test tube a’mostto the liq
uid, then bring it out. Recordyour observationsin the data table.
16. Carefullyadd5 mLof 3 M hydrochloricacid to test tube E.
17. Strike a match, carfullyadd one piece of magnesium ribbon into the acid in test tube E,
and quicklyplacethe lighted match directlyabovethe mouth ofthe test tube. Record
your observations,including the color and odorof the gas, in the data table.
18. Disposeof the contents oftest tubes A—E
as directed by your instructor.
CommonGases
CommonGases Page4
-
/&
•
Name:
__________
Class/LabPeriod:
-
CommonGases
DataTable
Physicaland ChemicalProperties of CommonGases
Name and Formula of Gas
,
Color and Odor
Test TubeA
.
Burning Splint Test
‘
•
Name and Formula of Gas
TestlubeB
,
Color
Nameand Formula of Gas
•
Color and Odor
Test Tube C
Litmus Test
Nameand Formula of Gas
Colorand Odor
Test Tube D
GlowingSplint Test
•
Test Tube E
,
Nameand Formula of Gas
Colorand Odor
MatchTest
Flinn ChemTopic Labs
—
Chemistryof Gases
Page5- CommonGases
Post-Lab Questions
1. Identifythe commongas or gases prepared n this experiment:
(a) Contributesto industrial smog and air pollution?
_______________
(b) Lightestelement in the universe?
_________________
(c) Neededfor the burning of fossilfuelsand for respiration in animals?
____________
(d) Dissolvesreadilyin water and is used as a fertilizer?
(e) Combustible?
_______________
(0
Extinguishesa flame?
_______________
(g) Has an odor?
_______________
(h) Requiredfor photosynthesis?
2. Explainthe observationsof the glowingsplint test for oxygen.
3. Explainthe observationof the litmus test forammonia.
4. Circleand labelthe physicaland chemical properties in the followingdescription of
chlorine:
“Chlorineis a greenish-yellowgas that dissolvesin water and is toxic to humans. It
combines violently with sodium metal to form sodium chloride,a white solid that melts
at 800 °C.”
5. Consult a PeriodicTable: Name the elements that exist as gases at room temperature
and give their symbolsor formulas.Hint: Recallthat some elements exist as diatomic
moleculesin their free state.
6. (a)Which gaseouselements (seeQuestion#5) are toxic? (b) Which gaseous elements are
consideredinert or unreactive?
CommonGases
/.
Name:
Chemistry Lab: Bayport- Blue Point HS
0
A Heated Discussion
Analyzing (lie Value and Uses of Different Temperature Scales
July 8, 2003
?
Hot Enough for You? Scientists Ask Fast Enough for You
BVKENNETHCHANG
Today, temperatures in New York City are expected to be in the 90’s, and
most New Yorkers will think of the temperature as a warm, soggy,
sensation on the skin.
Physicists have a different notion. To them, temperature is, in a simple
sense, speed. “It’ssome measure of the random motion of the various
degrees of freedom,” said Dr. Robert J. Schoelkopf, a professor of applied
physics at Yale.
For example. molecules of air or water bounce into one another like the
Ping-Pong balls in the machine that picks lottery numbers. The collisions
transfer energy between the molecules, producing a bell-curve distribution
of speeds, and the average kinetic energy of the molecules corresponds to
temperature. While the speeds of individual molecules constantly change,
the distribution of speeds remains the same, determined by the
temperature. The higher the temperature, the faster, on average, the
molecules jostle into one anoiher at cooler temperatures, the molecules
uidlv.
move more lan
While scientists have a precise definition of temperature. measuring it
precisely is an art still being refined.
Most thermometers, including the familiar mercury thermometer invented
by Daniel Gabriel Fahrenheit in the early 1700’s.have been based on the
fact that the jostling of molecules causes gases and liquids to expand
when heated. Technological innovations have led to new types of
thermometers, including those that use temperature-dependent electrical
resistors or measure infrared light radiating from warm bodies. At the
frontiers of science, scientists have devised techniques to take the
temperatures of ultracold atoms, surfaces of distant stars and trillion-
0
/13.
degree firestorms of colliding atoms.
Biologists have also only begun to understand how livingcreatures
measure and regulate temperature.
Dr. Schoelkopf and colleagues at Yale have invented a thermometer that
measures temperature based on electrical noise not that different from the
static heard on radios. (Radio static is itself asort:of temperature
measurement Radiation left over by the Big Bang has cooled to a few
degrees above absolute zero in the past 13 or so billion years, suffusing
the universe in a bath of microwaves that can be picked up by microwave
receivers and heard as a static hiss.)
The new thermOmeter,.clçscribedlast month in thejoumal Science,
consists oftwo pieces of metal on a silicotichip separated by athin strip
of insulator. Under the strange rules of quantum mechanics, electrons can
occasionallyhop back and forth across the gap
a low hiss of electrical
iioise. With rising temperatures, electrons bounce off one another and the
atoms in the metal with more momentum, producing more electrical
nbise. The loudness of the noise thus giyes the temperature.
—
That idea is not new but until now such thermometers have needed to be
carefully calibrated.
.
he innovation of Dr. Schoelkopfsgroup is to.apply a voltage that pushes
a number of electrons across the gap, regardless of the temperature. That
allows the thermometer to calibrate itself
“It has a certain simplicity, which is attractive,” said Dr. Wes Tew, a
physicist at the National Institute of Standards and Technology in
Gaithersburg. Md., who was not involved with the research.
The device’s simplicity and accuracy over a wide temperature range could
make it useful as a reference thermometer, especially at ultracold
temperatures.
The studs’of heat goes back at least as far back as the second century B.C.
when an engineer named Philo of Byzantium observed that gases expand
when heated and contract when cooled. Galileo Galilei is generally
credited as the first to turn this observation into a thermometer around
1600. But while early crude thermometers could measure relative rises
and falls in temperature, the lack of a universal temperature scale made it
difficult to compare readings of different thermometers.
Fahrenheit. the German physicist who invented the mercury thermometer,
devised the temperature scale still in use in the United States, setting the
/
freezing point of water at 32 degrees and the boilin point at 212 degrees.
Anders Celsius. a Swedish astronomer, set up a competing temperature
scale inreverse, putting th boiling point of water at zero degrees and the
freezing point at 100 degrees. Others later inverted the scale, putting the
freezing.point at zeroand the boiling point at 100.
Experiments showed that’under constant pressure,:any gas steadily shrinks
in volume as the temperature drops. in 1848, a Scottish chemist, William
Thomson, later Lord Kelvin, realized that extrapolating this trend, the
shrinking gas would disappear entirely at about minus-273 degrees
Celsius, and he proposed a new temperature scale that defined the new
zero as the temperature where the gases would disappear.
Because temperature is a measure of speed,.this coldest possible
temperature,’ known as absolute zero or minus-459.67 degrees on the
Fahrenheit scale, is where everything comes to almost a,complete stop.
Sdentists have since reflnec the temperature standard several times.
Because the boiling and freezing points of water vary with altitude, the
Kelvin scale is now set by the triple point of water
a precise
temperature and pressure where ice, liquid water and steam coexist. The
triple point is, by definition. 273.16 kelvin. “That’sth,e only temperature
we know,” Dr. Tew said. “Allthe other temperatures must be
experimentally determined.”
—
‘
‘
In the latest tweaking of the standard in 1990. water at one atmosphere’s
pressure no longer boils at exactly 100 degrees Celsius, but 99.974
degrees (or in Fahrenheit, the boiling point is no longer 212 degrees, but
211.95 degrees).
To help people calibr
thermometers, the standard offers 17 wellte
measured temperatures including the triple point of hydrogen (minus434.8 degrees Fahrenheit), the melting point of gallium (85.6 degrees
Fahrenheit) and the freezing point of copper (1,984 degrees Fahrenheit).
Dr. Schoelkopf has begun testing, his noise thermometer against some of
the standard temperatures. He believes that it should work for
about one-hundredth of a degree
temperatures ranging from very cold
above absolute zero
to room temperature, although accuracy tails off at
the upper end of that range. “We can try to see what the limits of our
thermometer are,” he said.
—
—
But the noise thermometer, or indeed any other thermometer. is useless
for experiments where physicists cool atoms to a few billionths of a
degree above absolute zero. Jr would be impossible to cool a thermometer
to the temperature it was trying to measure. “We’re talking really, really
/
cold.” said Dr. WilliamD. Phillips, a physicist at NIST w.h shared the
Nobel Piize in Physics in 1997 for developing techniques to cool atoms
with lasers.
Instead, physicists revert to the notion of temperature as speed. They turn
off the electromagnetic fields that trap the atoms and then a short time
later, shoot a flash of laser light to illuminatethe size of the expanding
cloud. “That will give you a good measure of what the velocities of the
atoms are,” Dr. Phillips said.
At the high end of the temperature spectrum. astronomers use a different
trick. Since they cannot stick thermometers into distant stars, they take
advantage of a property of matter: heated, it emits light. The distribution
of wavelengths of emitted light follow a characteristic bell curve, but the
peak shifts depending on the temperature. The peak wavelength of light
from the sun. for example, is green. which corresponds to,à surface
temperature of about 10,000 degrees Fahrenheit. The hiss from the cosmic
background microwaves tells the average ernperature in the universe:
above absolute zero.
minus-454.8 degrees Fahrenheit. a few degr
es
Ear thermometers that measure body temperature work by the same
concept. People also emit light, but at longer infrared wavelengths not
visible to the human eye. A sensor in the thermometer scans for the peak
wavelength of infrared light, which tells the temperature of the ear drum
area. The ear drum shares the same blood vessels as the hypothalamus, the
region of the brain that regulates body temperature.
No one yet knows how internal thermometers work, and only in the past
few years have biologists begun to understand how the body senses
external temperatures.
In 1997, researchers led b’ Dr. David J Julius. a professor of cellular and n
’
University of California at San Francisco. were the first to identi
a protein
cells. At temperatures above 108 degrees Fahrenheit. the protein, which thr
opens a pore to let in sodium, calcium and potassium ions. The ions charge
message of pain to the brain.
“Most mammals. certainly humans and primates. have a pretty welldefined threshold for where hot stimuli become noxious,” Dr. Julius said.
(The same protein responds to capsaicin. a molecule in chili peppers,
producing the fiery taste.) Three other proteins that detect various degrees
of warni and hot have since been found.
A couple of years ago, Dr. Julius isolated a protein that sends sensations
of cool at temperatures below 77 degrees Fahrenheit.
/&,
In March in the journal Cell,’Dr. Ardem Patapouti
n,
a professor of cell
biOlogyatthe Scripps Research institute in San Diego, reported finding a
protein receptor that detects painfullycold temp&atures below 59 degrees
Fahrenheit. Dr. Patapoutián’s group has spliced the gene that produces this
protein.in other types.of cells like hamster ovaries that then became
sensitive to cold.
In the Cell paper, Dr. Patapoutian quoted from a John Updike essay:
“Cold is an absence, an absence of heat, and.yet it feels like a presence.’
of his research
Dr. Patapoutian has been meaning to tell Mr. Updj
e
him
because
that’s
how it works,”
send
these
“I
have
to
findings.
papers
Dr. Patapoutian said.
For the living ceatures, cold is a separate sensation, although probably
not one that will be felt today.
ht
Copvri
2003 The N
York Times Company
/7
Name:
Chemistry Lab: Bayport-BJuePoint HS
A Heated Discussion
Analyzing tIle Value and Uses of Djfferent Temperature Scales
RelatedNew YorkTimesArticle
“HOTENO JGHFOR YOU?SCIENTISTSASK, FAST ENOUGHFOR YOU?”
By KENNETH CHANG, July 8, 2003
Objectives
Students will:
1. Explore the colloquial definition of temperature.
2. Learn about how scientists define temperature by reading’and discussing the article
You?ScientistsAsk, Fast Enough/or You?”
“Hotfo’r
Enough
3. In groups, measure how the phase of water changes with temperature; record and graph
their results and convert their findings between the three main temperature scales.
:
Resources/Materials
“Hot Enough for You? Scientists Ask, Fast Enough for You?” article
HOT PLATES
WATER
ICE
THERMOMETERS
BEAKERS
GOGGLES
ICE BATHS
HOT BATHS
I
CALCULATORS
Pre-Lab Discussion
What is the temperature today?
How does that compare with the temperature from yesterday?
How is temperature measured (with what instrument and in what units)?
Why is knowledge about temperature important?
.
I
Procedure
u?”
1. Read the article “Hot Enough for You? Sci
ntistsAsk,Fast
Enough for Y
focusing on the following questions which are to be answered on looseleaf:
How do scientists define temperature?
On what principle about gases and liquids is a thermometer based?
How does the newly invented thermometer measure temperature?
Why is the new thermometer useful?
What are some of the different temperature scales used around the world?
What is absolute zero, and how is it defined?
What is the triple point? Why is it important?
4
How do.ear thermometers work?
What are “internal thermometers”? How are scientists investigating how they
work?
j. How does the protein identified by Dr. David Julius regulate temperature inthe
organisin in which it is produced?
a.
b.
c.
d.
e.
f
g.
h.
i.
2. You will be addressing the following question in today’s lab experiment:
,
How does temperature affect the phase of water?
a. Working with your lab Group, perform the ativity that has been assigned to your
group based on your Group number.
b. In your experiments, you will need to record the temperature ofthewater (either
in the boiling state or the ice cube) inside the glass beaker.
c. To heat up the water, students will use a hot’plate. To cool down the water,
students will use an ke bath.
d. On the observation form. you will convert the temperatures you recorded in all 3
temperature scales (Celsius. Fahrenheit. and Kelvin).
3. You must complete the observation chart included in this lab packet. You will be
makin an observation AND recording the temperature every 30 seconds for 15 minutes.
4. After you have finished collecting your data (15 minutes) you will make a graph
of your data on graph paper. You must have axes labels, units and a TITLE!
Temp
(units)
You are to do
this on
GRAPH PAPER!
Time (minutes)
For all evennumberedgroups
You will beginWithan ice cube and slowly heat t on a
:
hot plate for 15 minutes. You will make observations about the temperature chan
and
e
the
hot
physical appearance on
plate.
You will begin with boiling water and slowly cod! it in
For all odd numberedgroups
:
an ice bath for 15 minutes. You will make observations abQut the temperature change
and physical appearance on the hot plate.
GROUP I will measure the temperature change of an ice cube experiencing the addition
of heat over time. Group members will measure data in degrees Celsius.
Materials needed: hot plate, thermometer, ice cube, and g!ass beaker.
GROUP 2 will measure the temperature change ofboiling water experiencing a loss of
heat over time.. Group mbers will measure data in degrees Celsius.
Materials needed: ice bath, thermometer, boiling water, and glass beaker.
GROUP3 will measure the temperature change of an ice cube experiencing the ad
tion
of heat over time. Group members will measure data in degrees Fahrenheit.
Materials needed: hot plate, thermometer, ice cube, and glass beaker.
GROUP 4 will measure the temperature change of boiling water experiencing a loss of
heat over time. Group members will measure data in degrees,Fahrenheit.
Materials needed: ice bath, thermometer, boiling wat’er,and glass beaker.
.
GROUP .5will measure the temperature change of an ic cube placed in a solution of salt
water experiencing the addition of heat over time. Group members will measure data in
degrees Celsius.
Materials needed: hot plate, thermometer, ice cube, salt and glass beaker.
GROUP 6 vill measure the temperature change of boiling salt-water solution
experiencing a loss of heat over time. Group members will measure data in degrees
Celsius.
Materials needed: ice bath. thermometer, boiling water, salt and glass beaker.
GROUP 7 will measure the tem
erature
change of an ice cube in a solution of salt water
experiencing ihe addition of heat over time. Group members will measure data in degrees
Fahrenheit.
Materials needed: hot plate, thermometer. ice cube, salt and glass beaker.
GROUP 8 will measure the temperature change of boiling salt-water solution
experiencing a loss of heat over time.
€roup members will measure data in degrees Fahrenheit.
Materials needed: ice bath, thermometer, boiling water, salt and glass beaker.
Post Lab Ôuestions
& Analysis
1. How does temperature
2. Answer
•,
the following
affect the phase of water
questions
discussing
how water reacts to temperature
changes:
a.
What happens to the molecules of water as temperature is increased or decrëase
?
b.
c.
What is the starting temperature of an ice cube
What starts to happen physically to the ice cubeast
mperature
d.
How
e.
Briefly define the following:
can the phase change between liquid and gas be observed?
Freezing
Boilin2
Cotidensailon
Phase change
Solid
Liquid
Gas
is increased?
3. Groups 5-8 performed the experiment using a salt-water solution. Use the
following chart to find the Group that you are to compare data with. Answer
following questions:
S
Groups I & 5 share data
Groups 3 & 7 share data
Groups 2 &.6 share data
Groups 4 & 8 share data
How will temperature affect the phase changes of a salt solution?
.
How does this compare with that of water?
Collect data and make a general conclusion of how boiling point elevation and free±ing
point depression are evidenced by your experiment. compared with the standard paints of
water.
i7&
e
Observation Form: How does temyerath
Group’#:
Temp Scale used:
ya1
Thser
Temp
(.°C)
(minuI
sL
0-0.5
Solution Type.:
Observations
Temp
(K)
Temp
(°F)
,
:
•
S
0.5-1
1-1.5.
1.5-2
.
••
,
2-2.5.
2.5-3
3-3.5
3.5-4
44.5
4..5-5
5-5.5
5.5-6
6-6.5
6.5-7
?
affect the phase of water
•‘
,
_______
.,
.
.
I
.
5
0
.
,
S
S
•
5
S
.
0
0
•
,
S
:
:
1_7•5
7
7.5-8
8-8.5
8.5-9
9-9.5
9.5-10
10-10.5
10.5-11
11-11.5
11.5-12
12-12.5
12.5-13
—
3-I
1
—.I
1
3.
13.5-14
14-14.5
14.5-15
0
.
0
,
0
0
.
0
__________
Name
Date
Class
I
Chemic
ExpIorin
nqes
Ch
Most reactions can be classified into five major types. As you carry
out this experiment, you’ll observe examples of each of these types.
In doing so, you will also learn to recognize many of the physical
changes that accompany reactions.
CHEMIRD
AND
ILQVWORHSHE
MI
CflEMIRII
6
EM
PR
QAL
PRO
C[0IIHE
What are some of the physical changes that
indicate that a reaction has occurred?
For each of the following reactions, record in
the data table all changes that you observe.
Synthesis Reaction
1. Place 50 mL 0.1M CuSO
in a 125-mL
4
flask.
2. Place 1.6 g granular copper and 0.8 g
powdered sulfur on a watch g’ass and
mix together thoroughly with a spatula.
3. Heat the flask on a hot plate set at high
until the solution begins to boil.
4. Stir the Ca/S mixture into the boiling
solution.
4
CuSO
5. Continue boiling until a black solid
forms.
Decomposition Reaction
1. Place 100 mL of saturated Ca(OH)
solu
2
tion (limewater) in the 250-mL flask.
2. Add finely ground CaCO
to a large test
3
tube until it is one-fourth full. Stopper
the tube with the stopper/glass tube/rub
ber tubing assembly, and clamp the tube
to the ring stand.
3. Light a laboratory burner, and begin to
heat the test tube. Submerge the end of
the rubber tubing into the limewater so
that any gas produced in the tube will
bubble through the limewater.
4. Continue heating the CaCO
until you
3
observe a change in the limewater. The
2
causes limewater to
presence of CO
become cloudy.
o0JECTIYES
• Observe physical changes that take place
during chemical reactions.
• Compare changes that take place during
different types of chemical reactions.
MATERIALS
C
0
a
0
C)
I
0
C
0
:2
0
C,
U
0
8
C
125-mL flasks (4)
balance
hot plate
watch glass
spatula
stirring rod
lab burner
file
new penny
250-mL flask
ice
tongs
100-mL graduated
cylinder
large test tube and
one-hole stopper
with glass tube and
rubber tubing
attached
ring stand
test-tube clamp
0.IM CuSO
4
granular copper, Cu
powdered sulfur, S
3,
CaCO
finely ground
saturated Ca(OH)
2
solution, limewater
GMHCI
0.5M 3Na
2CO
0.5M CuCI
2
0
C,
0
SAFETY
PRECAUTIONS
a
Wear an apron and goggles. Use care when
handling hot objects. Dispose of the reaction
mixture and products as instructed by your
teacher.
Chemistry:Concepts and Applications
ChemLab 6
41
S!ngle-Displacement Reaction
1. Place 30 mL 6M HC1 in 125-mL flask.
2. Using ,a file, cut six,0.2-cm notches
evenly spaced around .the perimeter of a
new penny.
3. Place the penny in the flask of acid and,
leave it in a fume hood overnight.
•
•Double-Displacement Reaction
1. Add 25 mL 0.5M 3Na
and 25 mL
2CO
0.5M CuC1
to a 125-mL flask.
2
2. Swirl the flask gently until you observe
the formation of a precipitate.
Combustion Reaction
1. Light a laboratory burner and adjust
the air and gas supplies until the flame
is blue. Observe what happeps.
2. Using tongs, hold a flask or beaker with
• ice in it about 10’cm over the flame for
• approximately one minute. Move the
flask away from th flame and observe
the bottom of the flask.
ANALYZE
ANO
CON.CLOOE
1. Making Inferences Which observa
tions noted during each of the reactions
indicated that a reaction had occurred?
3. Making Inferences Write the name
and formula of the
a) black solid formed in the synthesis
reaction.
b) gaseous product of the decom
osition
.
reactior
c) solid product of the decomposition
reaction.
d) pale blue precipitate in the doubledisplacement reaction.
e) liquid product of the combustion
reaction.
4. Observing and Inferring Explain
how the penny changed during the sin
gle-displacement reaction. What would
happen if a pre-1983 penny, which is
solid copper, were used?
0
0,
C
0,
E
0
0
I
0
,
a
0
C
0
0
>
(a
C.)
0)
8
C
a,
’
0
2. Comparing and Contrasting What
did all of the reactions have in common?
42
Chemistry: Concepts and Applications
5. Relating Concepts Is energy a
reactant or product of the combustion
reaction?
ChemLab6
>‘
0
0
ANO
ASSESS
APPLY
S
/73-;
Were there any physical changes that
often occur during a reaction that you
did not observe while doing this
ChemLab? If so, what were they?
2. Write balanced chemical equations for
all of the reactions carried out.
3. Why do you think pennies are no longer
made from only copper metal?
‘I
••
•
ANO
ODSEHYAIJONS
DATA
I
Observations
Reaction
Synthesis
.
Decomposition
S
displacement
Single
C)
Double
displacement
Combustion
Chemistry: ConceptsandApplications
ChemLab6
43
Name
Date ___________ Class
_________________________________
___________
Reactions Lab
Typesof Chemical
S
6.
/7
14
Text reference: Chapter9. pp. 215—217
Pre-LabDiscussion
There are many kinds of chemical reactions and several ways to classify
them. One useful method classifies reactions into four major types. These
are: (1) direct combination, or synthesis; (2) decomp9stion, or analysis;
(3) single replacement; and (4) exchange of ions, or double rep’acement.
Not all reactions can be put into one of these categories. Many, however,
can.
In a synthesis reaction, two or more substances (elements or cornpounds) combine to form a more complex substance. Equations for
synthesis reactions have the general form A + B —p AB. For example,
the formatioi of water from hydrogen and oxygen is written 2H
2 + 02
20.
2H
A decomposition reaction is the opposite of a synthesis reaction. In
decomposition, a compound breaks down into two or more simpler sub
stances (elements or compounds). Equations for decomposition reactions
A + B. The breakdown of water into its elements
have the form AB
is an example of such a reaction: 2H
20
2 + 02.
2H
tanc
in a compound is
In a single replacement reaction, one ub
replaced by another, more active, substance (an element). Equations for
single replacement reactions have two general forms. In reactions in
which one metal replaces another metal, the general equation is X + YB
XB + Y. In those in which one nonmetal replaces another nonmetal,
AX + Y. The following equations illustrate
the general form is X + AY
these types of reactions:
Zinc metal replaces copper(1l) ion:
—‘
—+
—
—
—
Zn(s)
+
4(aq)
CuSO
—*
+
4(aq)
ZnSO
Cu(s)
Chlorine (a nonmetal) replaces bromide ions:
2(g)
C1
+
2XBr(wj)
—÷
+
I(aq)
2KC
9(t)
Br
In a double replacement reaction, the metal ions of two different ionic
compounds can be thought of as “replacing one another.” Equations for
AD + GB. Most
this type of reaction have the general form AB + CD
replacement reactions, both single and double, take place in aqueous solu
tions containing free ions. In a double replacement reaction, one of the
products is a precipitate, an insoluble gas, or water. An example is the
reaction between silver nitrate and sodium chloride in which the precipi
tate silver chloride is formed:
—
3(aq)
AgNO
•
+
NaC1(aq)
—
AgCI(s)
+
3(aq)
NaNO
All of the types of reactions discussed here may be represented by
balanced molecular equations. Reactions involving ion exchanges may be
represented by ionic equations also. In this investigation you will be con
cerned only with molecular formulas and equations. In a balanced equa
69
tion, the number of atoms of any given element must be the same on both
sides of the, equation. Multiplying the coefficient and the subscript of an
element must yield thesame result on both sides of the bdanced equation.
In this investigation you will observe examples of the four types of
reactions described above. You will be expected to balar
the equations
ce
representing the observed reactions.
Purpose
Observe some chemical reactidns and identify reactants and products of
those reactions. Classify the reactions and write balanced equations.
Equipment
burner
crucible tongs
microspatula
test tubes, 15x 180-mm (7)
test tube holder
test tube rack
‘
‘
wood splints
sandpaper, fine
evaporating dish
safety goggles
lab apron or coat
Materials
zinc, mossy (Zn)
e,
10 cm (Cu)
copper wii
magnesium ribbon, 5 cm (Mg)
copper(II) carbonate (CuCO
3)
6 M hydrochloric acid (FICI)
4)
1M copper(II) sulfate (CuSO
0.lM zinc acetate (Zn(C
30)
2H
0.1 M sodium phosphate 4)(Na
3PO
1M sodium sulfite (Na
3)
SO
Safety
In this investigation ‘ou will be working with open flames, heating chem
icals, handling acids, and producing gaseous products. You should review
the safety procedures for these activities given on pages ix—x.
Burning magnesium produces a very bright, hot flame. Make sure you
hold the burning metal at arm’s length and do not look directly at it.
Remember never to smell a chemical directly. Review the accepted
method of wafting gases toward your nose as illustrated on page xi.
Pay special attention to the safety symbols beside certain steps in the
procedure. Refer to page xi to review the special precautions associated
with each symbol.
Wear safety goggles and protective clothing at all times when working
in the lab.
Procedure
PART A SYNTHESIS
1. Use fine sandpaper to clean a piece of copper wire until the
wire is shiny. Note the appearance of the wire.
2. Using crucible tongs, hold the wire in the hottest part of a
burner flame for 1-.2 minutes. Examine the wire and note any
change in its appearance caused by heating.
3. Place an evaporating dish near the base of the burner.
Examine a piece of magnesium ribbon. Using crucible tongs,
70
Name
.
14
.
1
/7rP
Reactions(continued)
Typesof Chemical
hold the sample in the burner flame until the magnesium starts
to burn. DO NOT LOOK DIRECTLYAT THE FLAME.HOLD THE
BURNING MAGNESIUM AWAY FROM YOU AND DIRECTLY
OVEI THE EVAPORATING DISH. When the ribbon stops burn
ing, put the remains in the evaporating dish. Examine this prod
uct carefully.
PART B DECOMPOSITION
4. Place 2 heaping microspatulas of copper(II) carbonate
3)(CuCO in a clean, dry test tube. Note the appearance of the
sample.
3
O
5. Using a test tube holder, heat the Cu
strongly for about
3 minutes. E
ctinguish the flame and then insert a burning wood
is present,
2)
splint into the test tube. If carbon dioxide gas (CO
it will put the flame out. Note any change in the appearance of
the residue in the test tube.
PARTC SINGLEREPLACEMENT
6. Stand a clean, dry test tube in the test tube rack. Add about
5 mL of 6 M hydrochloric acid (HCI) to the tube. CAUTION.
Handle acids with care. They can cause painful burns. Do not
inhale any JICI fumes. Now carefully drop a small piece of zinc
metal (Zn) into the acid i’n the test tube. Observe and record
what happens.
7. Using a test tube holder, invert a second test tube over the
mouth of the test tube in which the reaction is taking place. See
Remove the inverted tube after abQut 30 seconds
1.
Figure l4
insert
and quickly
a burning wood splint into the mouth of the
the presence of hydrogen gas.) Note the
indicates
tube. (A “pop”
of
the
substance
in the reaction test tube.
appearance
I..
Figure 14-1
.
solution
8. Add about 5 mL of I M copper(ll) sulfate (CuSO
4)
in
of
zinc
metal
Place
small
tube.
amount
a
to a clean, dry test
the solution. Note the appearance of the solution and the zinc
before and after the reaction.
71
PARTD DOUBLEREPLACEMENT
9. Add about 2 mL of 0.1 M zinc acetate 2H
)(n(C
3O
to a
add
about
tube.
2
mL
of
0.1
M
test
sodium
Next,
clean, dry
solution to the test tube. Observe
phosphate tribasic (Na
4)
PO
what happens and note any changes in the mixture.
10. Add about 5 mL of 1 M sodium sulfite 3)(Wa
solution to
2SO
a clean, dry test tube. To this solution, add about 1 mL of 6 M
HCI. Note the odor given off by wafting some of the gas toward
your nose. DO NOT SMELL THE GAS DIRECTLY.
0
Observations and Data
.
DATATABLE
Before reaction
Sample
A.
Synthesis
After reaction
,
.
1.Cu
2.Mg
.
B. Decomposition
3. CuCO
3
.
C. Single Replacement
4.Zn+HCI
5. Zn
+
.
.
4
Cu50
0. Double Replacement
6. Zn(C
)
0
3
H
2
7. Na
3
SO
72
+
+HCI
.,
PO
3
Na
4
.
Name
S
14 Typesof ChemicalReactions(continued)
Equatiohs
0
Balance each of the equations by inserting the proper coefficients where
needed. Write the’•names of the reactant(s) and product(s) below the
molecular equation for each reaction.
PARTA SYNTHESIS
1.
Cu(s)
+
02(9)
—
CuO(s)
2.
Mg(s)
-
02(g)
—p
MgO(s)
PARTB DECOMPOSITION
3.
(s)
3
CuCO
CuO(s)
—
+
2(g)
C0
PARTC SINGLEREPLACEMENT
4.
Zn(s)
+
I-ICJ(aq)
5.
Zn(s)
+
4(aq).
CuSO
+
2(aq)
ZhCI
—p
—
4(aq)
ZnSO
2(g)
H
,+
I
Cu(s)
PARTD DOUBLEREPLACEMENT
6.
30)(aq)
2H
Zn(C
7.
2Saq)
3(Na
O
+
+
3Paq)
4(Na
O
HC1(aq)
-
—i
(’iq)
3O
2H
NaC
NaCI(aq)
+
+
20(1)
H
+
3s)
4)2(Zn
P0
2(g)
S0
I
Conclusionsand Questions
1. In this experiment, what method was used to test for the presence of
for CO
2
gas? Write a balanced equation for
2
CO
gas? What is another te
t
this test.
S
73
/1
•
•
:
2. What test was used to identify hydrogen gas? Write a balanced equation
to represent this test.
,H
H
3. Balance the equations below and identify the type f reaction rep
resented by each equation.
+
+
a. AgNO
3(aq)
Cu(s)
3)2(aq)
Cu(N0
Ag(s)
b. BaC1
+
+
4()
BaSO
2(aq)
2Saq)
4(Na
O
NaC1(aq)
+ NaBr(aq)
+ Br
c. C1
2(g)
I(áq)
NaC
2(1)
—p
d. KCIO
+
(g)
2
0
3(s)
lçs)
K
I
—
.
—‘
.—
e. AIC1
3(aq)
f. H
(g)
2
+
+
2(g)
Q
4OH(aq)
NH
—p
t
74
—
20(g)
H
I(aq)
4C
NH
+
3(s)
Al(QH)
Page1 Preparingand TestingHydrogen Gas
•
-
’,
/k
Preparing and TestingHydrogen Gas
A MicroscaleApproach
Introduction
Hydrogenis the most abundant element in the universeand the second most abLrndantele
ment in livingthings. Despiteits abundance,very little hydrogen is found on Earth in the
‘free state as hydrogengas. This is because hydrogen is easilyoxidizedand combines readily
with many other elements,includingcarbon, oxygen,and nitrogen. How can hydrogen gas
‘be prepared in the lab?
Concepts
• Hydrogengas
•
Physicalproperties
• Chemicalproperties
•
Flammability
Background
Hydrogengas was first studied in 1766by Henry Cavendish,who isolatedthe “inflammable
air” produced in the reaction of metals with acids.The true chemical nature o this gas was
not understood until about 20 years later, when Lavoisiercorrectly explainedthe reaction
that occurs when the gas combineswith oxygentQmake water. Lavoisiernamed the gas
“hydrogen”from the Greekwords meaning “water-former.”Mostof the hydrogen on Earth
is found in water and in petroleum-basedproducts (oil and gas). Hydrogen gas is also the
most abundant element in the universe—itis the “fuel”that makes our sun and stars burn
brightly.
Experiment Overview
The purpose of this experiment is to collect hydrogen gas bywater displacementand study
its properties. Hydrogengas will be generated by the reaction of zinc metal with hydrochlo
ric acid.
Pre-Lab Questions
1. Readthe Procedure section and the accompanyingSafely Precautions. Whathazards are
associatedwith the use of hydrochloricacid?
2. Write a balancedchemical equation for the reaction ofzinc metal with hydrochloricacid.
3. What are the twomajor gases in air? Which one will probablyreact with hydrogen in the
“TestforIVlixtureofAir and HydrogenGas?”Whatis the likelyproduct of this reaction?
Materials
Hydrochloricacid, HC!,3 IVI,20 mL
Zinc,granular, Zn, 1 g
Soapsolution, 2 mL
Barstraw
iJ-Iype pipet bulbs, 2
J3cr
cn
PreparingandTesling Ilvdru
Gas
Bunsen burner
Gas-deliverystopper, size 00
Plastic cup, 10-oz
Test tube, small
Toothpicks,wood,3
Preparing and TestingHydrogenGas Page2
-
/15.
Safety Precautions
Zinc dust may be flammable. Hydrochloricacid is toxic by ingestion and inhalation and is
ills immedi
severely corrosiveto skin and eyes. Notify your teacherand clean up all acid sj
is
flammable.Avoid.contactof
all
chemicalswith eyes and,skin. Wear
ately. Hydrogengas
chemical splashgoggles and chemical-resistantgloves and apron. Washhands thoroughly
with soap and water beforeleaving the laboratory.
Procedure
1. Placeabout 0.5 grams of granular zinc into a
small test tube and insert the gas-deliverystop
per (seeFigure 1).
Gas-DeliveryStopper
2. Fill a pipet bulb with water so that the hydrdgen
by the displacementofwater.
cted
gas can be coil
4
3. Set up the gas generatoras shown in Figure 2.
To generate the hydrogengas,remove the gas
ery
deli
stopper,fillthe test tube about 4/5 full
with 3 M hydrochloricacid, and replace the stop
per. Thereshouldbe only.a smallair space
betweenthe acidand the bottom of the stopper..
Thiswill reducethe time neededto.flush the air
out of the gas generator.
4. Placethe pipet-buibfull of water on top of the
gas-deliverystopper to collectthe hydrogen gas
by water displacement(seeFigure 2).
5. Collecta pipet-buibfull of the gas.
4
HydrochloricAcid
Zinc
Figure 1.
Pipet
Bulb
unit
Gas-generating
Gas-DeliveryStopper
/
Testfor Pure Hydrogen Gas
6. Totest for pure hydrogengas, bring a pipet-bulb
fullof the gas to a Bunsen burner, mouth down.
Holdingthe pipet bulb horizontally, placea
flamingtoothpicknear the mouth of the pipet
bulb and quicklysqueeze the bulb of gas. Record
your observationsin the data table.
PipetBulbs
Figure 2. Collecting the gas
Testfor Mixture of Air and Hydrogen Gas
7. Fill a pipet bulb about ¼ full ofwater. Collect enough hydrogen gas to displacethe water
in the bulb.
8. Totest the hydrogen—air
mixture,bring the pipet bulb to a Bunsen burner, mouth down.
Holdingthe pipet bulb horizontally,place a flamingtoothpick near the mouth of the
pipet bulb and quicklysqueezethe bulb of gas. Recordyour observationsin the data table.
Flinn ChemTopic”
Labs
—
Chemistry of Gases
Page3- Preparingand TestingHydrogen Gas
Relative Density of Hydrogen Gas
Soap
9. Insert a bar straw so that it telescopesinto the gas-delivery
stopper of thegas-generatorurlit (seeFigure3).
10. Whilethe hydrogengas is beinggenerated,pla
ce a filmof
solution
the
ofthe straw
mouth
soap
(usingyour finger)at
so that a bubbleof hydrogengas can be formed.
11. Whenthe bubbleis approximately1 cm in diameter,raise
the apparatusjust abovepour headand gently blowupward
at the bubblein order to jar the bubbleoose without
breakingit. Recordyour observationsin the data table.
12 Disposeof the test tube contents as directedbyyóur
Instructor.
PreparingandTestingHydrogenGas
______
I
BarStraw
Gas-Delivery
Stopper
Zinc
Figure 3.
Preparing and TestingHydrogenGas Page4
-
Name:
Class/LabPeriod:
Preparing and TestingHydrogen Gas
Data Table
I
Testfor Pure HydrogenGas
•0
TestforMixtureof Air and HydrogenGas
RelativeDensityof HydrogenGas
Post-Lab Questions
1. Is hydrogengas lighter or heavierthan air? Explainbasedon your observations.
2. Comparethe observationsmade when pure hydrogenwas tested versus when a mixture of
hydrogenand air was tested.
(a) Whichreaction was more explosive?Why?
(b) Writea balancedchemical equation for the reactionof hydrogenand oxygen.
Flinn Chemlopic”Labs
—
Chemistryof Gases
Page5- Preparing and Testing Hydrogen Gas
3. Summarizethe physicaland chemica!propertiesof hydrogen gas by indicatingwhether
•the followingstatements are True or False:
(a) Hydrogen,gas is colorless.
(b) Hydrogendoes not diffusein air.
_______________
(c) Hydrogenis very solublein water.
_______________
(d) Hydrogenhas a greater density than air.
_______________
(e) Hydrogenis flammable.
_______________
(1) Hydrogensupports combustion.
_______________
• (g) Hydthgenand air will explodewhen ignited.
4. The reaction of metalswith acids is a general reaction that workswith a wide variety
of differentmetalsand mineral acids.Writebalancedchemical equations for the
reactionsof:
_______________
(a) Zincth
sulfuricacid.
•
(b) Aluminumwith hydrochloricacid.
Preparingand TestinjHvdrugcii (;is
Producing H
drogen Gos
Metal
FromCalcium
Procedure:
1.
2.
3.
4.
Putyour safety goggles
on.
e top withwater.
flaskalmostto tF
FillanErlenmeyer
Fill a beakeraboutthree-fourths full withwater.
Add2 dropsof phenolphthalèin
solutionto boththe beakerand the
is an indicator.
Erlenmeyerflask Phenolphthalein
5. Preparetwotest tubesfor hydrogenco ection. Fillthemboth with
• water andinvertthe full tubes into the beakerof water Pkiceyour
finger over the mouthof the test tube while inverting.Thi will
out of the test tubes whenyou
helppreventthe water from coming
turn them upsidedownand placethem into the beaker.
6. Obtaina smallpieceof calciummetal(peasize) anplaceit. intothe
centerof the Erlenmeyerflask Attach the rubber stopper/rubberglass tubing connector to the openingof the flask Extend the
rubber-glass
tubingintothe beakerof water. What doyouobserve
happening?
7. As the hydrogen
gas is producedandbubblesintothe beaker, place
oneof the test tubes over the bubblesto collectthe gas by water
Continue
to fill the test tube with hydrogengas until
displacement.
allof the water inthe test tube hasbeendisplaced.
8. Usinga match,lighta woodensplint.
9. Test for the presence of hydrogengas by inserting a glowing
wooden
splintintothe mouthof the inverted test tube.Be sure to
of the test tube awayfrom other people.What do
aimthe opening
youobservehappening?
1O.Repeat
steps 7-9 for the secondtest tube.
11.Makenoteof the changesthat havetaken placein the Erlenmeyer
flask The water has turned pink and there shouldbe a white
precipitateat the bottom. Swirlthe flask if yourdon’tsee it.
that follow.
12.Pleasecleanupand answerthe questions
Questions for Lob ProducingHydrogenGasfrom Calcium Metal
birections4 Pleaseanswer on ‘a sepc1r
ate
pieceof paper. •1
-
1.
2.
3.
4
5.
6.
7.
8.
9.
•
What is the chemical symbolusedfor”calcium?
In what periodandgroup is calciumlocated? (Give the #‘s)
What isthe family name for the group that containscalcium?
How many’valenceelectrons does calciumhave?
What type iondoes calciumbecomewhenit reacts with other elementsor
compounds? boes calcium like to gainor loseelectrons?
What type Ofelementiscalcium?
(Metal,’nonmetal,semimetal)
lookedlike before it wasplaced.in the
bescribewhatthe calciumphysically
flask filled withwater?
What are someof the chemical properties this ‘metalhas? Listfour.
Thecalciummetal reacts with the water to produce hydrogengs. Write a
balanced equationin the spacebelowto demonstrate this reaction. Be sure
to includeboththe reactants and products and their phaseof mattei’’
I
+
10. Wasthe reactiOnbetweencalciumandpjjeferendothermicor e.xothermic?
Howcanyou tell?
usedfor?
11. What wasthe phenolphthalein
12. Howdid youknowthat hydrogengas wasproduced?
13. The hydrogen burns In the presence of the glowingwooden splint to
producewater. When mixedwith oxygen, hydrogenignitesand makes a
bark or pop noise. Write a balancedequationin‘the spacebelowto
demonstratethis reaction. Be sure to includeboth the reactants and
productsandtheir phaseof matter.
‘
+
+
14. bo youthinkother metalswouldhavea similarreactionwith water? If so,
whichones?
be more or less
15. Wouldsodiumbe moreor less reactive?Wouldaluminum
reactive?
16. At the end of the reaction the directions ask you to observe a white
precipitate?What is a precipitate? What is the name of the white
precipitateformed?.(Hint Lookat the reactionyou wrote out for question
#9)
k
1!
Name______________________________
Class
pate____________
__________
9 Laboratory Investigation
Reactivity of Metals in Single-Replacement Reactions
Problem
Which metals will replace each other in single-replacementreactions?
Mateiials (per group)
•
four small pieces of the following metals: coppez. magn
sium,
the
solutions
of
dilute
nng
foUo
compounds: HQ, Cu
2,
d
12-wellspot plate (or12 small test tubes)
paperand pencil
Procedure
iron
2,
MgCI
3
FeCl
‘
1. Makesmalllabelsfor each ofthe wells on the spot plate.Each label should list one ofthe metali
being used and one p1the compounds Thereshouldkel2 labels in all.
2. Placea piece of copperin eachof fourwells on the spot plate.
3. Add enough HO to cover the copperin the first well. Labelthis well with the label that lists
the copper in the thirdwell
2,
copperand HO. Coverthe copperin the second well with Cud
with MgCI
fourth
and
the
with
Labeleachwell
well
2,
3.
FeC1
accordingly.
copperin the
4. Repeatsteps 2 and 3 using magnesium in clean wells on the spot plate.
5. Repeatsteps 2 and 3 us
ng iron in clean wells on the spot plate.
6. In the datatablebelow, recordany signs of chemical change that you observe in eachof the
wells. Possiblechanges couldinclude changes in color orstate,or the productionof bubbles.
,
Observations
C Prentace-Hall,
Inc.
•
Chapter9
23
..
.
Name_
9 Laboratory Investigation
.
..
,
/X7
Date____________ C’ass
__________
(continued)
(.
1. Whichof the metals reactedwith the most compounds?
2. Whichreacted with the fewest compounds?
Analysis and Conclusions
1. Describe what happened in thechemicalreactions you observed.
2. List the metals you tested from most reactive to least reactive.
3. Which of the metals you tested were able to replace the hydrogen in HG?
0;
I..
4. Where would .hydrogen as in
acid, be placed in the activity series you listed?
’ce
geferc,
blc
T
Do your experimental results
5. Compare your results with C
etni5trq
the
table? Explain possible reasons for disagreement.
agree with
6. On YourOwn Testthe same threemetals for reactivity with water. Can any of these metals
replacehydrogenin water?Is any metal capableof replacinghydrogen in water?
1)
24
Chapter9
C Pr.mic.-Haft. M
c.
DATE
NAME_
___________
Double Replacement Reactions & Solubility Rules (Table F)
Purpose: Observe the formation of various precipitates and, based on your observations.
formulate a hypothesis, regarding general solubility rules.
Materials: I test plate, pipets, safety goggles, lab apron, & beakers containing solutions:
A)
B)
C)
D)
0.1 M AgNO.
0.1 M Ba(NO
1)2
0.] M Cu(N0
1)2
0.1 M Pb(NO
1)
2)
3)
4)
0.IM KC1
0.1M KOH
0.IM KI
0.IM K,S0
4
Safety Warning: Bases such as KOH, will dissolve animal & vegetable tissue including
skin. You must wear goggles during the lab to protect against the possible splashing of
these solutions.
Procedure:
1. Obtain a test plate. Clean & dry it if necessary. Place it over the data sheet.
2. Place 4 drops of each solution in the tray in the row or column indicated.
Solution A in the 1 4 wells of row A.
Solution B in the 1St4 wells of row B.
Solution C in the 1 4 wells of row C.
Solution D in the IS 4 wells of row D.
Solution I in the 4 wells of column 1.
Solution 2 in the 4 wells of column 2.
Solution 3 in the 4 wells of column 3.
Solution 4 in the 4 wells of column 4.
Note: Do not touch the tip of the dropper on the tray.
Do not tip the tray.
Place the droppers back in the proper cups.
3. Record observations for the 16 reactions on the data table provided. (Include
in your observation color, appearance of the solution, & / or any precipitates
that form.
Conclusion:
1. Write the equation for the dissociation of each salt on the sheet provided.
2. For each precipitate formed in the experiment, circle the equation for the
double replacement reaction that occurred. Use the sheet provided.
3. Which ions were soluble? Where there exceptions?
4. Which ions were insoluble (formed a precipitate)? Where there exceptions?
5.
Compare your answers to questions 3 & 4 with Reference Table F?
V
Write anequation for the dissociationof
ach
• A.
1
•AgNO
4
• B.
3),
Ba(N0
4
C.
)2
Cu(NO
4
D.
1),
•Pb(NQ
1.
KC1
4
•
.2.
KQH
3.
KI
4.
SO
4K
:
4
salt in solution water).
/73.
For each double replacement reaction thaf occuned in the experiment. cikle the
precipitate formed.
4
AgCI
KOH
4
I
AgO
KI
4
Ag!
1.
AgNO +KCI
2.
3
AgNO
+
3.
.
AgNO
+
4.
2AgNO
5.
),
B’a(NO
+
6.
3)J
Ba(NO
1)
Ba(N0
4
.
2KCI
4
•
+
2KOH
4
+
2K1
+
4
K,S0
KNO
+
KNO
+
-4- KNO
2SO
.Ag
4
BaSO
+
Ba(OH).
‘2
2KNO
+
•,÷
2KNO
+
2KNO
3
2KN0
4
BaSO +2KN0
3
2KCI
4
CuSO
+
2KOH
4
Cu(OH).
Cu(NO
+
2K]
3
Cu!,
12.
1)Cu(N0
+
.SO
4K
9’
4
CuSO
+
2KNO
13.
)Pb(NO
+
2KCI
4
4
PbSO
+
1
2KN0
14.
L
Pb(NO
+
2KOH
3
,
Pb(OH)
15.
1)Pb(N0
+
2K1
4
,
Pbl
16.
.
Pb(NO
+
SO
K
4
4
PbSO
8.
3)2
‘Ba(N0
9.
),
Cu(NQ
.+
10..
1)
Cu(NO
11.
2S0
4÷K
.
+
+
+
2KNO
+
2KNO
2KNO
+
2KNOI
1
2KN0
+
2KNO
—i-.
te
MicropI
fDI
.‘
U)
U,
C’)
C’
U
C
C
C
C
U
C
0
C.
U
C.)
Chemistry: Concepts and Applications
Lab Manual
Page1 Introductionto ReactionRates
-
Introduction to Reaction Rates.
The “Blue Bottle” Reaction
Introduction
Howfastwill a chemicalreaction occur?If a reaction is too slow,it may not be practical. If
the reaction is too fast, it may explode.Measuringand controlling reaction rates makes it
possiblefor chemistsand engineersto make a variety of products, everythingfrom antibi
otics to fertilizers,in a safeand economicalmanner. The purpose of this experiment is to
investigatehow the rate of a reaction can be measured and how reactionconditionsaffect
reaction rates.
Concepts
• Kinetics
•
Reactionrate
• Collisiontheory
•
Oxidation—reduction
1
Background
Kineticsis the study ofthe rates of chemicalreactions.Asreactants are transformedinto
products in a chemicalreaction, the amount of reactantswill decreaseand‘theamount of
products willincrease.The rate of the reaction can be determined byrneasuring the concen
tration of reactants or productsas a function of time. In some cases, it is possibleto use a
simple visualclue to determine a .reactionrate. Thus, if one of the reactant is coloredbut the
productsare colorless,the rate of the reaction can be followedby measuring the time it takes
for the color to disappear.The averagerate of the reaction is then calculatedby dividingthe
molar concentration(M)of the coloredreactant by the time neededfor the color to disappear.
Dependingon howfastthe reaction occurs, the rate wouldbe reported in units of either
Wsec or M/min.
Reactionsinvolvingthe organic dyemethylene blue providea convenient exampleto study
reaction rates. Methyleneblue (abbreviatedMB)existsin two forms,a reducedform and an
oxidizedform, that havedifferentcolors.The reducedform of methylene blue (MBrd) j5
is blue. The reduced form is easilyconvertedto the
colorless,whilethe oxidizedform (MB
0)
oxidizedform by mixing it with oxygenin the air (Reaction1). The oxidizedform, in turn,
can be convertedbackto the reduced form bytreatment with a reducing agent, such as
dextrose,a reducing sugar.
MBrd
+
Reaction]
MB
2—
0,O
Blue
Colorless
with
In this experiment,we will study the rate of reaction of the blue, oxidizedform MB
0
dextroseand potassiumhydroxideto give the colorless, reduced form MBrd (Reaction2). If
the initial concentration of MB
in solution is known, the rate of the reaction can be deter
0
mined by measuring the time neededfor the blue color to disappear.
0MB
Blue
Ii I4
diicl Ion to ReactionRates
+
dextrose
+
KOH—* MB
Colorless
Reaction2
Introduction to Reaction Rates Page2
-
/
•
Experiment Overview
The purpose of this experiment is to investigatehow changing the temperature o the reac
tants or how changing the concentration of potassiumhydroxidewillaffectthe rate of reac
tion of methylene bIuë The basic process is alwaysthe same—whena colorlesssolution
containing MBrd is shaken,it turns blue (Reaction1). The time neededfor the solution to
turn colorless (Reaction2) will be measuredand will thep be used to determine the average
rate of reaction.
Pre-LabQuestions
•
1. Definethe terms oxidation and reduction.Note: Consultyour textbook,if necessary,for
definitionsand examples.
2. In the part of this experimentin which methyfeneblue changes from blue to colorless,is
it being oxidizedor reduced?Whatreactant is causing this change?Is this reactant acting
as an oxidizingagent or a reducingagent?
3. Collisiontheory offersa simpleexplanationfor howreactions occur—reactingmolecules
must first collide.In order for collidingmoleculesto be convertedinto products,they
must collidewith enough energyand with a suitableorientation to break existingbonds
(in the reactants)and form newbonds (in the products).Anyfactorthat changes either
the total nunTherofcollisionsor the averageenergy of the collidingmoleculesshould
affectthe reaction rate.
ing
the temperature should affectthe rate
(a) Usingcollisiontheory,predict how increa
of a chemicalreaction.State the prediction in the form of an if/then hypothesisand
givea reason for your hypothesis.
(b) Usingcollisiontheory,predict how increasing the concentrationof a reactant should
affectthe rate of a chemicalreaction. State the prediction in the form of an if/then
hypothesisand givea reason for your hypothesis.
Materials
“Bluebottle”solution for Part A, 10 mL*
0.1 M, 12 mL
Dextrosesolution, 06,C
12
H
1 mL
Methyleneblue solution, 0.1%,
Potassiumhydroxidesolution, KOH,3.0 M, 6 mL
Water,distilled or deionized
Washbottle
Labelingor marking pen
Metricruler
Stopwatchor clock (watch)with second hand
•
Beakers,100- or 150-mL,4t
Beral-typepipets, thin-stem, 4
Graduated cylinder,1O-rnL
Hot plate or warm water
Ice or coldwater
Test tubes, medium, 3
Testtube rack
Thermometer
Stoppersto fit test tubes, 3
*Theblue bottle solution contains dextrose,potassium hydroxide,and methylene blue.
tSeveralgroups may share beakersto make water baths at different temperatures.
FlinnChemTopicLabs Kinetics
—
Page3- Introductionto ReactionRates
Safety Precautions
Potassium hydroxidesolution is a corrosiveliquid;it is particularly dangerousto eyes and
may blisterand burn skin. Avoid contact with eyes and skin and clean uj all spills immedi
ately. MEthyleneblue is slightly toxic by ingestion. Wearchemical splashgoggles and
chemical-resistantgloves and apron. The dextrOse(sugar)solution will attract ants. Rinse
off all work areaswith water and wash:hands thoroughly with soap and water before
leaving the laboratory.
Procedure
Part A. Effect of Temperature
L Obtainfour100-or 150-mLbeakersand
• makewaterbathsat approximatelythe
followingtemperatures:10 °C,20 °C,
30 °C;and40 °C.In orderto obtain
tion
times, avoidtern
• convenientrea
peraturesapve 40 °C or below10 o
Tie a knot
2. Obtain four,thin-stem pipets and place.
a mark 2 cm fromthe bottom on each
pipet bulb.
3. Fill each pipet bulb to the 2-cm mark
with the “bluebottle”solution. Tie a
knot in the stem of each pipet to seal it.
(SeeFigure 1.)
4. Place one pipet into each of the four
water baths from step 1. Let the pipets
stand in the bath for 3—5minutes.
Recordthe temperature of eachwater
bath in the datatable.
Label
Solution
2 cm
5. Removethe pipet from the 20 °Cwater bath, start timir
ç,
fivetimes and immediatelyreturn it to the ‘ater bath.
Figure 1.
then quicklyshakethe pipet
6. Stop timing whenthe blue color fades completelyand the solutionturns colorless.
Recordthe elapsedtime in secondsin the data table.
7. Repeatsteps 5 and 6 with the other three pipets. Recordall time and temperature read
ings in the data table.Note: Tryto shake the pipets the same wayeach time. Aftershak
ing, return the pipetsto their respectivewater baths.
8. Disposeofthe pipetsas directed by your instructor.
Part B. Effect of Concentration
9. Obtainthree mediumtest tubes and stoppers and labelthem #1—3.
10. Usinga graduatedcylinder,add 3.0 mL of dextrosesolution to each ofthe three labeled
test tubes.
Introductionto ReactionRates
Introduction to Reaction Rates Page4
-
/71
11. Addone drop of methylene blue solutionto each test tube.
12. Measure1.0mLof3.0 M potassiumhydroxidesolution into a clean graduated cylinder,
then add 2.0 mLof distilledwaterto get a finalvolumeof3.0 mL.
13. Pour the contents of the graduatedcylinderinto test tube #1. Stopper the test tube and
shake gentlyto mixthe solutions.
14. Measure2.0 mLof 3.0 M potassiumhydroxidesolution into a clean graduated cylinder,
then add 1.0mLof distilledwater to get a finalvolumeof3.0 mL.
15. Pour the contents of the graduatedcylinderinto test tube #2. Stopper the test tube and
shake gentlyto mixthe solutions.
16. Measure3.0 mLof 3.0 M potassiumhydroxidesolution into a clean graduated cylinder.
17. Pour the contents of the graduatedcylinderinto test tube #3. Stopperthe test tube and
shake gentlyto mixthe solutiOns.
18. Allowthe test tubes to sit undisturbedat room temperature until the blue color fades.
Note: This may take a fewminutes.
19. Checkthe temperature ofthe solutionsto be sure theyare allabout the same temperature.
Recordthe temperature in the data table.
20. With your finger firmly on the stopper,shake test tube #1 vigorouslyfivetimes nd
immediatelystart timing.
21. Stop timing when the blue color fadescompletelyand the solution turns colorless.
Recordthe elapsedtime in secondsin the data table.
22. Repeatsteps 20 and 21 using test tube #2 and then againusing test tube #3.
23. Disposeof the contents of the test tubes as directed by your instructor.
FlinnChemTopic”
Labs
—
Kinetics
Page5—Introductionto ReactionRates
Name:
Class/LabPeriod:
_____________
Introduction to Reaction Rates
I
Data Table
Part A. Effect of Temperature
Temperature, °C
Reaction Time, sec
Part B. Effectof Concentration
TestTube
2
1
3
ReactionTime,sec
Temperature,°C
.
.
Post-Lab Questions
1. Howdidthe reactiontime change as the temperature waschanged in Part A?
2. Howis the rate of a reaction related to the time of reaction?
3. What effectdoes temperature have on the rate of the “bluebottle”reaction?
4. Accordingto a general “rule of thumb” for chemical reactions, the rate of a reactionwill
roughly doublefor every 10 °C increasein temperature. Dothe kineticsof the “bluebot
tle” reactionfit this general rule?
Introductionto ReactionRates
Introduction to Reaction Rates Page6
-
•
5. On a separate sheet of paper,make a graph çf the results in Part A byplotting the reaction
time in seconds on the y-axisversus the temperature in kelvinson the x-axis.’
6. Usingthe graph, estimatehowlong it wouldtake forthe reaction to occur at 275 K and at
325 K.Discusswaysthe graph could be improvedto give,better estimates.
=
7. Usethe “dilution”equation (M
o calculatethe concentration of potassium
)
2V
M
1V
B.
each
tube
#1—3
i
n
Part
in
test
hydroxide
1
M
=
=‘
concentrationof KOHbefore mixing
concentrationof KOHafter mixing
Samplecalculationfor test tube #1:
M2
—
1V
2
V
=
volumeof KOHbefore mixing
volumeof KOHafter mixing
‘
)(3.OM)(1.OmL
(6.O’mL)
050M
8..The concentration of methyleneblue in Part B is approximately2.0 x 10 M.Dividethe
ction time in secondsto calculatethe average
concentration’of methyleneblue by the re
rate of the reaction in units ofWsec for each test tube #1—3.
9. Doesthe rate of the “blue bottle”reaction depend on,the concentration of potassium
hydroxide?Discussin generalterms the effectof reactant concentration on the rate of a
chemicalreaction.
10. Howmuch did the rate of the reaction,change when the concentration of KOHwas
doubled(test tubes #1 and 2) or tripled (test tubes #1 and 3)?
FlinnChemlopic” Labs
—
Kinetics
Name
Date ____________ Class
___________________________________
ed
____________
“
Ratesof Reaction
Lab32
lext reference: Chapter17. pp. 47
_475
Pre-Lab Discussion
•
•
The rate of a chemical reaction is the nine required for a given quantity
of reactant(s) to be changed to product(s). Reaction rate usually isexpres
sed in terms of moles per unit tune. This rate is affected by several factors,
including the nature of the reactants, concentration f the reactants, tern
perature, pressure, and the presence of catalysts. in this experiment, you
will study the effects of temperature and concentration.
TtiA chemical reactidn is the result of çffective collisions between pa
des of .nts.
Increasing the temperature of a system raises the aver
ract2
age kinetic ‘éner&vof the particles of the system. This results in more
collisions and, of greater importance, more effective collisions per unit
time. This affects the rate of the reaction.
At constant temperature, increasing the concentration of one or more
of the reactants increases the number of particles present and, hence, the
number of collisions. This affects the rate of the reaction.
In this experiment, two solutions will be mixed, and the completion
of the reaction will be marked by a color change. One solution contains
The other contains the hydrogen sulfite ion (HS0
3)
the iodate ion.(
3).
10
and soluble starch. The entire reaction takes place in two stages. The ionic
equations for these stages are:
I.
3(aq)
10
2. 5V(aq)
•
±
3(aq)
3HS0
-4- 6H
’(aq)
—
-4- 10
3(oq)
F(aq)
—
-4-3S0
42T(aq)
(aq)
3l
+
3H(aq)
-4- 3H
2O(1)
In the presence of starch molecules (not shown), molecular iodine (12)
produces a characieristic blue color. The rate of the entire reaction can
be determined by timing the interval between the time the two solutions
are mixed and the appearance of the blue color. By varying the concentra
tion of one of the reactants (at constant temperature) and then varying
the temperature alone, you can observe and record the effects of these
two factors on reaction rate.
This experiment should provide a better understanding of reaction
rates and the factors that affect these rates.
Purpose
the concentration of a reactant has on the
rate of a chemical reaction. Study the effect that changing the temperature
has on the rate of a chemical reaction. Formulate hypotheses about how
reaction rates are affected by changes in temperature and in concentration
of reactants.
Study the effect that
changing
163
Equipment
beal 25Q-rnL
er.
beakers. 100-mL(2)
timer (StOp watch or cluck
with second hand)
safety goggles
lab apron or coat
graduatedcylinders. ]O-mL (2)
test tubes. 18x]50-mm(2i
thermometer
Materials
Solution A (with JO. ion)
distilled water
ice cubes
Solution B (with HSO. ion and
soluble starch)
Safety
Avoid spilling reagent solutions on your skin or clothing. \ash off any
spills immediately with cold tap water. Note the caution alert symbols
here and with certain ‘steps in the Procedure.” Refer to page xi to review
the precautions associated with each symbol. Always wear safety goggles
and a lab apron or coat when working in the lab.
Procedure
,
I’
PARTA
1. Using a clean. do’, l0-mL graduated çyIin
er.
measure
exactly 10.0mL of SolutionA and pour it into a ]00-mL beaker.
2. Using a second 10-mL graduate. measure exactly 10.0mLof
Solution B and pour it into a second ]00-mL beaker.
3. Prepare to time the reaction. While one lab partner pours
Solution A into Solution B, the second partner should im
mediately start timing the reaction. Pour the solutions back and
forth several times from one beaker to the other to ensure
thorough mixing. Then allow the mixture to stand At the instant
a color change occurs, the partner timing the reaction should
note the elapsed time. Record this in your data list. Rinse and
dry the beakers and graduated cylinders.
4. Measure exactly 10.0mL of Solution B into one of the beak
ers. Using a clean graduated cylinder, measure exactly 9.0 mL of
Solution A into the other beaker. Dilute this solution by adding
exactly 1.0 rnL of distilled water. Follow the step 3 instructions
for mixing the solutions arid timing the reaction. Record the
elapsed ume in your data list. Rinse and thy the beakers and
graduated cylinders.
5. Repeat step 4 four more times, using increasingly dilute
samples of Solution A. Use the following ratios of Solution A to
distilled water (in mnL):8 to 2; 7 to 3; 6 to 4; and 5 to 5. Rinse
and dry the beakers and graduated cylinders after each trial.
Record elapsed times in Part A of Observations and Data.
1
164
Name
•32 Ratesof Reaction(continued)
PART B
6. Measure 10.0rnL of Solution A into one test tube and 10.0
mL of Solution B into a second test tube.
7. Half mi a 250-mLbeaker with cold t.ap water. Add ice cubes
to the. water and stir carefuliy with the thermometer. Continue
stirring (and adding ice as needed) until the temperature of the
ice-water mixture is about 5°C.
Figure 32-1
8. Place thetwo test tubes in the ice-water bath arid let them
stand until the solutions are at the same temperature as the ice
water (Figure 32-1).Always rinse and wipe the thermometer
a.Iter removing ii from a sohition.
9. When the solutions are at the same temperature as the ice
water, prepare to time the reaction. One lab partner should start
timing the reaction the instant the second partner pours Solution
A into Solution B. Quickly pour the mixture back and forth from
test tube to test tube several times and return the mixture to the
ice-water bath. At the instant a color change occurs, note the
time elapsed. Measure the temperature of the mixture im
mediately. Record the exact temperature and elapsed time in
‘our data table. Discard the mixture as instructed. Rinse arid dry
the test tubes.
10. Repeat step 6.
11. Prepare a water bath at a temperature of about 15°C.Repeat
steps 8 and 9 at this new temperature. Record your observations
in your data table.
12. Repeat these procedures using warm baths at the following
temperatures: 25°C;35°C; 45°C. Use warm tap water to prepare
these baths. Rinse and dry the test tubes after each trial.
165
ObservationsandData
PARTA
I
SoJutio B (rnL
)
10
20
H
Solution A irnL
,Tirne(sec
)
(mL
0
10
10
i
10
8
2
10
7
3,
10
6
4’
5
10
PARTB
DATA TABLE
Trial
:
Time(sec)
Temperature(CC)
•
1
.i
2
3
4
I
‘
.
,
,.
I
.,.
•
:
.1
Calculations
1. Plot your data from Part A on the grid provided. Draw a line through
the plofled points to produce a curve showing the effect of concentration
of reactants on reaction rate.
I
166
Name
________________________
Ratesof Reaction(continued)
24
0)
E
C
°
0)
20
16
12
8
4
0!
j
0
1
I
2
3
I
I
4
5
I
I
6
I
I
7
i
8
9
10
ml of SolutionA
2. Plot your data from Pan B on the grid provided. Draw a line through
the pinned points to produce a curve showing the effect of temperature
on reactiOn rate.
32
28
24
U
a)
a)
20
E
16
C
t
t
0)
12
S
4
0
ii’
0
5
10
15
20
25
30
35
40
45
50
lemperalure (CC)
167
and Questions
ConclusionS
I. Basedon your experimental data. make a general stateme
ht
(hvpoth.
esis).aboutthe effect of concentration of reactants on reacticrn ra
t’.
2. Make a similar hypothesis about the effect of temperaturt on reacuon
rate.
3. Whatother factors affect the rate of a reaction?
4. How doesthe collision theory relate to the rate of a chemical reac
icn?
(
(
168
‘S.
%
5-.,
g
Determining Reaction Rate
Problem
Observations
Howdoes concentrationaffectreactionrate?
Volume
of Volumeof Volumeof Reaction
Water
MCI
Time
3
O
S
2
Na
Beaker (ml)
(mL)
.)
(mL
(a)
Materials (per group)
3 graduatedcylinders
5 125-mLbeakers,labeled1—5
0.20 M Na
’
3
O
S
2
2.OMHCI
distilledwater
sheetof white‘paper
black markingpen
timer or stopwatch
,
2.
,
3,
,
Procedure
1. Use graduatedcylindersto measureand
add 0.20 M Na
and distilledwaterto.
3
O
S
2
the beakersas desôribedbelOw:
Beaker
3
O
S
2
Na
1
2
3
4
5
10.0mL 20.0 mL 30.0 mL 40.0 mL 50.0 mL
Water 40.0mL 30.0 mL 20.0mL 10.0mL 0.0 mL
2. On a sheetof paper, use the markingpen to
writethe letterX smallenoughso that it can
be coveredby a beaker..
3. PlaceBeaker1 on the X. Usethe remaining
graduatedcylinderto add 5.0 mL of 2.0 M
HCI to the beaker.Be carefulto use proper
safetyprecautionswhenworkingwith acid.
4. Recordthe timeat whichthe HCIis added.
Immediatelygive the beakerone gentle swirl
and place it back on the X. Look through
the solutionat the X and record the time at
whichthe X is no longervisible.
5. RepeatStep4 for the remainingbeakers.
744
—
1. Whatvisibleindicationis therethat a
chemicalreactionis occurring?
2. Howis the concentrationof Na
3
O
S
2
affectedby the volumeof distilledwater?
3. Whathappensto the reactiontime as the
volumeof distilledwaterincreasesrelativeto
the volumeof Na
?
3
O
S
2
4. Preparea graph of time versusvolumeof
3
O
S
2
Na
by plottingtime alongthe x-axis
and volumealongthe y-axis.
Analysis and Conclusions
affect
1. Howdid the concentrationof Na
3
O
S
2
the rate of the reaction?
2. Doesyour graphsupportyour conclusionfor
Question1? Explain.
3. In this investigation,what are the variables?
Whatis the control?
4. Basedon your data,whatdo you think might
be the ratelawfor this reaction?
5. On YourOwn With your teacher’sper
mission,repeatthis experimentkeepingth
concentrationof Na
constantand
3
O
S
2
HCI.
concentration
of
varyingthe
Unit Four: Thermodynamicsand Kinetics
398
ENERGY
REACTIONRATESANDACTIVATION
4.3.4 CATALYSTS,
Concepts to Investigate: Reaction rates, catalysts, activation energy, autocatalysts; doliision
theory.
solutionfrom drugstore(higherconcentrations
Materials: PartI: Hydrogenperoxide(H
)
0
2
are avaiJablefrom chemicalsupplycompanie andwork better,but must be handledwith
dioxide (Mn0
activatedchar
),
2
greatercaution),apparatusshownin Figure’K, manganese
potassiumiodide(KI),
)
4
(KMnO
,
potassium
coal, calciumcarbonate(CaCO
),
3
permanganate
spatula; Part2 Sugarcubes,activatedcarbon(fireplaceash),tongs,ceramictile or ashtray,
matchor burner.
Principles and Procedures: A catalyst is a substance that speeds up the rate of a chemical re
action and can be recovered at the end of the reaction in its original form. Often only a trace
of a catalyst is sufficient to accelerate a reaction A catalyst speeds up a reaction by ‘provid
ing a set of elementary steps with more favorable energetics than those that exist n’its ab
sence. Catalysts are classified as heterogeneous when the reactants and catalyst are in differ
ent phases (for example, solid catalyst and liquid or gas reactants) or homogeneous when the
catalyst is in the same phase as the reactants and products.
y
required to
Catalysts increase the rates of reactions by decreasing the activation ener
in
collision
order
the
to react, colliding particles must
initiate a reaction. According to
theory,
have a total kinetic energy equal to or greater than ‘theactivation energy, the minimum amount
of energy required to initiate a chemical reaction. If the minimum energy is not available, the
particles remain intact and no change results. If the energy is available, the particles enter a
transitional structure called an activated complex that results from an effective collision and
that persists while old bonds are breaking and new bonds are forming. Figure L shows the en
ergy relationship among reactants, products, activated complex and catalyzed activated corn
plex. Note that the presence of a catalyst (curved dotted line) reduces the activation energy
required. As a result, a larger percentage of the collisions occurring in a catalyzed reaction
meet the energy requirements to react, and the reaction proceeds more rapidly. In the reaction
illustrated, the products are more stable than the reactants, and the reaction is exothermic and•
is accompanied by a release of heat (—i
1i1). The reverse reaction is endothermic. Heat is ab
sorbed and the reactants are more stable than the products. Note that I1 is positive in an en
dothermic reaction.
J
‘
Aciivated
Complex
id
:_
K
L
/
‘
aj,is
,c
Re
Produci.s
Time
‘
4.3 Chemical Kinetics
399
Part 1: Catalysts: Hyd
ogen
peroxide is a colorless liquid used as a rocket propellant, dis
infectant, and bleaching agent. You may have used a dilute hydrogen peroxide solution to
sterilize a wound. Hydrogen peroxide slowly decomposes into water and oxygen:
+ 02(g)
(aq)—
2O
2H
(1)
j
Thisprocess can be accelerated by the addition of numerous substances, particularly salts of
such metals as iron, copper, manganese, nickel or chromium. It should be noted that these
substances accelerate the decomposition of hydrogen peroxidL but are not consumed in the
process. Such substances are known as catalysts.
Set up a gas collection device as illustrated in Figure K. Fill the test tube with water,
cover the mouth, and invert into a container of water so no water is lost from the tube. Place
three grams of manganese dioxide in a large test tube and clamp to support as shown. In
sert a stopper assembly equipped with a delivery funnel and bent glass tube as illustrated.
Add 5 mL of 3% hydrogen peroxide through the delivery funnel. Coiiect the test tube of gas,
and set aside. Continue collecting gas in additional tubes until the reaction ceases.
r,
stopp
Now add more hydrogen peroxide and continue until you have collected 5 test tubes of oxy
gen. Note that the manganese dioxide is not used up in the reaction. It remains visible in the
tube, and promotes the decomposition of hydrogen peroxide repeatedly. Manganese dioxide
is therefore considered to be a catalyst, and the reaction can be written:
S
‘
2H202(aq)
M002
)2H20(l)
+02(g)
Note that the manganese dioxide is written above the arrcw, indicating that it is not changed,
but only catalyzes the reaction Unstopper the first tube of gas collected, and test for the pres
ence ofoxygen by inserting a glowing splint or smoldering match. Test each of the remain
ing tubes in the same manner. The flame should glow brightest in the final tube because it
contains relatively pure oxygen, while the first tubes may contain some air displaced early in
the reaction. Decant the hydrogen peroxide from the reaction flask and add fresh hydrogen
peroxide to the old manganese dioxide. Does manganese dioxide continue to promote the
production of oxygen?
Repeat this activity using 3 grams each of activated charcoal, potassium permanganate,
potassium iodide, and calcium carbonate. Record your results in Table 3. Which of the ma
terials appear to be catalysts?
Part 2: Catalysts in combustion: Hold a sugar cube with a pair of tongs above a ceramic
tile, ashtray or other fireproof surface, and try to burn it with a match or laboratory burner
(Figure M). Since the melting point of sucrose is only 185°C, the sugar cube melts before it
Table 3: Testsfor Catalysts
Rate of Bubbling
manganese dioxide
activated charcoal
calcium carbonate
potassium permanganate
potassium iodide
Glowing Splint Test
Catalyst?
400
Unit Four: Thermodynamics and Kinetics
burns. Dip a second cube in very fine powdered
activated charcoal (carbon) or ash from a fire
place or ashtray. When a match is brought to
this cube, the cube burns readily. The carbon
acts as a catalyst in the combustion of sugar.
Questions
ashes,aclivalcd
charcoal,or carbon
powder
(1) What is the purpose of a catalyst?
(2) Describe in your own words the meaning of “activation energy” and “activated com
plex.”
(3) Although the heat of formation (LIH = 285.8 kJ/mol) and the free energy change
G = —237.2kJ/mol) for the reaction of hydrogen and oxygen to form waterare quite’
(L
and oxygen (02) are mixed at room temperature, they do
high, when hydrogen (H
2)
not combine spontaneously to form water (H
Explain.
20).
(4) A catalyst lowers the activation energy for a reaction. Explain.
4.3 Chemical Kinetics
395
ON REACTIONRATE
4.3.2 THE EFFECTOFTEMPERATURE
Concepts to Investigate: Reaction rate, temperature.
Materials: Part I: Vinegar, baking soda (NaHCO
beakers, side arm flasks or flasks fitted
3),
with stoppers and tubing; Part 2: Alka Seltzer® tablets, thermometer, plastic film canisters
(optional).
Principles and Procedures:
Part 1: The effect of temperature on neutralization of vinegar: Determine the approxi
mate rate of carbon dioxide production (in mL of carbon dioxide per unit time) as a function
of temperature using the apparatus described in 4.3.1 (Figure G). Repeat the investigation us
ing undiluted vinegar at temperatures of approximately 0°C, 15°C, 30°C, 70°C. Use an ice
bath or hot plate to achieve the reqpired temperatures. Report your results in Table 2 and plot
them in Figure H.
1
C
I
(
2
-J
E
H
-
* HC
0
3
H
2
3
NaI-1C0
—*
NaC
O
H
2
2 (g) H
CO
0
2
I
temperature
Part 2: The effect of temperature on antacid reactions: Heartburn (acid indigestion,
sour stomach) is a common problem associated with anxiety or an acidic diet. An exces
sively acidic stomach may cause considerable discomfort that can be relieved by neutral
ization of stomach acids. Numerous products are sold to combat heartburn, one of which is
and
Alka Seltzer®. Alka Seltzer® contains calcium dihydrogen phosphate 4)2(Ca(H
P0
Table 2: The Effect of Temperature on Reaction Rate
Temperature, C°
ReactionRate,mL C0
/s
2
Unit Four: Thermodynamicsand Kinetics
396
sodium hydrogen carbonate (NaHCO
3).
follows:
+ 2NaHCO
3(s)
Ps)0
4)2(Ca(H
20(l)
2
2C0
(g)+ 2H
stances
When placed in water, these sul
2
+ Ca
react as
)
(aq) + 2Na* (aq)
42
(aq) + 2HP0
Note ihat one of the products is carbon dio*ide gas. It is this gas that causes you to burp af
ter swallowing a glass of dissolved Alka Seltzer®.When Alka Seltzer! is placed in waler it
fizzes vigorously. The rate of the above reaction can be gauged by the apparent rate of bub
bling. Place a fresh tablet of Alka Seltzer® in ice water, water at room temperature, and wa
ter that is near boiling (Figure 1.) Which one fizzes fastest?
• ,,S
II.iIJ,
ij
1,1’
:::
•
.
,q.
_.
Ps)O
3
(Ca(H
)2
2C0, (g)4 2H
O(I)
apprwumalely900c
2NaJ1CO,(s)
+
Ca(aq)4 2HPO
(aq)
2
+
2Na(aq)
0°C
An interesting alternative to this activity is illustrated in Figure J. Put on goggles and a
lab coat. Fill one plastic film container one third full with ice water, and.a second with wa
ter at room temperature. Do not use hot water because water may splatter and cause burns!
Place an Alka Seltzer®tablet in the canister, seal, stand back, and record the time at which
the top pops off. Repeat two or three times at each temperature and determine average val
ues.
Questions
(1) Describe your observations of the relationship between temperature and rate of reac
tion.
What
would you predict concerning the rate of reaction if both the concentration and
(2)
temperature were increased?
(3) Food spoils rapidly at room temperature, but when placed in a refrigerator or freezer,
food can be kept for extended periods. Explain.
(4) Explain the difference in the “fizzing” of Alka Seltzer® at different temperatures.
Page1 ExploringEquilibrium
—
ExploringEquilibrium
It Works Both Ways
Introduction
The word equilibrium has two roots: cqui, meaning equal, and libra, meaning weight or
balance. Our physicalsense of equilibrium—in the motion of a seesawor the swing of a
pendulum—suggestsan equal balanceof opposing forces.Howdoesthis physicalsense of’
Let’sexplore the nature and consequences
equilibrium translate to chemical equilibr
üm?
of equilibrium in chemicalreactions.
Concepts
‘
,
Reversiblereactions
•
Chemicalequilibrium
• Complex-ionreaction
•
indicators
Acid—base
..
Background
•
Physicalchanges,such as melting ice or dissolvingsugar,are often introduced by noting
that these processescan be easily reversed.Some common examplesof chemical change,
such as burning woodor spoilingfood,generallycannot be reversed.Acloser lookat chemi
‘cal change, however,revealsthat many chemicalreactions are also reversible.
Considerthe followingexampleof a.reversibl•echemical reaction.At high pressures and in
the presence of a catalyst,nitrogen and hydrogen.react to form ammonia.If the temperature
is high enough, however,ammonia decomposeso reform its constituent elements.The
reaction can go both ways!This reversiblereaction is representedsymbolicallyusing double
arrows (Equation1).
2(g)
N
+
2(g)
3H
3(g)
2NH
Equation 1
What hajpens when nitrogen and hydrogenare allowedto react?In a closedsystem, the
concentrations of nitrogen and hydrogen will decreaseand the concentration of ammonia
will steadily increaseas the reaction proceeds in the forwarddirection. Soon, however,the
concentration ofammoniawill be large enough that the reverse reactionwill begin to take
placeat a significantrate as well. Eventually,as the reaction occurs in both the forwardand
the reversedirections,the number of ammonia molecules being formedwill becomeequal
to the number ofammonia molecules being consumed.At this point, no further changes
willbe observedin the overallconcentrations of nitrogen, hydrogen,and ammonia.This is
the point of chemicalequilibrium.
Chemicalequilibriumis definedas the state where the rate of the forwardreaction equals the
rate of the reversereactionand the concentrations of reactants and products remain constant
with time. Notethat this definitiondescribesa dynamicpicture of equilibrium.The reactions
continue, but there is an equal balanceof opposingreaction rates.
What happens when the equilibrium is disturbed?Any factor that changes the rate of the
forwardor the reversereaction will change the amounts of reactants and products that are
present at equilibrium.Reactionconditions that are known to affectthe rates of chemical
reactions includethe concentrations of reactants and the temperature. In this experiment,
ExploringEquilibrium
ExploringEquilibrium Page2
—
we will investigatehow changes in reaction conditionsaffectthe amounts of reactants and
products present at equilibrium.
Experiment Overview
The purpose of this experimentis to explorethe nature ofchemicalequilibriumand to identify
conditionsthat affectthe positionof equilibrium.Twodifferentreversiblereactionswillbe
studied.
Reactionof iron(1I1)nitrate with potassiumthiocyanate will be used to study complex-ion
2
equilibrium. Iron(III) ions react with thiocyanate ions to form FeSCN
complex ions
(Equation2). The effectsof changing the concentrations of reactants and of changing the
reaction temperature will be investigated.
3(aq)
Fe
+
2aq)
FeSCN
SCN1aq),
Equation 2
The properties of an indicatorwillbe used to study acid—base
equilibrium.An indicator is a
form
substances
that
havedifferentcolors.
dye that can gain or lose hydrogen ions to
Equation 3 summarizes the reversiblereaction of the indicatorbromcresol green (H
n). HIn
represents an uncharged indicator moleculeand 1n an indicatoranion formedafter the
moleculehas lost a hydrogen ion. The color of the indicator in the presence of either excess
H or OH-ions (see Equation4) willshow how changing the concentration of a product
affectsthe equilibrium shown in.Equation 3.
HIn(aq) t
GolorA
(aq)
H
W(aq)
+
OHiaq)
—
+
In
aq)
ColorB
zuation 3
E
Equation 4
90(I)
H
Pre-Lab Questions
1. True or False:At equilibrium,no more reactants are transformedinto products. If false,
rewritethe statement so that it correctlydescribes the nature of chemical equilibrium.
2. True or False:At equilibrium, the concentrations of reactants and products are equal.
If false,rewrite the statement so that it correctly describesthe nature of chemical
equilibrium.
3. Paper coatedwith cobaltchlorideis sold commerciallyas moisture-sensitivetest strips to
estimate relativehumidity levelsbetween20 and 80 percent in air. The following.
reversiblereaction takes place with water:
2(CoCI
s)
Blue
+
20(g)
H
t:
96H(s)
CoCl
2O
Pink
(a) Whatcolor do you think the paper will be when the humidityis low (20%)?What
cuIrir will il h when the humidity is high $0%)?
(hI ‘l’licI:si
itili
with a color chart to esl;iiii;itc inteniiecliatehumiditylevels.Predict
cnlor that might he ohscrved whcu the humidity is about 50%.
strips coilie
inli;Ic
Flinn Chemlopic’ Labs
—
Equilibrium
Page3- ExploringEquilibrium
Materials
’mcresol green indicator,0.04%, 1 mL
Bro
0.1 M, 4 mL
lron(I1I)nitrate solution, Fe(N0
3),
Hydrochloricacid,HCI,0.1 M, 2 rnL
Potassium thiocyanatesolution, KSCN,0.1 M, 4 mL
Sodium hydroxidesolution, NaOH,0.1 M, 2 mL
Sodium phosphate(monobasic)solution,
0.1 N, I mL
2P0
4,NaH
Water,distilledor deionized
Washbottle
‘
Beaker,50-mL
Beakers,250- or 400-mL,2
Beral-typepipets,,graduated, 8
Hot plate
Ice
Labelingor marking pen
Stirring rod
Test tubes, small, 6,
Testtube rack
Thermometer
Safety ‘Precautions
Potassium thiocyanate is toxic by ingestion. Dilute hydrochloricacidandsodium hydrox
idesolutions are skin and eye irritants. Jron(III)nitrate solution is also a possib/eskin and
body tissue irritant;it willstain clothes and skin. Avoid contact of all chemicalswith eyes
and skin. Cleanup all chemical spills immediately. Wearchemical splashgoggles and
chemical-resistantgloves and apron. Washhands thoroughly with soap and water before
leaving the laboratory.
,
‘,
Procedure
Part A. Complex-Ion Equilibrium of Iron(III) and Thiocyanate Ion
1. Fill two beakers(250-or 400-mL)half-fujiwith tap water.Addice to one beakerto
prepare an ice-waterbath (0—5°C) for use in step 8. Heat the secondbeaker on a hot
°C) for use in step 9. Donot boil the water.
plate to preparea hot water bath (70—80
2. Observeand recordthe initial colorsof the Fe(N0
3)
and KSCNsolutions.
2’:
3. Preparea stocksolution of FeSCN
In a clean50-mLbeaker,measure 40 mL of distilled
and
water.UsingseparateBeral-typepipets for each solution, add 1 mL of 0.1 M Fe(N0
3)
2 mL of 0. 1N KSCN.Mixthoroughly with a stirring rod.
4. Labelsixclean test tubes 1—6.Usinga graduated,Beral-typepipet, add 1 mL of the
2
FeSCN
stocksolution to each test tube 1—6.
5. Add10 drops ofdistilledwater to test tube 1. Gentlyswirlthe test tube to mixthe solu
tion and recordthe color of the solution in the data table.Testtube 1will be used as the
control solutionfor comparison purposes in steps 6—10.
6. Add10 drops of 0.1 M Fe(N0
to test tube 2. Gentlyswirlthe test tube to mix the solu
3)
tion and comparethe color of the resulting solution to the control in test tube 1. Record
the color comparisonin the data table.
7. Add10 drops of 0.1 MKSCNto test tube 3. Gentlyswirl the test tube to mix the solution
and compare the color of the resulting solutionto the control in test tube 1. Recordthe
color comparisonin the data table.
ExploringEquilibrium
ExploringEquilibrium Page4
-
8. Add10 drops of distilledwater to test tube 4 àr
d placethe sample in an ice-waterbath.
tube
from
the icebath and compare the color of the
the
After3—5minutes, remove test
solution to the control in test tube 1. Record,the color comparisonin the data tble.
9. Add10 drops of distilledwater to test tube 5 and place the sample in a hot water bath at
70—80°C.After2—3minutes, remove the tube from the hot water bath and conipare the
color of the solution to the control in test tube 1. Recordthe color comparison in the
data table
10. Totest tube 6, add 10 drops of 0.1M 4.NaH
2PO
solution in the data table.
Recordthe colorand appearanceof the
11. Washthe contents ofthe test tubes downthe drain with eccess water and rinse with dis
tilledwater.
Part B. Acid-Base Equilibrium Of Bromcresol Green
12. Obtain 2’mL of distilledwater in a clean test tube and add 5 drops of 0.04%bromcresol
green. Swirl gently and record the color of the solution in the data table.
13. Add3 drops of 0.1M1-ICIsolutionto the test,tube. Swirlgently and record the new color
of the solution in the data table.
14. Add0.1MNaOHdropwiseto the solution until the originalcolor is restored. Shake
gently and record the number of drops of NaOHaddedand the color of the solution in the
data table.
15. Continueadding0.1MNaOHdropwiseuntil a total of 5 drops of NaOHhavebeen added
in steps 14and 15combined.
Canthe process be reversedto obtain a color that is intermediate between that in steps 13
and 14?
16. Add0.1 M HCIagain dropwisevery slowlyuntil the solution reaches a “transition” color
the two colors recorded above (steps 13and 14).Swirl gently between
midwaybetwe
n
drops to avoid overshootingthe transition color.Recordthe number of drops of HCI
required and the color in the data table.Note: It may be necessaryto add half a drop
ata time.
17. Washthe contents of the test tube downthe drain with excesswater and rinse with dis
tilledwater.
FlinnChemTopic”Labs
—
Equilibrium
Page 5- ExploringEquilibrium
“7
Name:
Class/LabPeriod:
ExploringEquilibrium
Data Table
Part A. Complex-IonEquilibriumof Iron(III) and ThiocyanateIons
Colorof KSCN
Solution
Colorof Fe(NO
3)
Solution
Test tube 1
•C9lorofcontrolsolution (step5)
Test tube 2
Colorafteradditionof Fe(N0
3)
Test tube 3
Colorafteradditionof KSCN(step7)
Test tube 4
Colorofsolutionafter cooling(step8)
Test tube 5
Co!orofsolutionafter heating (step9)
Test tube 6
Co]orafteradditionof 4NaH
2PO
,•
(step6)
,
(step10)
Equilibriumof BromcresolGreen
Part B. Acid—Base
Initialcolorof indicatorsolution (step12)
ColorafteradditionofHCI(step13)
Colorafteradditionof NaOH(step14)
Numberof drops ofNaOHadded(step14)
Amountof HCIrequiredto obtain“transition”color (step 16)
Transitioncolor (step16)
Post-Lab Questions (Usea .eparate sheet of paperto answer the followingquestions.)
1. Writethe chemicalequation for the reversiblereaction of iron(1II)ionswith thiocyanate
ions in Part A.Labelthis Equation A.Usethe informationin the data table to write the
color of each reactant and product underneath its formula.
2. Howdid the color of the solution in Part A change when additionalreactant—either
in step 6 or KSCNin step 7—wasadded?Explain the observed
color changes:
3)
Fe(N0
Addingmore reactant to an equilibrium mixture of reactants and products increases
the rate of the (forward/reverse)reaction and thus (increases/decreases)the amount
of product.
3. Howdo the results obtained in steps 6 and 7 demonstrate that both reactants and prod
ucts must be present at equilibrium?
4. Howdid the colorof the solution in Part A change when it was cooled (step8) or heated
(step9)? Howdo these results demonstrate that the reaction shownin EquationAdoes
ExploringEquilibrium
Exploring Equilibrium Page6
—
indeed occur in both the forwardand reverse directions?
ions combinedwith iron(III) ionsand removedthem from solution.
5. In step 10, 4-H
2P0
Howdid the color of the solutionin Part A change when 4NaH
was added?Explain
2PO
the observedcolorchange:Removingone of the reactants from an equilibriummixture
of reactants and products decreasesthe rate of the (forward/reverse)reactionand thus
(increases/decreases)the amount of product.
I
6. Afterobservingthe effectof 4NaH
on the equilibriummixture in step 10,a student
2PO
3 and SCN-ions were still present in solution. Suggest additional
doubtedthat both Fe
couldbe
doneto provethat both reactants are still present at this point.
experimentsthat
7. Writethe chemical equation for the reversiblereactionof bromcresol green with water in
Part B. Labelthis Equation B.Hint: Referto Equation3 in the Background section.
8. Usethe color changes observedfor the indicatorbeforeand after adding HCI(steps 12
and 13)to predict the colorsof the HIn and 1n formsofbromcresol green. Writethe col
ors of HIn and 1n underneath their formulasin EquationB. Explainyour reasoning.
vard pr
Hint: AddingHC1increasesthe concentration of H ions.Which reaction, for
I
reverse,wouldthat increase?
9. Explain the observedcolor change:Addingmore productto an equilibrium mixture of
reactantsand products increasesthe rate of the (forward/reverse)reaction ana thus
p
(increases/decreases)the amount of product.
10. In step 14,hydroxideions reactedwith and removedW ions from solution (seeEquation
4 in the Background section).Whatcolor changewas observedwhen NaOHwas added?
Explain the observedcolor change:Removingone ofthe products from an equilibrium
mixture of reactants and products decreasesthe rate ofthe (forward/reverse)reaction and
thus (increases/decreases)the amount of product.
11. Whatform(s)of the indicator were most likelypresent when the transition color was
observedin step 16?Howdoesthis observationprovidevisual proof that not all reactions
“go to completion?”
FlinnChemlopic”Labs
—
Equilibrium
Date___________
Class
Name
_________________________________
•
____________
and
ChemicalEquilibrium
Principle
Le Chatelier’s
Lab3
Textreference:Chapter18. pp. 524—527
Pre-Lab Discussion
In most of the chemical reactions you have studied so far, at least one of
the reactants has been “used up.” The point at which a reactant is used
up marks the end of the reaction, and the reaction is said to have “gone
to completion.” Under ordinaiy circumstances, the prOduct(s) of such
reactions are not able to react to re-form the original reactants. Thus,
these are “one way” reactions. They proceed in one direction only.
Many other chemical reactions do not go to completion. Rather, the
products of these reactions remain in contact with each other and react
to re-form the original reactants. Such reactants are said. to be reversible.
In a reversible reaction, the forward and reverse reactions proceed at the
same time. When the rates of the twç reactions are equal, a state of
chemical equilibrium is said to exist. Under such conditions, both the
forward and reverse reactions continue with no net change in the quan
tities of either products or reactants.
A state of equilibrium is affected by concentration and temperature
and, if gases are involved, by pressure. If a system at equilibrium is sub
jected to a change in one or more of these factors, a stress is placed on
the system. According to Le Chatelier’s principle, when a stress is placed
on a system at equilibrium, the equilibrium will shift in the direction that
tends to relieve the stress. Equilibrium will be reestablished at a different
point, that is, with different concentrations of reactants and products.
In this experiment, we will study two equilibrium systems. The equilib
rium equation for the reversible reaction of the first system is:
3
Fe
+ SCN
(light brown)
±
Fe 2(SCN)
(red)
The addition of any substance to the system that increases the concentra
3
tion of Fe
or SCN will favor the forward reaction. This will cause the
shift to the right. The addition of any substance that de
to
equilibrium
of these ions will have the opposite effect.
the
concentration
creases
•The equilibrium equation for the second system is:
422CrO
(yellow)
+
3O
2H
±
207Cr
(orange)
+
20
3H
The addition of an acid to this system increases the H
concentration
3O
and causes the equilibrium to shift to the right. The addition of any sub
concentration will have the oppo
3O
stance that causes a decrease m H
site effect.
By studying these two systems, you should achieve a better under
standing of equilibrium systems and their responses to stress.
175
Purpose
•
is
Study equilibrium systeI
Le Chatelier’s principle.
and their responses to stress
described by
Equipment
beaker, 100-mL
graduated cylinder, 10-niL
test tubes, 13 X 100-mm (5)
test tube rack
•
Materials
:
0.1 MFeCI
3
0.1 MKSCN
I,
0.IMKC
distilled water.
•
Safety
6
•
dropper pipet
marking pencil
safety goggles
lab apron or coat
0.1 4MK
2CrO
0.1 2O
Cr
7MK
I
L0MHC
1.0M NaOH
fl A
Handle the HCI and NaOH solutiOns with care. They are corrosive sub
stances and can injure .the skin or eyes. Flush any spills with cold water
and report tFiem to your teacher. Note the caution alert symbols here and
with certain steps in the “Procedure.” Refer to page xi to review the
precautions associated with each symbol. Always wear safety goggles and
a lab apron or coat when working in the lab.
Procedure
PARTA
1. Using a marking pencil, number four test tubes 1 through 4
and stand the tubes in a test tube rack.
2. Measure out 5 mL of 0.1 M FeCl
and pour it into a 100-mL
3
beaker. Add 5 mL of 0.1 M KSCN to the same beaker. Dilute the
contents of the beaker with distilled water until the solution is
a light reddish-orange color. Divide the solution equally among
the four numbered test tubes. Set test tube I at one end of the
rack to be used for color comparison.
3. Using a dropper pipet, add 0.1 M FeCl
3
drop by drop to the
solution in test tube 2 until a color change occurs: Record your
observations in Part A of “Observations and Data.” Rinse the
pipet.
4. Repeat step 3, but instead of FeCI
add the following solu
3,
tions drop by drop to the test tube indicated. Rinse the pipet
after each use.
0.1 M KSCN
test tube 3
0.1 M KCI
test tube 4
Record your observations.
5. Discard the solutions. Wash and rinse the test tubes and
invert them in the rack to drain.
PART B
6. Using a marking pencil, number four test tubes 5 through 8.
Stand the tubes in a rack.
176
Name
34
andLeChatelier’s
ChemicalEquilibrium
Prindple
(continued)
A
A
Pour 5 mL each into
7. Measure out 10 mL of 0.1 M 4.K
2CrO
test tube 5 and test tube 6. Rinse the graduated cylinder and
Diyide this equally between
Cr
7.MK
measure out 10 mL of 0.1 2O
test tube 7 and test tube 8.
8. Using a dropper pipet, add 1.0 M HCI drop by drop to test
tube 5 until the color changes. Record your observations.
9. Repeat step 8 with test tube 6. As soon as the color changes,
rinse the pipet and use it to add 1.0 M NaOH drop by drop to
the solution until the color changes again. Record your observa
tions for this step.
10. Using the pipet, add 1.0 M NaOH to test tube 7 until the
color changes. Record your observations.
11. Repeat step 10 with test tube 8. As soon, as the color
changes, rinse the pipet and use it to add 1.0 M HC1 to the
solution until the color changes again. Record your observations.
Observations and Data
PARTA
Color
test tube 2
test tube 3
test tube 4
PARTB
Color Change
test tube 5
test tube 6
test tube 7
test tube 8
—
Conclusionsand Questions
1. Write equilibrium equations for the reversible reactions that take place
m Part A and Part B.
177
to the
3
:2. Using Le Chatelier’s principle, explain how the addition of FèC1
solution in test tube 2 (6tep 3) affected the equilibrium that existed in the
of the other
solution,. Give similar explanations for the addition of ech
substances (step 4).
J
:‘
H
:
3. Using the equilibrium equation for the reaction and Le Chatelier’s prin
ciple, explain the color changes noted in Part B.
S
178
Page 1 Gas PhaseEquilibrium
-
Gas Phase Equilibrium
Pressure and Temperature
Introduction
Manyimportant reactionsthat take place in the atmosphere involveequilibrium concentra
tions of gas phase reactants and products. Whatvariablesaffectthe position of equilibrium
for reactions in the gas phase?
Concepts
I
chemical equilibrium
•
tlier’s
LeCh
Gas-phasereactions
•
Nitrogen oxides
principle
Background
Burning fossilfuelsfor energy “drives”our societyand our economy.It is also a major
source of environmentalconcerns and challenges.The releaseof large amounts of carbon
dioxidefrom the combustionof oil and gas, for example,is a subject of controversybecause
of its pos
contribution to globalwarming. In addition to carbon dioxide,burning fossil
ible
fuelsalso producesa variety of sulfur and nitrogen oxides.Sulfur oxidesare formedvia the
•oxidationof sulfur-containingimpurities in coaland oil (Equations 1 and 2) and are a major
cause of acid rain. Nitrogen oxidesare formedwhen nitrogen and oxygen—themain compo
nents of air—combinewith one another incar engines, power plants, orin car exhaust
(Equations 3 and 4). Nitrogen oxidesare a major component of photochemicalsmog and air
pollution.
S(s) ÷ 02(g)
2(g)
2S0
2(g)
N
2N0(g)
+
—‘
02(g)
2(g)
S0
Equation 1
3(g)
2S0
Equation 2
+
02(g)
:
2N0(g)
Equation 3
+
02(g)
t
2(g)
2N0
Equation 4
As can be seen froth Equations 2—4,most of the gas phase reactions that take place in the
atmosphere are reversiblereactions. Conditionsthat affectthe position of equilibrium for
gas phase reactions are therefore of enormous importance in determining the environmen
tal impact of burning fossilfuels. In this experiment,we will consider the properties of
nitrogen dioxideand investigate howthe principles of equilibrium applyto its reactions.
is a toxic, reddish-browngas with an irritating odor.It is primarily
Nitrogen dioxide(NO
2)
responsiblefor the brownishhaze that hangs over many of the world’slargest cities due to
air pollution. Nitrogen dioxideis also quite reactive.In the presence of sunlight, for exam
ple, it undergoesa light-induced “photochemical”reaction to produce ozone (Equation5).
High levelsof nitrogen oxidesin the atmosphere are associated,therefore, with high ozone
levelsas well.
light
<
+
Equation 5
2(g)
N0
2(g)
O
3(g)
> NO(g) + 0
GasPhaseEquilibrium
Gas Phase Equilibrium Page 2
—
The high reactivityof nitrogen dioxidemeans that it reacts even with itself—twomoleculesof
which is a colorlessgas at room
204,
2 combine to form the “dimer,”dinitrogentetroxide,N
NO
reversiblereaction
and quicklyreaches a
Formationof
is
(Equation6)
temperature.
a
204
N
position of equilibrium.The relativeamounts of N0
present at equilibrjumdepend
2and
204
N
LeChâtelier’s
and
Principle.
temperature, accordingto
on pressure
:
2(2N0
g)
Red-brown
Equation 6
4(Ng)
2O
Colorless
Experiment Overview
The purpose of this experimentis to study the effectsof changing the temperature and pres
in a sealedtube at equilibrium.LeChâtelier’s
sure on the relativeamounts of NO
4
2O
2 and N
Principlepredicts how a change in conditionswillaffectthe equilibrium for a reversible
chemicalreaction—thereactionwillshift in a directionthat tends to reducethe effectofthe
imposedchange. Theeffectof changingthe temperature dependsonwhether the reactionis
exothermicor endothermicas written, whilethe effectof changing the pressure dependson
number of gaseousmoleculeson the reactant versu product side of the reaction equation.
•the
Pre-Lab Questions (Show al/work on a separate sheet of paper.)
1. DrawLewiselectrondot structures fOrthe nitrogen oxidesmentioned in the Background
and dinitrogen tetroxide N
section:nitric oxideNO,nitrogen dioxideNO
2,
4
2O
2. Usethe electron dot structures ofNOand NO
2 to explainwhy these moleculesare consid
ered highly reactive.
3. Although both N
2 and 02 are naturallypresent in the air we breathe, high levelsof NO
and NO
2 in the atmosphereoccur mainly in regions with large automobile or powerplant
emissions.The equilibriumconstant for the reactionof N
2 and 02 to give NOis very
small.The reaction is, however,highlyendothermic, with a heat of reaction equalto
+180ki (Equation7).
2(g)
N
+
02(g)
+
180 kJ :
Equation 7
2N0(g)
(a) UseLeChâtelier’sPrincipleto explainwhy the concentrationof NOat equilibrium
increaseswhen the reactiontakes placeat high temperatures.
(b) UseLeChâtelier’sPrincipleto predict whether the concentration of NOat equilibrium
should increasewhen the reaction takes placeat high pressures.
Materials
2
Sealed,jumbo pipet bulbs containing NO
2,
Beakers,250- or 400-mL,2
Ice
Water
Hot plate
Thermometer
Forcepsor tongs
White paper (for background)
FlinnChemlopic” Labs Equilibrium
—
Page3- Gas PhaseEquilibrium
Safety Precautions
Nitrogen dioxideis a highly toxic gas. The gas willbe supplied in sealedpolyethylene pipèt
bulbs.Do not cut the pipet bulbs orpuncture them in any way. Do not continue with the
procedure if you see any breaks or tearsin the bulbs or if you see yellow stains on your
hands. Immediately take any leakingpipets to the fume hood and notify your teacher. Wear
chemical splashgogglesand chemical-resistantgloves and apron. Washhands thoroughly
with soap and waterbeforeleaving the laboratory.
Procedure’
1. Filltwo beakers(250-or 400-mL)half-fullwith tap water.Heat one beaker on a hot plate
to prepare a hot-waterbath (75—80
°C) for use in step 4. Addice to the secondbeakerto
for
ice-waterbath
(0—5
°
C)
an
use in step 5.
prepare
2. Measurethe roomtemperature and record it in the data table.
3. Obtaintwosealedpipet bulbs filledwith nitrogen oxidesfrom your teacher.Observeand
record the colorofthe gas at room temperature.
4. Usingforcepsor tongs, placeone pipet bulb in the hot-waterbath for 2—3n
inutes.
Measurethe temperature of the bath and observethe color ofthe gas. Recordthis data in
the data table.
‘
5. Useforcepsor tongs to remove the pipet bulb from the hot-waterbath, then immersethe
bulb in the ice-waterbath. Measurethe temperature of the bath and observethe color of
the gas. Recordthis data in the data table.
6. Alternate immersingthe pipet bulb in the hot-waterand ice-waterbaths.Arethe color
changesrepeatable?Recordall observationsin the data table.
7. Placethe bulb on a piece ofwhite backgroundpaper.Doesthe gas return to its original
colorwhen the bulb returns to room temperature?
8. Takethe secondpipet bulb and hold it verticallyat one end. Squeezeon the bulb and
bend it over to compressthe gas into a smallervolume. Tryto squeezethe gas into about
one-halfits originalvolume.
9. Observeand record any immediatecolor changes that occur when the gas is compressed.
10. Continue squeezingthe pipet bulb in this manner for 2—3minutes. Observeany further
colorchanges that may occur. Comparethe color of the gas againsta white background
with that in the first pipet bulb, which should be at room temperature.
11. Return all pipet bulbsto your teacher for disposal.
GasPhaseEquilibrium
Gas PhaseEquilibrium Page4
-
Name:
________________
Class/LabPeriod:
______
Gas Phase Equilibrium
Data Table
Effectof Temperature and Pressure on 204theN0
—N
Equilibrium
Room temperature
,
Colorof gas at room temperature
Temperature of hot-water bath
,
Colorof gas in hot-water bath
,
r
bath
Temperature of ice-wat
Colorof gas in ice-waterbath
Observations upon further
heating and cooling
Colorof as when
volume was initially reduced
,
.
S
,
Final color of gas after
volume was reduced
Post-Lab Questions
1. Writethe chemicalequation for the reaction of NO
2 to formthe dimer N
204.
color of each compoundunderneath its formula.
Includethe
2. Whatcolor changewas observedwhen the gas was cooled?In what direction did the
equilibriumshift?
3. Whatcolor changewas observedwhen the gas was heated?In what direction did the
equilibriumshift?
Flinn Chemlopic” Labs
—
Equilibrium
Page5- Gas Phase Equilibrium
4. Areboth reactantand product gasespresent in the uri
in;iJ
temperature?Explain.
uqiiilihrniiii
inixtui* ;il
ruuIll
5. Usethe results ofthe heating and cooling experimentsLudecide whether the dimeriz;
tion reactionof NO
2 is endothermic or exothermic.lcwrite the diemical equation for the
reactionto includethe heat term on the reactant or product side, as needed.
6. UseLeChâtelier’sPrincipleto explainthe effectof temperature on the gas phase equilibri
um involvingNO
2 and N
204.
7. Writethe equilibriumconstant expression(massaction expression)for th nitrogen oxide
equilibrium.Doesthe value of the equilibriumconstant dependon temperature?
8. Accordingto Boyle’sLaw,what happenedto the pressure insidethe bulb when the bulb
wassqueezedto halfits original volume?UseLeChâtelier’sPrincipleto predict how this
reaction.
—N
pressure change shouldaffectthe position of equilibrium for the 204N0
9. Discussthe color changes observedwhen the gas volume was reduced.Dothe changes
agree with the predictionmade abovefor the effectof pressure?
10. Whatother factorsor conditions might haveinfluencedthe color changes observedwhen
the bulb wassqueezed?Hint: Did any of the other gas variables (P,V,T,n) change?
GasPhaseEquilibrium
Name
Date
Class
20 Laboratory Investigation
S
Changes During Red ox Reactions
Problem
What changes occurduring redox reactions?
Materials (per group)
24 well-reaction plates
miaopipettes containing the following solutions:
1.OMHcI
1.0 M Cu(N0
3)2
3)2
1’.OMZn(N0
small pieces of Mg, Cu, Zn
Procedure
1. Place 10 to 12 drops of HO into each of the wells of three well-reaction plates. Add one piece
of Mg to one of the wells, Cu to a second well, and Zn to a third well. Record your observations
in the data table below.
2. Place 10 to 12 drops of Cu(N0
3)2
observations
in the
record
your
•
3. Place 10 to 12 drops of Zn(N0
3)2
record your observations in the
in a dean well. Add a piece of Zn to the well. After 5 minutes,
data table.
in a dean well. Add a piece of Cu to the well. After 5 minutes,
data table.
.
4. Following your teacher’s instructions, discard all solutions and clean all well-reaction plates.
Observations
Reactants
Observations
Substance
oxidized
Substance
reduced
HCI+Mg
HCI+ Cu
HCI+Zn
)2
3
Cu(N0
+
Zn
)2
3
Zn(N0
+
Cu
28
Chapter20
f_____
I
C -I4aII.
enti
P
Inc.
THEMIRRORE FLASK
PROCEbURE:
1. Add lOml of .25Mdextrosesolutionto a clean bottle.
of .1OMsilver nitratesolutionto a150m
1
1
beaker.
2. Add 30m
3. While stirring, add concentratedammoniumhydroxidedrop-wiseto the silver
nitrate solutionin the beakeruntilthe gray-biackprecipitateforms. Continue
adding ammoniumhydroxidedrop-wisewith swirling untilthe silver diammine
ionic complexforms and the solutipnJUST becomesclear and colorless.
‘4. Add 15m1Qf.80M potassiumhydroxideto the beaker. The silver hydroxide
splid usuallyprecipitatesagain,so add concentratedammoniumhydroxide
.
drop-wisewith swirling untilthe solutionjust clear
5.’ Pourthe contentsof the beakerinto your bottle and put the cap on. Swirl so
the liquid contactsthe entire innersurface. Continuefor five minutes.
6. Pourthe liquid into the waste containerand hnsethe bottle.
Describeall of the chemicalchangesthat you obsdrvedduring the lab. Write as
many chemicalreactionsas possibleto explain the observations.
What happenedto the silver during this process? Whatevidencedo you have
for this conclusion?
¼
Name_________________
Bayport- Blue Point High School
Chemistry Lab- Redox Unit
ACTWIT1E. Of METAL& RE.DOX
PURPOSE
The objective of this experiment is to rank in order sveral metals on the basis of their
chemical activity. The results can then be compared to the Activity Series in the
reference table to test for accuracy.
DISCUSSION
Active metals react with acids to produce hydrogen gas. The rate at which bubbles of
hydrogen are produced when various metals are placed in acid solution can be used to’
indicate the reactivity of the metals. In the reaction, the metal is oxidized while the .I{
from the acid is reduced to H
2(g). The most active metals are the most easily oxidized.
4MATERIALS
Chemicals
iron nail
galvanized nail
copper tack
aluminum nail
6 M HCI solution (500 mL concentrated HCJ solution diluted to 1.00 liter with
distilled or deionized water)
Eguirment
test tubes or smallbeakers
test tube rack
.
.TAZARDS
Concentrated solutions of HCI will burn skin and damage clothing. Fumes from acid can
be caustic and/or irritating; do this experiment in a well-ventilated area (hood, if
available). Goggles must be worn throughout this experiment.
Created by M.Romaine
Page 1 of 4
3/.
PROCEDURE
1. Place a different metal ineach of four test tubes
2. Add just enough 6 M HCI solution to cover the metal. Add the acid to each of the
test tubes at the same time so that you are able to compare the rates.
3. Record the relative activity of each metal based upon the rate at which hydrogen
is released.
4. Test the gas released by the galvanized nail with a burning splint to confirm that it
is hydrogen.
5. Rank the four metals from lowest to highest activity.
DISPOSAL
Dilute the acid with water and flush down the drain with water. Metal pieces can be.
discarded with solid waste.
.
.
I
i4TLPS
.
Some metals form an oxide coating on the surface that will prevent further oxidation;
these should be cleaned with steel wool before testing their reactivity. Even though’the
metals chosen forthis experiment may have an oxidd coating, they should give the
correct order of activity without cleaning because of the large differences in activity.
OBSERVATONS
How did each metal react in the presence of the acid?
METAL
OBSERVATION
I
Place the metals in order of reactivity (from least to greatest).
<
According to your Activity Series Reference Table, list the metals in order of reactivity
(least to greatest).
<
Created by M.Romaine
Page 2 of4
4.
0
POST
LAB QUESTIONS
1. Define:
I
OxidatiOn:
Reduction:
Oxidation
State:
________________________________________________________
Electrochemistry:
_________________________________________________________
ElectrolyticCeIU______________________________________________________________
VoltaicCell: ___________________________________________________________
the half-reactions for the reactions of copper & hydrogen, iron & hydrogen, and
aluminum & hydrogen: Be sure to include electrons afid oxidation numbers. in each
reaction, indicate which species is being oxidized and which is being reduced.
2.Wiite
COPPER & HYDROGEN
Half- reaction
Half. reaction
Net reaction
OXIDIZED______________
ON
REDUCED______________
& HYDROGEN
Half- reaction
Half- reaction
Net reaction
OXIDIZED______________
Created by M.Romaine
REDUCED______________
Page
3 of 4
tf
53
ALUMINUM & HYDROGEN
Half- reaction
Half- reaction
Net reaction
OXIDIZED______________
REDUCED_______________
1) Answer ALL parts of this question!
a) What is one problem associated with acid rain that, is illustrated by this
experiment?
b) What other problems are associated with acid rain?.;
c) Some metals are galvanized to prevent “rusting.” If zinc reacts with acid readily,
as discovered in this experiment, why is iron coated with zinc?
d) How could this hypothesis be tested in the laboratory?
.
a)
b)
c)
d)
Created by M.Romaine
Page4 of4
TheGold PennyLab
In this lab,youwill be convertinga regularpennyintoa “gold”penny. In doin
so, you arefollowinga traditionthat goesbackto theearliestdaysof chemistry.
Themodempracti e of chemistrystartedwiththe studyof alchemyin medieval
Europeandthe MiddleEast. Alchemistsbelievedthatby doingcertainchemical
reactions,you couldturncheapmetalsinto gold. Someof themdid, anyway
the otherswouldconvincepeoplethat they coulddothis andsellthemthe
secret. By thetime anybodyrealizedthatthe “secret”didn’twork,they’dbe long
gonewiththe money.
—
In this lab, you’llbe doinga reactionin whichcopperappearsto turnintogold.
We’lltalkaboutwhat reallyhappenslater.
Safety:
In this lab,’it is very importantthat you wearyourgogglesat all times,andwork
withthe chemicalsin thehood. Thechemicalsthat weare workingwith are
extremelydangerous,and cancauseseriousinjuryif an accidentoccurs!
“
Experiment:
Takea pennyanddropit intothe boilingsodiumhydroxideandzinc
1)
t for abouttwo minutes.
mixture. Le it s1
2)
Pullthe pennyoutof thewaterwith a pair,of crucibletongsandlet it cool
and dryon a papertowel. DO NOT’TOUbHTHE.PENNYWITHYOUR
BAREHANDS!
Recordthe appearanceof the pennyhere:
3)
Withthe crqcibletongs,holdthe pennyoverthe Bunsenburneruntilyou
observea colorchange. Oncethe colorhas changed,immediately
removethe pennyfromthe heatand let it cool on the barebenchtop.
Don’tput it on a papertowel the papertowelwouldcatchfire.
—
‘—Recordthe appearanceof the pennyhere:
What happened?
Whathas happenedis thatthe zincthat was boilingin the sodiumhydroxide
formeda thin layerovertheoutsideof the penny. Whenthe pennywas heated
overthe Bunsenburner,the silveryzinc coatingmixedwiththe copperof the
pennyandcausedthe pennyto turn “gold”.
© 2000 CavalcadePublishing
For chemistry help, visit www.chemfiesta,corn
Basically,youformeda mixtureof zincand coppercalledbrass. Brassis a metal
that hasfrequentlybeenconfusedfor gold,especiallybypeoplewhodon’tsee
gold very much.
Repeatthe experiment:
Dothe experimentoveragain,exceptthistime changesomethingaboUtthe way
you did it. Changeonly onething! Hereare somesuggestionsaboutwhat’you
mightwantto try:
Leavethe pennyin thesodiumhydroxide/zinc
solutionfor a different
of
time
length
Heatthe pennyfor a differentlengthof time overthe Bunsenburner
Trycleaningthe pennyoff withwaterbeforeheatingit.
-
-
In the sectionbelow,writedownwhathappenedin yOursecondexperiment
:
Whatdid you do differentlythe secondtimethanthefirst?
Here’swhat happenedthe secondtimethatdidn’thappenthefirst time:
Questions:
What’san alloy? Is brassan alloy,or is it different?Whatdo youthink,
1)
and why?
2)
Couldyou turnothermetalsinto gold if you didthe right reaction?Whyor
why not? You mightwantto searchfor informationaboutalchemyto
answerthis question.
3)
Why do you think you gota differentresultthesecondtime you did the
experiment?Explainwhatyou think happened.
© 2000 Cavalcade Publishing
For chemistryhelp, visit www.chemfiesta.com
Date
Name
_________________________________
•
Class
___________
of Iron
Corrosion
____________
Lab
44
Text reference: Chapter 22. pp. 655—660
Pre-Lab Discussion
Corrosion is a redox reaction in which a free metal is oxidized, or “cor
roded,” ‘•by some oxidizing agent. In nature, the oxidizing agent is fre
quently atmospheric oxygen dissolved in water. Perhaps the most familiar
free iron, Fe,
,
example of corrosion is the rusting of iron. In this reactioi
3
2
and Fe
ions. In the reduction half-reaction, oxygen,
is oxidized to Fe
ions.
°2, reacts with water to form 0H
The rusting of iron usually is considered to be a destructive change,
and considerable time and money are expended to prevent it. One method,
‘which you will observe in this experiment, makes use of a “sacrificial”
metal. If iron is placed in contact with a more active metal (one that is
more easily oxidized), the more active metal will be corroded instead of
the iron. Metals situated below iron on the table of standard electrode
potentials can function as sacrificial metals.
In this experiment, indicators will be used to signal the formation of
2
Fe
ions and 0H ions as products of the corrosion of iron. One such
2
indicator, the hexacyanoferrate(1Il) ion, turns blue in the presence of Fe
of 0H ions.
ions. The other, phenolphthaiein, turns pink in the presen
This experiment should help promote a better understanding of corrosion
and of oxidation-reduction reactions.
‘p
Purpose
Observe the corrosion of iron and investigate conditions related to corro
sion.
Equipment
balance
burner
graduated cylinder, IOO-mL
beaker, 250-mL
ring stand
iron ring
wire gauze
glass stirring rod
dropper pipet
petri dishes (2)
steel wool or emery paper
metric ruler
safety goggles
lab apron or coat
MateriaJs
0.1 M 6K
(potassium hexacyanoferrate(IlI)]
3Fe(CN)
copper wire, 5-cm
agar powder
zinc strip, 5-cm
1%phenolphthalein solution
iron nails (4)
distilled water
•
Safety
Observe all general lab safety precautions. Always wear safety goggles
and a lab coat or apron when working in the lab.
227
Procedure
1. Set up a ring stand, ring, and wire gauze. Add 200 mL of
distilled water to the 250-mL beaker and heat 1* boiling. While
the water is being heated, clean four iron nails with steel wool
or emery paper.
2. Using the balance, measure out 2.0 g of agaz powder. When
the water is boiling vigorously, put out the flame and slowly add
the agar to the water, stirring constantly. When the agar has
Rinse the dropper
dissolved, add 10 drops of 0.1 M N)
3Fe(
6.K
pipet and add five drops of phenolphthalein solution.
3. Place one. bent iron nail and one straight iron nail into one
of the petri dishes. Be sure the nails do not touch and are
positioned as hbwn in Figure 44-1(a).
1
Figure 44-
$
e of the remaining nails with copper wire and
4. Tightly wrap on
the other with the inc;strip. Place these nails in the other petri
dish, as shown in Figure 44-1(b).
5. When the agar solution is cool enough, pour it into each petri
dish to a depth of 0.5 cm. Make and record observations in the.
time remaining. At the end of the lab period, place the dishes in
your drawer to be observed on the following day.
Observationsand Data
Petri dish (a):
Petri dish (b):
228
Name
44
Corrosionof iron (continued)
and Questions
Conclusions
1. Explain why corrosion occurred at the sites indicated by the blue color
inpetri dish (a).
2. What is the whitish material surrounding the zii
cwrapped
iail in petri
dish(b)?
the zinc-wrapped nail showed no evidence of corrosion,
3. Explain w
y
while the copper-wrapped nail did.
.
.
4; .Woulda magnesium wrapping prevent the corrosion of iron? Explain.
5. Why would sodium metal not be appropriate as a sacrificial metal in
this experiment?
.
229
Unit Four: Thermodynamids and Kinetics
354
4.1.3 CHEMICALHAND-WARMER
Concepts to investigate: Exothermic reactions, rusting, corrosion, oxidation.
Materials: 100 mesh iron powder (unoxidized electrolytic powder, 100 mesh or finer), ver
miculite (from garden store), salt, plastic ‘bag, twist-tie.
Safety: Wear goggles and lab coal.
p
‘
Principles ann’ Procedures: Frostbite is a condition that results from, the formation of ice
crystals in body tissues. Circulation is reduced drastically, and tissues become numb and turn
a grayish blue. Gangrene, the death of tissues due to lack of oxygen, will set in if blood flow
ceases. Those who work outdoors in cold climates’often carry chemical hand-warmers to pre
vent frostbite in emergency situations. In this activity you will make your own hand-warmer.
Mix’25 g of unoxidized iron.powder (iron powder must be gray or black,’not red or
and 5 g of vermiculite together in a small plastic
chlàridé)
brown), I g of table salt (sodium
bag that is.supported by a beaicer (Figure E). When all the materials are evenly distributed,
add approximately 5 mL of water, remove all the air and seal the bag. To activate your handwarmer, open the bag and allow oxygen to enter. Gently knead the bag to mix the contents
and note any changes in temperature (Figure F). Unoxidized iron rustsrapidly when exposed
to salt water and oxygen, generating a substantial amount of heat:
4Fe(s) + 30
2(g)
le
LVI=-1652kJ/mo
2’O.
2Fe
(s)
” your hand-warmer by removing the air from the bag and re
Is it possible to “turn off
sealing it? Try it and monitor the temperature of the bag for 10 to 15 minutes. What happens
if you once again open the bag and allow air to re-enter?
What is the role of sodium chloride in this reaction? Repeat the procedure described
above, but,without the salt. Compare this reaction to the previous one. Does sodium chloride
facilitate the oxidation of iron?
25 Fe powder
I NaCI
5 vermiculite
5 ml. H,O
E
4Fe(s)
+
2(g)
30
—,
3
2O
2Fe
(s)
H= -1652k)
F
Questions
(1) Is this an exothermic reaction? How do you know?
(2) ‘Why is it necessary to open the hand-warmer bag to start‘thereaction?
• 4.1 Thermochemistry
S
355
(3) What effect does removingthe,air and sealingthe bag have upon the temperatureof
the hand-warmer?
(4) Does sodium chloridefacilitatethis reaction?Explain.
(5) Salt is often added to melt ice on streets. Driverscomplaiflthat this salt causes their
cars to rust. Is this a valid complaint?Explain.
G
I
U
•
I
--
II
‘In
,
>
l.
ni
S
I
S
Page1 Propertiesof Acidsand Bases
—
Properties of Acids and Bases
Idenflfkation.and CIassificat
oñ
Introduction
Acidsand basesare useful reagents in the chemistrylaboratoryand playan important role in
biologyand nature. What are acidsand bases?What propertiescan be used to distinguish
acidsand bases?Let’sexplorethe properties of acidsand basesand identifythe characteristic
featuresthat willallowus to classifysubstances as acidsand bases.
Concepts
• Acidsand bases
•
Activemetals
•
pH Scale
• Indicators
•
Conductivity
•
Neutralization
Background
4he origin of
The word acid is derivedfrom the Latinverb acerewhich means “to (be)sour.”‘l
the word acid revealsa characteristic physicalproperty of acids—theytaste sow Lemons,
oranges, and grapefruitsare calledcitrus fruits because they contain citric acid, an acidic
compoundwhichgivesthem their sour taste. Althoughtaste is an interestihg property of
the foodswe eat, it is NOTa property that we will use in the chemical.laboratoryto classify,
compoundsas acids or bases.The followingpropertiesare typicallyused toclassify com
pounds as acids or bases.
Conductivity. Someacidsand bases ionizecompletelyinto ions when dissolvedin water.
Solutions that contain large numbers of dissolvedions conduct an electric current and are
called electrolytes.Other weakeracidsand bases may ionize only partiallywhen dissolvedin
water and may conduct electricityonlyweakly—theyare calledweak electrolytes.
Substancesthat do not produce dissolvedionswill not conduct electricityand are called
nonelectrolytes.
Effect on Indicators. Indicators are organic dyesthat change color in acidic or basic solu
tions. One of the oldestknownacid—baseindicatorsis litmus, a natural dye obtained from
lichens. Its use was describedas early as the sixteenth century. Litmus paper,prepared by
soaking paper in a solution of the dye, is often used as a general test for acids and bases.
Phenolphthaleinis another indicator that shows a color change as solutions change from
acidic to basic. Althoughthese indicators are usefulfor broadlyclassifyingsubstances as
acids or bases,they are not able to distinguish among differentlevelsof acidityor basicity.
Byusing combinationsof different indicators,however,it is possibleto obtain a spectrum of
color changesovera wide range of acidity levels.Universalindicator and pH paper are two
products that use combinationsof indicators to rank substances from most acidic to least
acidic, or most basic to least basic.
ThepH Scale. The pH scaleis a numerical scalethat is used to describethe relativeacidityor
basicityof a solutionand is relatedto the concentrationof H
3O ions.TheabbreviationpH
stands for “powerof hydrogen”—adifferenceof one unit on the pH scale correspondsto a
3O ions.Pure water contains extremely
poweroften differencein the concentrationof H
PropertiesofAcidsandBases
Propertiesof Acidsand•ases
—
Page2
small, but equal,concentrationsof H
3O and0H ionsdue to self-ionization(Equation1).
Acidsahd’basesare substancesthat alter the cöñëëntrationsof H
3O and OH-ions in solution.
2O(l)
H
+
20(I)
H
‘—
aq)
3O
H
+
OW(aq)
Equation 1
Reaction with Metals.Acidsreact with so-calledactivemetals—reactivemetals such as mag
nesium and zinc—toproducehydrogengas and solutionsof metal ions.The reactionof dif
ferent metalswith acidsis a well-knowntest used to rank metals from most activeto least
active.Reactionof a single activemetal with a varietyofdifferentsolutions is one of the best
methods to identifyacidsand to compare their relativeacidity.
Neutralization reactions. Acids‘andbasesreact‘witheach other to giveneutrql products—
solutions that are neither acidic nor basic.The products of neutralization of an acid and a
base are an ionic compound(generally,referred to as a salt) and water.The amount of acid
that will react with a specificamount of base is governedbystoichiometry.Neutralization
reàctiôns are frequentlyused In the laboratoryto determinehow much of an acidic or basic
compoiind is present in a substance.
Experiment Overview
The purpose ofthis cperiment is to explore’the propertiesof aqueous solutionsand to classify
them as acidic,basic,or neutral. The resultswillbe usedto developworkingdefinitionsand to
analyzethe pH scalefor identifyingacidsand bases.
Pre-Lab Questions
‘
,
Acidwit, acid rain, and an acid test—these familiar expressionssuggest some interesting
properties and uses ofacids.
1. The phrase acidwit is definedin the dictionaryas humor that is sharp, biting, or sour in
nature. In the laboratory,acids present a hazard becausethey are corrosive.Whatis
meant by the term corrosive?Howdoesthis relateto acidwit?
2. Acid rain is recognizedas a growingdanger to the environment. Brieflydescribetwo
problems associatedwith acid rain.
3. (Optional)The phraseacid test has entered the popularvocabularyto describea severebut
conclusivetest ofwhether something is authentic. Whatis the origin of the term acidtest?
Materials
Aceticacid solution, CH
3COOH, 0.1 M, 6 mL
Ammoniasolution, NH
0.1 M, 4 mL
3,
Hydrochloricacid solution, HCI,0.1 M, 6 mL
Magnesiumribbon or turnings, Mg, 5 pieces
Phenolphthaleinsolution, 0.5% in alcohol, 2 mL
Sodium hydroxidesolution, NaOH,0.1 M, 8 mL
Universalindicator, 1 mL
Washbottle and distilled or deionizedwater
Conductivitytester
Forceps
Litmus paper, neutral, 5 pieces
pH test strips, wide range
Pipets,Beral-type,5
Reactionplate, 24-well
Stirring rod
White paper (for background)
Flinn ChemTopic Labs
—
AcidsandBases
Page3—Propertiesof Acidsand Bases
Safety Precautions
0
All of the acidsand basesused in this lab are corrosiveto eyes, skin, and other body tissues.
They are toxic by ingestion.Avoid contact of all chemicalswith eyes and skin. Notify your
teacherand clean up all spills immediately with largeamounts of water.Magnesium metal
’ne. Keep away from flames.
is a flammablesolidand burns with an intense fla
solution and is flammable.It is moderately toxic by
based
Phenolphthaleinis an a1cohoi
from
flames
other ignitionsources. Wearchemical splash goggles
and
ingestion.Keep away
and chemical-resistantgloves and apron. Washhands thoroughly with soap and water
before leaving the laboratory.
Procedure
•Part A. Classifying Acids and Bases
1. Obtaina 24-wellreaction plate and place it on a piece ofwhite paper as shownbelow.
Notethat eachwellis identifiedby a unique combinationof a letter and number, where
‘the letter refersto the horizontal row and the number to the verticalcolumn.
•
•
2. Labelfivepipets 1—5and fillthem with solution,as shown below.
Label
Solution
1
Hydrochloric
Acid
2
Acetic
Acid
3
Distilled
Water
4
Ammonia
5
Sodium
Hydroxide
3. FillwellsA1—A5
in RowAabout two-thirdsfullwith the correspondingsolutions 1—5.
Note: Thewellsin a 24-wellreaction plate have a 3-mL capacity.Addabout 2 mL (40
drops) ofsolutionto eachwell.
4. Testeach solutionin RowA (wellsA1—A5)
using a conductivitytester. Describeeach solu
tion as a strong conductor,weak conductor,or non-conductorand record any additional
observationsin DataTableA.Rinsethe conductivitytester with distilledwater and wipe
clean betweeneach test.
5. Testeach solutionin RowA (wellsA1—A5)
using a piece of neutral litmus paper. Record
the color ofthe paper in DataTableA.Note: Dipa stirring rod into the solution, then
Properlics of Acidsand Bases
Properties of Acids and Bases Page4
-
touch the stirring rod to the test paper.Whenused in this way,one test paper may be
used for more than one solution.Be sure to wipe the stirring rod clean before testing
each new solution.
Recordthe color of each solu
6. Add1 drop of phenolphthaleinsolution to eachwellA1—A5.
tion in DataTableA.
in RowB.
7. Add20 drops of solutions 1—5to the correspondingwellsB1—B5
8. Testeach solution in RowB (wellsB1—B5)
using a pH test strip. Usethe color chart on
the pH paper containerto assigna numerical pH value to each solution. Recordthe pH
valuefor each solution in DataTableA.
Recordthe color of each solution in
9. Add1 drop of universalindicatorto eachwellB1—B5.
DataTableA.
in RowC.
10. Add20 drops ofsolutions 1—5to the correspondingwellsC1—C5
11. Addone small piece of magnesium metal to each solutionin wellsC1—C5.
Observeany
that
takes
of
if
reaction
he
speed
placend compare
reaction, any, in each well.
apparent
Recordall observationsin DataTableA.
Reactions of Acids and Bases
12. Carefullyadd 20 drops of hydrochloricacid (solution 1), followedby 1 drop of,phenolph
thalein, to eachwellDl and D2.
Part B. Neutralization
13. Carefullyadd sodium hydroxide(solution5) one dropat a time to the solution in wellDl
until a stable color change occurs. Stir the solution in betweendrops. Recordthe number
of drops of sodium hydroxideadded (Trial1) in DataTableB.
14. Add10 drops of distilledwater to the solution in wellD2. Repeatstep 13to test the solu
tion in wellD2.Recordthe number of drops of sodium hydroxideadded (Trial2) in Data
TableB.
15. Carefullyadd 20 drops ofaceticacid (solution2), followedby 1 drop of phenolphthalein,
to each wellD5and D6.
16. Carefullyadd sodium hydroxide(solution 5)one dropat a time to the solution in wellD5
until a color change occurs. Recordthe number of drops ofsodium hydroxideadded
(Trial1) in DataTableB.
17. Add10 drops of distilledwater to the solution in wellD6.Repeatstep 16to test the solu
tion in wellD6.Recordthe number of drops of sodium hydroxideadded (Trial2) in Data
TableB.
Disposal
18. Usingforceps,remove ny piecesof unreacted metal fromwellsC1—C5.
Disposeof these
metal piecesas instructed by your teacher. Rinsethe contents of the reaction plate down
the drain with plenty of excesswater.
FlinnChemTopicLabs AcidsandBases
—
Page5- Properhesof Acidsand Bases
•
‘I
_____________________________________
Name:
Class/LabPeriod:
Propertiesof ‘Acidsand Bases
Data Table A. Classifying Acids and Bases
Solution
1
2
3
4
5
Hydrochionc
Acid
Acetic
Acid
Distilled
Water
Ammonia
Sodium
Hydroxide
•.
Test Property
Conductivity
.
Litmus Paper
,
‘
lein
Phenolphtha
pH Test Paper
‘
.
,
UniversalIndicator
Reactionwith
Magnesium
‘
.
,
Data Table B. NeutralizatIon Reactions of Acids and Bases
HydrochloricAcid
Numberof Drops of Sodium
HydroxideAdded (Trial 1)
Numberof Drops of Sodium
HydroxideAdded(Trial 2)
Propertiesof Acidsand Bases
AceticAcid
Properties of Acids and Bases
Priqc
6
24S
Post-Lab Questions (I’c
I.;’/)I)(/I(
:I-o/pa/.er
.cIl
to answer the followingqu
stions.).
Ii e cii tduct ivitvtest to identifyeachsolution in Part A as a strong elec—
I. UseIhe resuIt:
dk
electmivic, OF nonelectrolyte.
trolvtc, W(
2. Whichsoluhoiis in Uart A reactedwith magnesium metal?Writea balancedchemical
equationbr the reactionof eachacid in Part Awith magnesium.
3. Strongacidsionize completelyin water to form ions and are thus strong electrolytes.In
contrast, weak acidsdo not readilyionizein water—infact,lessthan 1%of the mole
cules are probablyionizedat any given time. Weakacidsare thereforeveak electrolytes.
Classifyeach acid as either a strong or weakacid.Whichreactedfasterwith magnesium
metal, the strong acid or the weakacid?
4. Writechemicalequationsfor ionizationof the strong and weakacids in water.Identifythe
common ion that is produced in acidic solutions.
5. Howcan litmus paper and phenolphthaleinbe used to tell whether a solution is an acid
or a base?Be specific.
6. Usethe combinedresults of the conductivityand indicatortests to identifythe basicsolu
tions in Part A. Classifyeach as a strong versus weak base.
7. Writechemicalequations for the ion-formingreactionsofthe strong and weak base n
water.Identifythe common ion that is produced in basicsolutions.
8. Comparethe pH data for the solutionswhich you labeledas acidsand bases.What pH val
ues can be assignedto acidsand bases,respectively?
9. Comparethe pH valuesof strong versus weakacidand strong versus weak base solutions.
Howdoes pH varywith the “strength” of an acid or base,respectively?
10. Explainthe color change observedfor the indicator in the neutralization reaction of
hydrochloricacid in Part B.Whatis the pH range of the finalsolution?
11. (a) Write separate, balancedequations for the neutralizationreactionsof hydrochloric
acid and aceticacidwith sodium hydroxide.
(b) Usethe stoichiometryof the balancedchemical equationsto explainthe number of
drops ofsodium hydroxiderequired for complete neutralizationof the acids.
(c) Didthe strong and weakacids require equal number of drops of sodium hydroxide?
Explain.
(d) Did addingwater to the acidsolutions change the number of drops of sodium hydrox
ide required for neutralization?Explain.
12. Completethe followingtable to summarize the properties ofacidsand bases.
Acids
Property
Bases
Conductivity
Litmus
Phenolphthalein
Reactionwith metals
.
pH
FlinnChemTopicLabs AcidsandBases
—
NAME______________________
DATE_____________
ACID, BASEINDICATORLAB (TableM)
Purpose: Your job is to find out how each of thhe indicafors reacts or changcs in soh.tons of various ph.
Materials:
30 small plastic cups
methyl orange
bromthyrnol blue
lein
phenolphtha
bromcresol green
thymol blue
various pH solutions (4-10)
Pr’ocedure:
Read all of the directions in the procedure before you begin.
1. Label the plastic cups according to the data chart below.
2.
Fill each cqp with 10 drops of the correct pH solution (4, 5, 6, 7, 8, 9, & 10) fEomthe labeled
bottles.
3. Predict the color of the solution in each cup based on Table M.
h
of ea
ops
4. Test each of the solutions h adding 2-3.d
indicator in the apropriate
blank in the’chart.
5. Put the color you see the indicator change to n the cnect
1hL
Common
I fl(liu.
II
1
Acid—Base Indicators
hitV
A It I\Jt
,,
I
Color Change
)I I It.IuI!.:(
( .‘ii,i-(.li;ii
t
ii
fl)(’tII\
Ot
I)lOfl
II.
)Iii
J)l
o.
oIj)IIt:
”
‘(1lu
-
iihorIec’
’:
Ii
iitniu’
.—
co
oflcr
l)rO
—
thvrno)I
((Ito IIo
v
—
re(l
.
)tu
I
t
tO)
too
tO)
hhie
I(
n’vto)1)11
v
veIIo
tn blue
cups.
DATATABLE
Indicat
pH
Solution
or
Prediction
4
A
Methyl
Orange
B
C
D
Bromthymol Phenolphtha Bromcresol
Blue
Green
lein
.
I
Results
•
‘
Prediction
5
,
:
.
Results
.
,
Results
Prediction
7
‘
8
.
,
Results
Prediction
I
,
Results
9
I
Prediction
.
Results
10
.
.
Prediction
6
Thymol
Blue
Prediction
Results
,
9617
4
-
I Page 1
-
Name:
1)
•
witha pH of9.0 to turnblue?
Whichindicatorwould cause a sodiumhydroxidesokflioti
th 1 brange
and pheno
C) bis
1fl
B) Iitnijsand methylorange
D) thymolblue and litmus
S
.
2)
•
3)
r
isaddedto’a beaker containing
Methyl orange indicatc
a solutionofHCIwitha pH of2.OiWhat colorchange
occws as NaOH(aq)isadded to thebeaker?
B) yellowto red
A) blue to red
C) red to yellow
Thynxlblueindicatorisaddedtoa solutioninwhichthe [H3(Y]= I x 10-11.Whatcolorwillthe solution..
appear?
B) blue
C) yellow
A) pink
I)) red
Household vinegarhas a pH of approximately3.0. Which‘ould appearyellowwhen added to a vinegar
.
.4)
D) red to blue
solution?
,
.•
C) litmus
• A) bromcresol’greçn
D) methylorange
B) phenolphthalein
•
What,isthe pH of solutionthatchangesboth methylorange and litmusindicatorsred?
5)
B) 2
C) 8
D) 4
A) ‘6
blueindicator.What colorwillthe
Niiiic acidis addedto a otas
bromthymol
ium
hydroxidesolutioncontaining
6)
is
solutionbe once allof thebase neutralizEd?
‘C) pink
B) yellow
D) colorless
A) red
.
.
•.
.
7)
showninthetablebelow.
The,resultsof testinga colorlesssokaionwiththreeindicators,are
Indicator
red litmus
bluelitmus
•
Result
blue
blue
phenolphthaleinpink
Whichformulacould representthesolutiontested?
A) NaOH(aq)
B) CJ
J(aq)
O
H
C) C61-11206(aq)
D) HC
I(aq)
.
Page1 Natural Indicators
—
Nàturàl Indicators
Acids,Bases,and the pH Scale
Introduction
Rosesare red,violetsare blue—or are they? Red roses, as well as many other flowersand
fruits, contain natural indicatorsthat are sensitiveto acidsand bases.The color of a natural
acid—base
indicatordependson pH. Oneofthe mostwell knowneffectsof natural indicators
in plants occurs in the hydrangeaor snowballplant. Hydrangeaflowersare bluewhen grown
in acidic soils,pink or red in basicsoils.Howdo the colorsof natural indicatorsvary with pH?
Concepts
• Indicators
•
Extraction
•
pH Scale
• Weakacid
•
Conjugatebase
•
Equilibrium
Background
II
Indicators are dyesor pigments that can be isolatedfrom a varietyof sources, including
plants, fungi, and algae.Almostany flower,for example,that is red, blue, or purple in color
contains a classof organicpigments calledanthbcyaninsthat changecolor with pH. The use
of natural dyesas acid—base
indicatorswas first reported in 1664by Sir Robert Boylein his
collectionof essaysExperimentalHistory of Colours.Indeed, Boylemade an important
contribution to the earlytheory of acidsand bases byusing indicatorsfor the experimental
classificationofthese substances.The idea,however,may actually have originated much
earlier—medievalpainters used natural dyestreatedwith vinegar and limewaterto make
differentcolorwatercolorpaints.
Acid—base
indicators are large organic moleculesthat behave as weak acids—theycan
donate hydrogen ions to water moleculesto form their conjugate bases (Equation 1). The
distinguishing characteristic of indicatorsis that the acid (HIn) and conjugate base (Inj
forms are differentcolors.
HIn(aq) + H
20(l)
(colorA)
t
1n(aq) + H
(aq)
3O
(colorB)
Equation I
The abbreviationfin represents an uncharged indicator molecule, and Inan indicator ion
after it has lost a hydrogen ion. The color changes of acid—baseindicators illustrate an appli
cation of reversiblereactions and equilibrium. Becauseindicators are weak acids, the reac
tions summarized in Equation I are reversible.Reversiblereactions can easilybe forced to
go in either direction,depending on reaction conditions.The actual color of an indicator
solution thus reflectsthe position of equilibrium for Equation 1 and dependson the concen
tration of H
ions (and hence the pH).
3O
There are three possiblecases. (1) Mostof the indicator moleculesexist in the form HIn and
the color of the solution is essentiallythe color of HIn. (2) Mostof the indicator molecules
exist in the form 1n and the color ofthe solution is essentiallythe color of In-. (3) The solu
tion contains roughlyequal amounts of the two forms and the resulting color is intermedi
ate betweenthat of Hin and 1n. The exact concentrations of H
3O at which cases 1—3will
NaturalIndicators
Natural lndkators Page2
—
predominate depend oq the structure of the indicator moleculeand the equilibilum con
stant for Equation 1. Differentindicatorsthus change color at different pH ranges.
Natural indicatorsolutions are obtainedby tr
ating flowersand fruits with a solvent to
remove (dissolve)the solublecomponents.This process,calledextraction, is sim
to the
lar
procedure used to make a cup oftea using a tea bag. Thesolidis crushed or ground and
extractedwith an appropriatesolvent,such as boilingwater,ethyl alcohol,or rubbing alcohol.
The color of an acid—base
indicator dependson the concentration of H
ions,which is
3O
most convenientlyexpressedusing the pH scale.The mathematical relationship between pH
and [H
is given in Equation 2.
]
3O
pH
=
Equation 2
]
3O
—log[H
The H
in water ranges from 1 M (10°)in 1 M hydrochloric acid to io’ M
3O
ition
concentr
in 1 Msodium hydroxide.In pure water, which is neutral (neither acidic nor basic),the H
3O
concentration is equal to iO M.The logarithm of the concentration is the “powerof ten”
exponent in these concentrationterms. Thus, the negativelogarithms (Equation2) of typi
cal H
concentrations are positivenumbers from 0—14.The pH scale ranges from 0—14,
3O
with 7 being neutral. cids have pH values less than 7, whilebases have pH values greater
than7.
Withinthe pH range ofacid solutions,either a more concentratedor a strong acidsolution
willhave a lowerpH than a less concentratedor a weakacidsolution,respectively.Thus, the
pH valuesof 0.1 and 0.01MHCIsolutions are 1 and 2, respectively,whilethe pH of 0.1 M
aceticacid is about 3. Onthe basicsideofthe pH scale, either a more concentrated or strong
basesolutionwillhave a higher pH than a lessconcentratedor a weakbase solution, respec
tively.Thus, the pH valuesof 0.1and 0.01M I’JaOHsolutionsare 13and 12,respectively,while
the pH of 0.1Mammoniais about 11.Rememberthat the pH scaleis logarithmic—asolution
of pH 3 is ten times more acidicthan a solutionof pH4, and 100times more acidicthan a
solutionof pH 5. Figure 1 summarizesthe pH scaleand the pH range ofacidsand bases.
0
1
2
3
.4
5
6
7
8
9
10
11
12
13
14
-4
Strong acids
Weakacids
Weakbases
Neutral
Strong bases
Figure 1. The pH Scale
Experiment Overview
The purpose of this experiment is to extract natural indicatorsfrom flowersand fruits and
designa procedure to investigatetheir color changes as a function of pH. Aset of standard
acidand base solutions of known pH (pH = 2—12)willbe provided.The results will be used
to construct color charts of the indicators. In Part B, the natural indicators willbe used,
along with other knownindicator solutions, to analyze the pH values of unknownsolutions.
FlinnChemlopic®
Labs AcidsandBases
—
Page3—Natural Indicators
Pre-Lab Questions
Phenolphthalein is a synthetic indicatorthat is ctlorless when the pH <8 and red when the
pH> 10.The pH range from 8—10is the “transition range” for phenolphthakin. When phe
nolphthalein is addedto solutions havinga pH between8 and 10, the indicator is intermedi
for phe
ate in color betweencolorlessand red, or variousshadesof pink. The color chan
es
summarizedin
the
and
indicators
colorcharts
twoother
following
nolphthaiein
(Thble
are
1).Areasshadedwith hash marks indicate pH intervalsin which the color of the indicator
changes from one form to another.Note:Alizarinexhibitstwo differentcolor transitions,
between5—7and 11—13,
respectively.At pH values greater than 12alizarin appears violet.
Table 1
pH
Indicator
1
2
Alizarin
5
6
7
8
9
10.
Colorless
Phenolphthalein
BromthymolBlue
4
3
12
Red
Yellow
Yellow
11
Blue
Red
1. Whatwillbe the intermediatecolorof bromthymolblue in a solutionof pH 7?
2. Acolorlesshouseholdsolution was tested with the three indicatorsshownabove.The
solution was colorlesswith phenolphthalein,yellowwith bromthymolblue, and orange
with alizarin.Whatisthe pH ofthe solution?Be as specificas possible.
Materials
Standard acid and basesolutions of known pH (pH2—12),5 mL each
Flowersor fruits (roses,violets,geraniums, pansies,petunias, peaches,cherries,
cranberries, grapes, plums), about 5 g
Indicator solutions, 5 mL each
Beakers,100-and 150-mL,1 each
Color pencils, 1 set
Thymolblue
Methylorange
Pipets,Beral-type,5
Reactionplate, 24-well
Bromthymolblue
Funnels and filter paper (optional)
Isopropyl(“rubbing”)alcohol, 50 mL
“Unknown”acidsand bases, 5 mL each
Hot plate (optional)
Mortarand pestle (optional)
Water,distilled or deionized
Safety
Precautions
Thestandardacidand basesolutions used in this experiment are body tissue irritants.
Avoidcontact of all chemicals with eyes and skin. Isopropyl alcoholis flammable. Keep
away from flames and other ignition sources. Wearchemical splashgoggles and chemicalresistant gloves and apron. Washhands thoroughly with soap and water before leaving the
laboratory.
NatLIIdI Indic.itI IFS
Natural Indicators Page4
—
--Procedur
Preparation. Extraction of, Natural Indicators
1. Obtainabout 5 g of’flowerpetals or fruit skin.Note: For smallfruits that are not easily
peeled,use the entire fruit.
2. Tear,chop, grind,.or crush the plant materialand place the piecesin a 150-mLbeaker.
•
3. Coverthe samplewith water or isopropylalcohol.Usea minimum amount of solvent
(approximately50 mL).
4. if water is used as the solvent,heat the mixture,to just belowthe boilingpoint using a hot
plate or bunsen burner setup. Donot heat the mixture if isopropylalcoholis used.
5. After15 minutes,’decantor filterthe mixture into a clean, 100-mLbeaker.The indicator
solutiOnshould be clear,not cloudy.’
•
Part A. Indicator Color Changes
6. Desigr a procedureusing the standard acidand basesolutionsof known pH to determine
the color changes for the natural indicatorsolution and the pH intervalsin which the
color changes occur.
7. Construct a data tableto recordthe results.
8. Showthe data tableand discussthe proposedjrocedure with your instructor.
9. Carry out the procedureand record the results:
Part B. Classifying Unknown Solutions
10. Designa procedureusing your natural indicatorsolution and at least one other synthetic
indicatorto determinethe pH valuesof unknownsolutions.Hint: Chooseindicatorsthat
will giveyou the narrowest range possiblefor the pH valueof each unknown.The color
charts for the availableindicatorsare shown in Table2.
11. Construct a data table to record the results.
12. Showyour data tableand discussthe proposedprocedurewith your instructor.
13. Carryout the procedureand record the results.
Table 2.
pH Value
ThymolBlue
MethylOrange
UromfhyrnolBlue
0
J1
j
2
3
Red
5
6
j
7
8
9
10
Yellow
11
Blue
Yellow
Red
Yellow
Blue
FlinnChemlopic®
Labs AcidsandBases
—
Page5- Natural Indicators
H
Name:_______________________________________
Class/LabPeriod:__________________________________
Natural Indicators
Post-Lab Questions
1. Assumethat the pH 2 color of the natural indicator represents its most acidicform (HIn)
(a) Whatis the pH range in which the rpôst acidicform predominates?
(b) Calculatethe lowestH
3O
concentrationat whichthe indicatorstill existsin this form.
2. Assumethat the pH 12color of the natural ‘indicatorrepresents its most basicform (m
l.
(a) Whatis the pH range in which the most basicform predominates?
‘(b) Caictilatethe highest H
3O
concentrationat whichthe indicatorstillexistsin this form.
3. For one ofthe unknownacid—base
solutions that you tested, explainwhy you chose the
combinationof indicatorsyou did to determine the pH valueof the solution.What is the
advantageof using multiple indicators, rather than a single indicator,todeterminethe
pH of a substance?
4. Construct a Results Tableto summarize the properties of the unknowns.
(a) Estimatethe pH value of each unknown.
eachsolution as acidic or basic.
(b) CiassiI
’
(c) Withineachclassof unknowns—acidsand bases—arrangethe solutions in order
from leastacidicto most acidicand least basicto most basic,respectively.
Natural Indicators
Name
‘
Date
___________
Class
___________
Lab
Acid-Base
Titration
39
Text reference:Chapter20. pp. 584—586
Pre-LabDiscussion
.
In the chemistry laboratory. it is sometimes necessary to experimentally
determine the concentration of an acid solution or a base solution. A
procedure for making this kind of determination is called an acid-base
titration, in this procedure. a solution of known concentration, called the
asured
volume of
standard solution, is used to neutralize a precisely m
the solution of unknown concentration to which one or two drops of an
1icator
have been added. if the solution of un
appropriate acid-base in
known concentration is acidic, a standard base solution is added to the
acid solution until it is neutralized. If the solution of unknown concentra
tion is basic, a standard acid solution is added to the base solution until
it is neutralized.
When carrying out an acid-base titration, you must be able to recog
•
• nize when to stop adding the standard solution, that is, when neutrallza
• tion is reached. This is the purpose of the acid-base md$cator mentioned
above. A sudden change in color of the indicator signals that neutralization
has occurred. At this point, the number of hydronium ions from the acid
• is equal to the number of hydroxide ions from the base.tThe point at which
this occurs is called the end point of the titration. When the end point is
reached, the volume of the standard ‘solution used is carefully determined.
Then. the measured volumes of the two solutions and the known concen
tration of the standard solution can be used to calculate the cohcentration
of the other solution. The following steps tell how to calculate the un
known concentration:
1. Write the balanced equation for the reaction. From the coefficients,
determine how many moles of acid reacts with I mole of base (or vice
versa). Use the coefficients to form a mole ratio.
2. if the mole ratio vs 1:1. the following relationship can be used to
calculate the unknown concentration:
=
>
molarirv of the acid solution
molarity of the base solution
volume of the acid solution
’a
1
=
volume of the base solution
The equation for this relationship can be rewriflen to find the solution
of unknown concentration. For example. if the molarity of the base
were unknown, the equation would be
where 3M
1
M
=
=
=.
X
Mb=
13
3. if the mole ratio vs not 1:1, the calculation of the unknown molar
isv is slightly more complicated. For example, if 2 moles of standard
201
2S
acid solution is neededto neutralize 1 mole of baseof ut
known.
tration. the following relationship exists:
MaX V
8=2(Mb>
The 2 in this equation
iS
concen
Vb)
known as the mole factor.
In Part A of. this experiment, yOu will determine the) molarity of a
solution of NaOH by titrating it with a standard solution of -IC1.The
equation for this reaction is
I
HC
‘+
NaOH
—.
NaCI +
2b
H
Because the mole relationship of H* to 0H is 1:1, no mole factor will be
needed in your calculations:
In Part B of the experiment, you will titrate household white vinegar.
Most commercial vinegar contain at least 4%acetic acid by,weight. You
will use the NaOH solution whose molarity you determined in Part A for
the utrat4on of he vinegar.
Up to this point in your laboratory work, most of your quantitative
experiments have required you to calculate mass relationships. This is
known as gravimetric analysis. Titration requires you té use volume
relationships. a technique known as volumetnc analysis.
This experiment should lead to a better understanding of theproper
ties of acids,and bases, neutralization reactions, and titration techniques.
Purpose
Determine the molarity of a NaOH solution by titrating it with a standard
HCI solution. Determine the molarity of a sample of white vinegar.
Equipment
burets. 50-mL (2)
buret stand
double buret clamp
graduated cylinder. J0-mL
Erlenmeyer flask. 250-rnL
beakers. 250-mL (2)
dropper pipet
pipet, l0-mL
suction bulb
safety goggles
lab apron or coat
-
Materials
0.100 M HCI(standard solution)
NaOH (concentration unknown)
phenolphthalein
Safety
distilled water
detergent solution
white vinegar
Fl
Follow all precautions for working with acids and bases. Note the caution
alert symbols here and with certain steps in the ‘Procedure.” Refer to
page xi to review the precautions associated with each symbol. Always
wear safet goggles and a lab coat or apron when working in the lab.
202
Name
39 Acid-BaseTitration
‘(continued)
Procedure
PARTA TITRA11ONOF BASEOF UNKNOWNCONCENTRATiON
1. Wash two burets with detergent solution. Rinse them
thoroughly. first with tap water. then with distilled water.
2. Obtain about 100 mL of standard acid solution in a clean.
A
dry 250-mL beaker. Obtain about the same’ amount of the base
of unknown concentration in a second 250-mi. beaker. CAU
TION: Handle thesesolutions with care. They can causepain
ful burns f they come iv contort with the skin.,
3. Pour about 10 mL of acid into one buret and rinse the inside
surface of the buret thoroughly. Allow the aqid to run out the
buret tip. Fill the buret to slightly above the 0.0-mL mark with
acid. Then allow the acid to flow out the buret tip until the
bottom of the rneniscus is at the 0-mL mark (see Figure 39-1).
Be’ sure there are no bubbles in the tip. If bubbles are present,
add a little more acid to the buret and allow it to drain through
..
the tip until it is free of bubbles and the meniscus is at 0.0 ml.
4. Repeat step 3 using the base solution in the second buret.
Figure39-1
with step 5 of the “Procedure,” one tab partner should
carry out the instructions while the secondpartner records the
data.
Starting
5. Place a 125-mL Erlenrneyer flask under tIW’acid buret as in
Figure 39-2. Holding a sheet of white’ paper behind the buret to
make the ‘scale easier ‘to read. allow exactly 10.0,mL of acid to
flow into the flask.
6. Add exactly 10.1)mL of distilled water to the flask. Then.
using a clean dropper pipet. add three drops of phenolphthalein.
Swirl ‘the flask to mix all the ingredients.
7. Place the flask on a sheet of white paper under the buret
(ont.aining the base solution. To avoid splashing. he sure the tip
of the buret is in the flask (See Figure *4-2).
8. Swirling the flask gently. begin the titration by adding NaOH
to the flask drop by drop (untiriu’ until a faint pink color re
’
occurs (the pink
mains for about 30 seconds. If overutration
color is too deep). follow ‘our Iea(’hers instructions for correct
ing this condition.
9. Note and record the exaci final volume reading on the scale
of the base buret. Discard the solution in the flask as instructed.
Wash and rinse the flask
F through fl. It is not necessary
JO. Repeat the titration step
.
to refill the buret.s. Simply read anti record the initial volumes
carefully
’
of the solutions in the hureL
mL
mL
Figure39-2
After one tab partner has completed two ratwns vi theJiaOH,
the tab partners should sv?fch roles, The recording partner
.clioukl complete two trials (step.c ! thruugh 9) u’hile the other
partner takes over the recording duties. NOTE: Be sure to read
and record the initial volumes ui the solutions in the burets at
the beginning of each trial,
fit
203
I.
PARTB TITRATIONOFWHITEVINEGAR
11. Using a pipet and, suction bulb,measure 10 rnL of white
mL
Erlénrñeyer flask. Add 100 mLof distilled
vinegar into a 25O
water.
12. Add three drops of phenolphthalein and carefuily titrate.
using the same NaOH solution used in Part A.
13. If overtitration occurs, add a measured amount of vinegar
to the flask (using the pipet) until the solution is colorless. This
time, reach the end point carefully by titratthg drop by drop with
the NaOH solution.
Observationsand Data
PARTA
DATATABLE
mall
TnaI2
HCI NaOH
Initial reading
.
.
TnaI3
HCI NaOH
HO
Tr al4
NaOH
HO
.
NaOH
I
.
,
Finalreading
.
Volumeused
.
.
PARTB
Total volume:
white vinegar
Total volume:
NaOH solution
=
______________
_____________
Calculations
PARTA
For each tnai. calculate the molarity of the NaOH solution using the
relationship M.
204
Thai]
______
Tha12
_______
Tha13
_______
Tha14
_______
‘Name :
Acid BosCLab Titration
-
Pleaseanswer the following questionsbased on the lab and experiment
results.
1. What was the averageMolarity of NaOH’from your 4 trials in Part A?
2. Why do you perform 4 trials fOr this lab?
‘3. What is the’ Titration equation?
4.. What does Ma stand for?’
5. What was the Ma for this lab’
6. What is an acid base titration used for?
-
7. What does the ‘equivalence’ or ‘end point’ mean?
8. What was the nameof the indicator used in this lab?
9. befine the term standard solution,
10.What was the standard solution used in this lab?
ii. A student performinga titration onon acidsolutionwith NaOHneversaw
acolor thange.Why didthestudent not seea colorchange?
acid
12.Write a balanced
equationfor the reactionbetween hydrochloric
andsodium
hydroxidefor thislab.Whattypeof reactionisthis?
‘13.Namesomesourcesof error for thislab.
14.If 30.0 mlof 0.500 M KOHis neededto neutralize10.0mlof HCIof
whatisthe Molcirityof the HCI?
unknown
concentration,
Name ____________________
Date ___________________
Titration ofVinegár
Lab
Purpose: To determine the molarity of an upknown concentration of
P1
)(HC
2
0
vinegar 3
which is also known as acetic acid.
Materials: 50 ml of HC
0
3
H
2
2 150 ml beakers
1 150 ml Erhlenrneyer Flask
2 Burets
50 ml of NaOH
Bromthymol Blue
Procedure:
1. Obtain 50 ml of an unknown concentration of acetic acid
in a clean 150 ml beaker.
)
0
3
H
2
(HC
rd
Obtain 50 ml thestand
ml beaker.
.
base solution in a separate clean 15Ô
3. Fill the acid buret with the acetic acid up to the 0 mark, Fill the
base burét with NaOH up to the 0 rnark
I
t
4. Measure 10 ml of NaOHfrom the bur
flask.
into a 150 ml Erhlenmeyer
5. Add 3 or or 4 drops of Bromthymol blue to the flask.
0
3
H
2
6. Begin titrating (adding) the HC
until the endpoint is reached.
from the buret to the flask
7. Repeat steps 4-6 for Trial 2.
8. Repeat steps 4-6 for Trial 3.
Data
Trial2
Tnall
Tnal3
Vinegar NaOH Vinegar NaOH Vinegar NaOH
InitialReading
FinalReading
/olume Used
Questions:
1. What was the standard solution used ih this experiment?
2. What was the molarity of the standard solution used in this
experiment?
3.
What is the titration equation?
4. Calculate the molarity of the HC
(Ma)
0
3
H
2
work)
your
from Trial 1. (Show.’
1
5. Calculate the molarity of the HC
0
3
H
2
work)
your
(Ma) from Trial 2. (Show
6. Calculate the molarity of the HC
0
3
H
2
your work)
(Ma) from Trial 3. (Show
7. Calculate the average molarity of the HC
0
3
H
2
(Ma).
Page1 —TotalAcidity
TotalAddit
Titration of FruitJuices
Introduction
The refreshingtaste of fresh fruit juices’isdue to a complexblend of flavorsand fragrances.
Fruit juices get their sweettaste from sugars, especiallyfructoseand glucose, and their sour
or tart taste from acids, such as citric acid and tartaric acid. The balanceof sugar to acid
content is one ofthe main factors responsiblefor the appealingtaste of fruit juices—too
much sugar, and the juice will taste bland, but too much acid, and the juice will taste sour.
The “total acidity”of fruit juices is determined by titration with sodium hydroxide.
Concepts
neutralization
• Acid—base
•
Stoichiometry
• Titration
•
Concentrationand molarity
Background
The main acids present in fruits and fruit juices are citric acid (in citrus fruits), tartaric acid
(in grapes), and malicacid (in apples).
OH
OH
OH
I
I
—CH
2—
C—CH
COOH
HOOC
HOOC—CH—CH—
COOH
HOOC
2—CH—
—CH
COOH
OH
COOH
Citricacid
Tartaricacid
Malicacid
Figure 1. Organic acids in fruits and fruit juices
The amount of citric acid in citrus fruit juices can be determined by titration with a standard
solution of sodium hydroxide.(Astandard solution is one whose concentration is accurately
known).Citric acid is a tricarboxylic acid—it has three ionizable or “active”hydrogenatoms
in its structure. Onemole of citric acid therefore reacts with three molesof sodium hydrox
neutralization reaction shown in Equation 1.
idevia the acid—base
OH
HOOC—CH
2—
CH
COOH
OH
+
COOH
Citricacid
3NaOH
C—CH
-O
2-—
CH
CO
+
2O
3H
Equation 1
2-C0
Citrateion
In the titration of a citrus fruit juice with sodium hydroxide,a sodium hydroxidesolution of
known molarity is carefullyadded to a measuredvolume of fruit juice containing phenolph
thalein as the indicator.The exactvolume of sodium hydroxidethat must be addedto reach
the phenolphthaleinendpoint (pH> 7) is measured and then used to calculatethe concen
tration of citric acid in the juice.
TotalAcidity
TotalAcidity Page2
—
Experiment Overview
The purpose of this experimentis to compare the citric acid content in a variet of fruit
juices. The concentration of cittic acid in each juice willbe determined by titration with
sodium hydroxidesOlution.Phenolphthaleinwill be added as an indicator to detect the
equivalence,point in the titration, that is, the point at which all of the citric acid has been
neutralized by reactionwith sodium hydroxide.
Pre-LabQuestions
1. Basedon your previousobservationsof their taste, rank the followingjuices with respect
to their acid content:Orange,white grape, pineapple,and grapefruit.
wn in Figure 1, determine the molecular
2. Usingthe structural formulaof citric acid sh
moIa
formulaof citric acidand calculateits
mass(g/mole).
3. A 10.0-mLsampleof pineapplejuice was titrated with 0.100Msodium hydroxidesolu
tiOn.The averagevolumeof NaOHrequired to reachthe endpointwas 12.8mL.
(a) Calculatethe number.of molesofsodium hydroxiderequired to reach the endpoint.
imber of moles of citric’aciddividedby the number of moles
(b) Usingthe male ratio (n
ofsodium hydroxide)for the neutralization reactionshown in Equation 1,determine
the number ‘ofmolesof citric acid in 10.0’mLof pineapplejuice.
(c) Multiply,the number of moles of citric a,cidbyits molar mass to calculatethe mass of
citric acid in 10.0mLof the Juice.
(d) The concentrationof acid in juices is usually expressedin grams of acid per 100mL
ofjuice.What is the concentration Ofcitric acidin pineapplejuice?
Materials
.
‘
Fruit juices (orange, white grape, pineapple,and grapefruit,etc.), 50 mL each
Phenolphthalein indicator solution, 0.5 or 1%, 2 mL
Sodiumhydroxide(standard)solution, NaOH,0.100 M, 100mL
Water,distilled or deionized
Beaker,50-mL,or small cup, 1
Beakers,100-or 150-mL,2
Beral-typepipet, graduated
Buret, 50-mL
Buret clamp
Erlenmeyerflask, 125-mL
Graduatedcylinder,25- or 50-mL
Ring stand
Testtube, large
Washbottle
White paper for background
F’linnChemlopiC Labs ChemistryofFood
—
Page3—TotalAcidity
Safety Precautions
-
Sodium hydroxidesolution is irritating to skin and eyes. Notify your teacherand clean up
ar chemical
all spills immediately.Avoidcontact of all chemicalswith eyes and skin. We
splash gogglesand chemical-resistantgloves and apron.All food-gradeitems that have
been brought into the lab are consideredlaboratorychemicalsand are for lab use only. Do
not taste or ingest any materials in the chemistry laboratory.Do not remove any remain
ing food items from the lab after they have been used in the lab. Washhands throroughly
with soap and water beforeleaving the lab.
!rocedure
1. Obtainabout50 mLof fruit juice in a 50-mLbeaker or small cup aridrecord the identity
of the juice in the data table.
2. Obtainabout 75 mL ofstandard sodium hydroxidesolution in a 150-mLbeakerand
record the precisemolarity of the solution in the data table.
3. Pour about 5 mLoffruit juice into a large testtube and add 1 drop of phenolphthalein.
4. Usinga graduated,Beral-typepipet, add 0.1 Msodium hydroxidein 1-mLpQrtiOnS until
the juice sampleturns pink or red. Gentlyswirlthe test tube while adding the sodium
hydroxidesolution.Recordthe volumeofsodium hydroxidein the data table.
5. Basedon this “rough titration” of 5 mL ofjuice, estimateand record th volumeof sodi
um hydroxidethat willbe neededto titrate 20.0mL of juice. Usethisestimate as a guide
to determinewhento “slowdown”in the titration (see step 13).Hint: If3 mL of sodjum
hydroxideis neededto reach the “rough”endpoint,beginadding sodium hydroxidedropwise (step 13)afterabout 8 mL of NaOHhavebeen added.
6. Rinse a clean50-mLburet with two 5-mL portions of the sodium hydroxidesolution.
7. Clampthe buret to a ring stand and placea “waste”beakerunder the buret. Fillthe buret
to abovethe zero mark with the sodium hydroxidesolution. Open the stopcockto allow
any air bubblesto escapefrom the tip. Closethe stopcockwhen the liquid levelin the
buret is betweenthe 0- and 10-mLmark.
8. Recordthe preciselevel(initial volume) of the solution in the buret.Note:Volumesare
read fromthe top downin a buret. Alwaysread from the bottom of the meniscusand
rememberto includethe appropriate number of significantfigures.See Figure 2.
Figure 2. Howto read a buret volume.
TotalAcidity
TotalAcidity Page4
—
?M.
9. Measure20.0 mL of fruit juice in’a graduated cylinder and transfer the juic into a
flask.Rinse
mL
125
the
meyer
Erle
graduatedcylinder with three 1O-mLportions of
distilled or deionized water and add the rinse solutions to the Erlenmeyer liask.
10. Add2—3drops of phenolphthaleinindicatorto the Erlenmeyerflask.
11. Positionthe flaskand the buret so that the tip of the buret is inside the mouth:of the
flask.Placea pieceofwhite paper under the flaskto makeit easier to detect the color
change at the endpoint.
12. Open the stopcockto add the estimatedamount ofsodium hydroxide(see step 5) to the
juice sample.Gentlyswirlthe flaskto mixthe contents.
13. Continue to add sodium hydroxideslowly,drop bydrop,while swirling the flask.Usea
wash bottle to rinse the sidesof the flaskwith distilledwater during the titration.
secondswhile swirling the flask,
14. When a faint pink color appearsand persists for 15—20
the endpoint has been reached.Closethe stopcockand recordthe finalburet reading
(finalvolume) for Trial,1 in .thedata table.
I
15. Pour the contents of the flask into the sink.and rinse the flaskwith distilledwater.
16. Repeatthe titration (steps 8—15)with a second20.0-mLsample of fruit juice.
17. (Optional)Iftime permits, completea second set oftitrations with a differentfruit juice.
Flinn ChemTopic Labs
—
Chemistry of Food
Page5—TotalAcidity
Name:-—
-.
Class/Lab Period:
Total’Acidity
Data Table
Titration of FruitJuice
Nameof JuiceTested
‘
RoughTitrationData(Step4)
‘
EstimatedVolumeof NaOHNeededto Titrate20.0 mLof Juice
PreciseMolarityoi NaOHStandardSolution
ThaiI
InitialBuret Reading
.
FinalBuret Reading
Volumeof NaOHAddedat Endpoint*
b
*Post
Thai2
‘
.
.
Calculation #1.
Post-LabCalculationsand Analysis
(Usea separate sheet of paper to answer the followingquestions.)
1. Determine the volume of sodium hydroxide added at the endpoint for each trial and enter
the results in the data table.
2. Calculate the number of moles of sodium hydroxide required to reach the endpoint for
each trial.
3. Based on the mole ratio for the neutralization reaction of citric acid with sodium hydrox
ide, determine the number of moles of citric acid present in 20.0 mL of juice.
4. Calculate the mass in grams of citric acid in 20.0 mL of juice for each trial.
5. What is the averageconcentration of citric acid in the fruit juice in units of grams of
citric acid per 100 mL of juice?
6. Compare the average citric acid concentration in different juices. Based on class data,
rank the juices from most acidic to least acidic. Does this ranking agree with the predic
tions made in Pre-LabQuestion #1?
TotalAcidity
nATE_____________
NAME
Determine the pH of an Unknown Solution Lab
Purpose: Your job is to determine the pH of an unknown solution using the inklicators
listed in Reference Table M.
25 small plastic cups
methyl orange
bromthymol blue
phenolphthalein
bromcresol green
blue
mol
th
red & blue litmus paper
5 unknown solutions
Materials:
Procedure:
You will develop a procedure to determine the pH.
Hints: 1.’ Think out the procedure first.
2. Write it down.
3. Construct a data table to rcord
your observations.
4. Write down any data.
5. Make your determination based on your data.
List your lab partners:
Solution 1:
Solution 2:
Solution 3:
Solution 4:
Solution 5:
omewo.rkAssignment:
Reactthe ChemMysterybelowanduseinformationalresourcesavailableat home
ReferenceTable,etc) to answerthe following:
(Internet, textbook,encyclopedia,
ChemMystery
TheLimestoneCaveman
You have been out for a walk when you come upon a man dead in a
limestone cave. You do not see any bruise, wound, or mark on his
body. You are asked to solve, the, mystery of how. the man died.
Upon searching the scene, you discover a deep hole in the limestone
floor that contains traces of acetic acid. Some ,bubbling is still
occurring. How might the man have died?
Answerthe following:
.
1. What is the chemicalformulafor aceticacidandcarbonicacid?
2. What is the commonnamefor householdacetic acid, andwhat is it used
for?
3. What is the chemicalformulafor limestone(alsomarble,or chalk)?
—
Name________
’-Lab-Activity—-
The Dissolving Cave:
and Acidic Solütioñs
A Reaction between CaCO
3
Ifltroduction
In this lesson the chemical effects of an acidic solution on limestone is
investigated.Limestoneis the same chemicallyas chalk or marble and will react with•
acidic solutions. When rainwaterpasses through he atmosphere, it absorbs some carbon
dioxide from the air. Water and carbon dioxide combine to form carbonic acid. This acid
is weak, but over long periods of time it can do mtich damage. When this acidic rainwater
passes through limestone, it will “dissolve” the limestone. This process forms many
caves. Rocks on the surface are also etched by rainwater.In this experiment students will
produce a solution of’carbonic acid. They will then observe ‘the changes in pH and a,
chemical reaction. Two indicators will be used to determine the pH changes of the
solutionsand during‘thereaction.
Materials
,
Calciumcarbonate(CaCO3)(chalk or marble chips)
BromcresolGreen indicator
BromthymolBlue indicator
100-migraduatecylinders
Household vinegar
‘‘
‘
,.
ReferenceTable M
Two 250-mi beakers,’
Goggles
Spoon
Soda Straw
s
Indicator
1. Fill a graduatedcylinder with 100 ml of distilled water. Transfer the water to a 250-mi
beaker. Repeat this stqp a second time.
2. Put four to 15 drops of Bromcresol green indicator in one of the beakers, and 15 drops
of Bromthymol blue indicator in the other beaker. Gently swirl the contents of each
beaker and place them on a white piece of paper.
Producing “Acid Rain:”
4. Put goggles on. Using a soda straw, slowly blow bubbles into each solution for about a
minute.Write down your observations on the data chart.
5. Based on the colors of each solution, determine the approximatepH of each solution.
Explainwhy you have chosen these pH values.
Note: Be sure to use Reference Table M: Acid Base Indicators. This table shows what
colors each of these indicatorshas for various pH conditions.
6.Now place a: small amount of calcium carbonate powder, chalk, or marble chip into
each-solution;-Write-your
observationsonthe
data-chart;
do
The Effects of Another Acidic SoJution -Vinegar:
Many ancient Greek and Roman monuments have stoçd against natural erosion for
centuries. However with acid rain, these ancient monuments, modern monumelits,
gravestone markers, and even concreteroadways are susceptibleto the corrosive nature
of Acid Rain.
7. Startwith an empty, clean and dry 250-mlbeaker.
8. Place a spoonflilof calcium carbonatein the beaker.This will be our “monument”
9. Using a graduated cylinder,measure and place 20m1of acetic acid (vinegar) into the
beaker containingthe calciumcarbonate.Record your observations.
Observations
Step 4.
Step5.
Step 6.
Step 9.
I
74
Questions
1. Describe 3 physical andJorchemicalproperties of the calcium carbonatç.
2. How does acid rain form?
3. Based on your observationsin the lab, what is the pH of acid rain?
4. What is the effect of acidrain on structuresmade of calciumcarbonate?
5. Using your observations,explain which is a stronger acid carboflicacid.or acetic
acid.
—
6. Write a balanced reaction between calcium carbonate and acetic acid? (Hint:
Water and a gas are two of the three productsproduced.)
7. What is the identity of the gas produced in step #9?
8. How could you test for the presence of the gas evolved in the reaction between.
calciumcarbonateand vinegar?
9. Two different indicators were tsed in this lab investigation. WoWd
phenolphthaleinbe an appropriateindicatorfor this lab? Explain.
10. Based on the information provided in the ChemMystery and lab activity, how
might the man have died in the cave?
References:
Experiment
http://vlc.atu.edu/phsc 1021/2002-falllexp09/expo9.html
ChemMysteiy
http://www.geocities.comlvannamontanalChemMystery.html
‘fl3
Name
______________________
SI.
+
Móde of O
k
a
Objectives:
CompoundsHydrocarbons
•.
To improve your knowledge of organic compounds.
S
2. To be ableto coflstruct models of simple organic compounds.
3. Tobeablelà transfer model structureto diagrams on paper.
Please readthefollowing carefully:
• You will each be given a molecular model building kit Each colored ball represents an
• element A black.l)all with four holes represents carbon and a yellow ball with one hole
.
Using the kit, “you will make three-dimensionalmodels of some
represents hydrogen
organiócompounds. Fromthese models, you will draw theirstructural formulas.
in the balls represent avaiiable electrons and those must be used up when
The hole
s
balls.
Wooden pegs are used for single bonds (2 shared electrons), while spnng
joining
are used for double and triple bonds (4 and 6 shared electrons, respectively).
• •
.
•
.
•
Be sure to use Reference Tables P and Q to help you construct your molecularmodels.
Reference Table P contains a list of organic prefixes. The prefixes show the number of
carbon atoms.in the compoun& For ecample, the prefix “eth” means that a hydrocarbon
compound contains two carbon atoms. Reference Table Qcontains informationabout the
Use the general formula to
three types of hydrocarbons: alkanes. aIken and alk
n.
help you determinethe structure of an organic compound. The “n” in the general formula
equals the numberof carbon atoms in the compound. For example, ethane is an alkane
ethene is an alkene and has the molecular formula
and has the molecular formula C
6
2H
!2
.c
’4
•
have only single bonds between carbon carbon atoms and carbon hydrogen
Mk
atoms. Alkçj have a double bond between two carbon atoms only and alkyjj have a
triple bond between two carbon atoms only.
Procedure:
—
—
+
S
1. Wrte the molecular formulas for each of the hydrocarbonscarbon molecules listed in the
boxes on the following pages.
2. Then,using the molecular models kit construct a model of each.
+
3. Draw a colorful picture of each model you builci
4. Answer the questions that follow.
+
Methane
Ethane
Propane
Butane
Ethene
Propene
Butene
Ethyne
Pentane
-
Propyne
Compounds that have the sam Isomerof Butane
molecular formula but different
structural formulas are called
isomers. For example n-butane and
methyl propane have the same
but the
molecular. formula, 10
41-1
,C
arrangementof the atoms are quite
different. In the box to the right,
drawan isomerof butane.
6909-I -Page]
Name:
_____________________________
I)
‘The compound C4HIO belongs to the series of’hydmcarbons with the general formula
A) C2H2n
2
•
B) CnH2n
2)
•.
•
C) CnH2n
D) CnH2n+2
What is the general formula for an alkyne?
C) CnH2n
A) CnH2n..6
2
B) CnH2n
D) CnH2n+7
The members of the alkaneseries ofhydrocarbonsare similar in that each member has thesame
C) empirical formula
A) structural formula
D) molecular formula
B) generalformula
_3)
‘
4)
Which set of formulas represents members of the same homologous series?
CH4O
C) C2H4,C3H6,C41-Ig
A) C,C1-14,
D) CF12,CH3, CH4
•B) C2H2,C2F14,C2H6
formula
The compound C2H2 belongs
tothe series ofhydrocarbons with thegeneral
_5)
•
A) C2nr12n2
B) C
H
_6)
7)
‘
C)
CnH2n.
2
H2n
D) C2,
number ofcovalent bonds in a molecule of methane is
Thetotal
C) 3
B) 2
A) I
D) 4
ofbutene?
What is the total number ofhydrogen
atoms in a molecule
C)10
B)6
A)8
D)4
ofethyne?
Whatisthenumber ofhydrogenatomsina molecule
‘C)8
B)2.
A)6
_8)
series?
Whichcompoundbelongs
tothealkene
B) C21-L4
A) C6H6
_9)
10)
11)
C) C2H2
JUPAC name endsin
molecule
contains atriple
Ifa hydrocarbon
bond., its
C) -ene
B) -y,ie
A) -one
ofthealkane series?
Whatisthegeneral formula
Cn}ln-f
A) -2
2
B) Cn1Hl2n
C) CnHpj.
D) Cn}12,?-4-2
•
D)4
D) C6H14
D) -ane
•
1
12)
I Page 2
does each compound contain only one double bond pmoIecule?
In which pair of b1rocarbons
C)
A) C4}lg and C2H
4
C2H6
C2H2and
—
13)
•
6909
-
-
C2H2.and C3H6
)-C6H6and-C7H
”-’D
••
Which structuralformularepresentsa compound that is a member of the alkene series?
HH
A)
C—H
H—C
C)
B)
•HH
I
I
H—C—C—H
D)
H—C—C—OH
I
HH
HH
H—C=C—H
HH
14)
A hydrocarbon molecule containing one tr pie covalent bond is classified as an
C) alkane
A) aikvne
D) alkene
B) alkadiene
.
,
.15)
—
Given the compound:
H H H H
I
I
I
I
H—C—C=C—C—H
I
I
H
H
Whatis the general formulaof the hydrocarbon series of which this compound is a member?
C) CnH2n
A) CnH2n
D) CnH2n..2
12n-{-2
B) Cn}
•
—
16)
A molecule of ethaneand a molecule of ethene both have the same
C) molecular formula’’
A) number of carbon atoms
D) empirical formula
B) number of hydrogenatoms
(
Alkane Data Table
a
•Nas.
Methane
-
tiki
Fov
awMg
D
•
4
CH
Ethane
T
•
•
Propane
•
•
: Draw 2
Butane
Pentane
•
Draw 2
•
•
-
-.
-.
-
Alkyne Data Table
Nas.,a
a
Fo,mt
Ethyne
Propyne
Butyne
Draw 2
Alkene Data Table
tz
Nas.
Fou.,i.ea
awMg
D
-
Ethane
Propene
Butene
Draw2
Name
I.
Modelsof Other OrganicCompounds Table R
—
Objectives:
1. To improve your knowledge of other organic compounds and their fimctional groups.
2. To be able to constructmodels of simple organiccompounds.
3. To be able to transfer model structure to diagrams on paper.
Please read thefollowing carefully:
You will each be gven a molecular model building kit. Each colored ball representsan
element., A black bail with four holes represents carbon; a yellow bali with one hole
represents hydrogen a red ball with two holes representsoxygen a blueball with three
holes represents nitrogen; arid a green ball with one hole represents any halogen (Cl
, F,
Br and I) Using the kit, you will make three-dimensional models of some organic
compounds. rom these models, you will draw their structural formulas.
•
.‘
The holes in the balls represent available electrons and those must be used up when
joining balls. Wooäen pegs are used for single bonds (2 shared electrons), while springs
are used for double and triple bonds (4 and 6 shared electrons, respectively).
•
Be sure to use Reference Tables P and R to help you construct your molecular models.
Reference Table P contains a list of organic prefixes. The prefixes show the number of
carbon atoms in the compound.. For example, the prefix “but” means that a compound
contains four carbon atoms. Reference Table R contains infonnation about different
classes of organic compounds and their functional groups. in the general formula, the
letter “R” represents the rest of the molecule. For example, methanol is an alcohol
ethanoic acid is an
3OH;
(functional group —01-1)and has the molecular formula CH
molecular
formula
and
has
the
acid
5 OOI{
2H
C
(functional group -COOFI)
orgamc
•
The way in which the molecular formula is written, the general formula and the
functional groups are the keys to determining the structuralformula of these organic
compounds..Watch for structures that have a double bond between a carbonand oxygen
atom.
Procedure:
I. Classify and write the name for each of the organic molecules listed in the boxes on the
following pages. Identifythe functional group by circling it in the molecularformula.
2. Then, using the molecular models kit constructa model of each.
3. Draw a colorful picture of each model you build.
4. Answer the questions thatfollow.
.
2
_______
5OH
2H
C
HCOOH
3CHO
CH
3CI
CH
OH
CH
32C
CH
5 OOCH
2H
3C
2
3N
CH
3 ONH
CH
2C
H
COCH
3CH
3OH
CH
HCOOH
Try this one: I,2 dichioropropane
9338-1 -Page 1
Name: _______________________________________________
—
I)
What class
compoundscan be representedas R—O1-l?
of organic
‘
A) alcohols
B) acids
2)
—CH
CH
3
The molecule
C)
D)
is a meffil)erofaclass
esters
ethers
of organic
compoukidscalled
0
A) ketones
B) aldehydes
‘C) alcohols
D) ethers
Methanal is the IUPAC name
A) ether
_3)
for an’
,
,
,
B) aldehyde
C) alcohol
D) acid
What type of compound is representedby the structuralformula shownbelow?
H H
HH
I
I
1.1’
H—C—C—0—C—C—H
II•
•Il
H H
HH
C) a ketone
D) an ester
A) an ether
B) an aldehyde
5)
The general
A)
structural
formula
for
aldehydes is
0
0
II
2 C—R
—
1
R
C) R—C”
\
OH
0
B)
R—C
‘
\
H
D)
—O—R
1
R
5
2S
6)
What type of compound is represented by the fo1lo
ng
stTucturaiformula?
HHO.
H
I
I ‘II
H—C—C—C—O—C—H
I
I I
H
HH
C) an ether
D) an aldehyde
A) a ketone
B) an ester
What type of compound is represented by the structural formulashownbelow?
_7)
0
3 \H
CH
C
2
A) an aldehyde
B) anether
—
8)
H
C) an ester
D) an acid
Which structural formula representsan acid?
H
H.O
A)
*
H—C—C—H
I
H
H
B)
_9)
C)
H—C—C—OH
I
I
H H
D)
H
0
I
.‘
H—C—C
\
I
OH
H
C).
0
ii
2 C—O—R
—
1
R
D)
—O—R
1
R
2
H
H—C—C—H
I
I
OHOH
H
What is the general structural formulafor a ketone?
0
A)
A—C”
OH
B)
0
I
2 C--R
—
1
R
9338-l-Page2
9338-1 -Page3
10)
—
What is the genei
l
sfructw’alformula for an ether?
0
—C—R
1
R
2
•
0
D). R—c(
B) 2,R
1—O—R
H
ii)
—
What is the struetural formulafor 1,2-dibromoethane?
•
H
A)
B)
12)
H
BrBr
D)
HC—C—C
H
I.
I
I
HHH
Br—C—Br
I
H
What is the structural formulafor ethanol?
HH
A)
B)
H
C)
H—C—C—H
II
HH
HH
II
H—C—C—OH
I
I
HH
H—C—C”
I
OH
H
HH
D)
H—CC—H
H
_13)
Thestructure H—C—<
isanexainpJeofwhaitypeofsubstance?
H
A) akelone
B) analdehyde
Br
C) H—C—C—Br
I •I
HH
-C—C—H
HI 1.
BrBr
H
•
H
H
C) an ester
D) an amine
Name
____________
Class____________
.,
$
--
Date
___________________________________
Lab
•Saponification
Textreference:Chapter25. pp. 740—742
Pre-Lab Discussion
Fats are esters formed from glycerol (an alcohol) and a long-chain organic
acid (a long-chain fatty acid). The formation of a fat can be represented
by the equation:
0
ii
‘I
H
•
HO—C—H
•
HO_C!_.H
•.
0
H
± 3RC—OH
HO—C—H
R—C—O-—C—H±
>
long-chain
fattyacid
I
•
0
H
3HOH
II
‘
R—C—O—C—H
glycerol
alcohol)”
(an
•
•
,,
a glycerol ester
(áfat)
Soaps are metallic’ salts of fatty acids. Soaps are made by boiling solid
fats or liquid fats (oils) with a solution of a strong base. This reaction is
If the fat from the equation above reacts with a
called saponification.
such
NaOH,
a soap and glycerol are formed:
as
strong base,
O
1-1
II
I
H
R—C—O—C—H
•
HO—C—H
0
O
R—C—O—C—H
+
3NaOH
I
>
3RCONa
asoap
0
HO—C—H
HO—C—H
H
glycerol
R—C—O—C—H
.
H
a glycerol ester
(a fat)
253
In condensed form, this equation can be written:
5
H
3C
(RCOO)
+
3NaOU
—
3RCOONa
+.
5(OH)
H
3C
-
in this experiment, soap will be made by reacting a liquid fat with
sodium hydroxide dissolved in ethanol.
Purpose
I,
Prepare soap from liquid fat and a strong base.
Equipment
beaker, 25-mL
graduated cylinders, 10-mL and
I 00-mL
evaporating dish
ring stand
iron ring
wire gauze
microspatula
test tube, l3XIOOLmm
glass stirring rod,
funnel
burner
watch glass
cork
rubber gloves
safety goggles
lab apron or coat
Materials
I
saturated NaCI solution.
litmus paper
filter paper
oil, cottonseed or olive
ethanol
30%NaOH solution
distilled water
Safety
6
fl A
Sodium hydroxide solution is corrosive. Wear rubber gloves, handle it
with care, and avoid spills on your skin or clothing. Flush any spills with
cool water and report them to your teacher. To protect against spattering,
carry out the experiment with a watch glass on top of the evaporating
dish. Tie back long hair and secure loose clothing when working with an
open flame. Ethanol is flammable; keep the burner flame away from it.
Note the caution alert symbols here and with certain steps in the “Proce
dure.” Refer to page xi to review the specific precautions associated with
each symbol. Always wear safety goggles and a lab apron or coat when
working in the lab.
Procedure
A
254
l. Measure out 4.0 mL of cottonseed or olive oil and pour it into
a clean, dry evaporating dish. Add 4.0 mL of ethanol and 2.0 mL
of 30% NaOH solution to the oil. CAUTION: Handle this solu
tion carefully.
2. Set up the ring stand, wire gauze, evaporating dish, watch
glass, and burner as shown in Figure 50-1. Heat the mixture
the flame away
p
gently with a very low flame. CAUTION: Kee
from the ethanol, which is higidy flammable.
Name
50
Saponification(continued)
Figure50-1
3. Continue heating for 10 minutes. Stop heating when the odor
of fat has disappeared and the oil has dissolved.
4. Allow the mixture to cool. During this time, place 10.0 mL of
distilled water in a 25-mL beaker and heat tc just below boiling.
5. When the evaporating dish is cool enough to touch, add the
hot water to the mixture in the dish. Then add 12.5 rnL of satu
rated NaCI solution and stir the mixture.
6. Filter the mixture, collecting the liquid in the baker.
Discard
the liquid and keep the solid on the filter paper.
7. In a test tube, dissolve I microspatula of your soap in 10.0
mL of distilled water. Test the solution with litmus paper and
record your observations.
8. Add about 1.0 mL of tap water to the solution in the test tube.
Cork the mouth of the tube and shake vigorously. Record your
observations.
I..,
Observations
Litmus test:
Shaking test:
.
255
Conclusions
and Questions
1. What do the processe of saponification and esterificatio
mon? How do these processes differ?
.1
i
have in com
.0
2. To what class Of compounds do fats belong? Soaps?
•
•
3. Describe the contents of the test tube after shaking it (step 8). Did any
white precipitat? (scum) appear? If so, explain its presence.
I.
4. •The formula for the lrocarbon
radical (R—) in the fat glycerol stear
hy
Write an equation showing the reaction of glycerol stearate
ate is H
17
.C
with sodium hydroxide to produce the soap sodium stearate.
I
5. How are soaps treated commercially before they are marketed?
I
256
z’97
Name___________________
Bayport-Blue Point High School
• Chemistry Lab- Nuclear ChemistryUnit
r
‘Ha If—Life.simu,ation
•
Objectiv
To detenninethe half-life Ofthe radioisotope “pennium”.
Discussion
This lab will help you to understand the idea bf half-life as it relates to our
iapter- NuclearChemistry. Usinghalf-lives is,.animportant part of carbon
cj
dating to detenmne the approximate age of artifacts.and fossils. Remember:
always use your reference tables to find the half-life of vanous isotopes!
In this experiment, heads up pennies will represent parent atoms of pennium
and tails down pennies will represent the daughter atoms from the decay.
Materials
Box
120 pennies
Stopwatch or clock with second hand
Procedure
1. Countout 120 pennies and lay them in your box, all HEADS UP!
a. Record this as 0 daughter atoms and 120 parent atoms in the
data chart provided.
b. This is recorded as time 0.
Created by M.Romaine
Page 1 of3
down 20
,times. Be sure to time how long it takes you lo shake the box 20
times. This represents the timing of the decay process.
3. Open the box and count the TAILS UP pennies, which represent the
the lid and shake the box with moderate
2. Cover
force Up &
daughter atoms. Remove all TAILS UP pennies from the box.
a. Subtract this number from the original number of heads-up
pennies to determine the remaining number of parent atoms.
b.. Record the time taken for shaking, the number of daughter
atoms and the number of parent atoms.
4. Repeat steps 2 & 3 seven more times, or until you have run out of
pennies in the box.
Data
Total
&
Observations
Elapsed
Time
I Number
I Atoms
NumberofParent
Atoms (HEADS up)
of Daughte,r
(TAILS up)
‘I
‘
0 seconds
I
.
P
.
.
Making
a Graph
You have to construct
a graph of
your data
radioisotope
penniuin.
Plot the number of remaining parent atoms
the time on the horizontal axis (x-axis).
to detenmne
the half-life
of the
on the vertical axis (y-axis) and
Quesiions
Define:
Radioisotope:
Created by M.Romaine
Page 2 of 3
‘9j.
9t
Pare
atom:
Daughter atom:
Half- Life:
1. Does exactly the same fraction,of penniuni atoms decay during eh
half-life? What does this suggest about half-life? Why are such
when actual atoms are involved?
ous
variations not likely to be ob
I
$
II
2. If you started with one MOLE of pennies, how many would remain
afier 10 half-lives? Show ALL work.
3. If you took a longer time to shake the box in this case, how would the
half-life be affected? Does changing conditions like this control the
half-life of a real atom? Explain.
Crealedby M.Romaine
Page 3 of 3
Name
Bayport-Blue Point High School
Unit
r.Chemistiy
ChemistryLab- Nuc1e
rtiuc1e
orl,
flss
S
Cj
Re
thn
a nuclearchainreactionusingdominoes.
Purpose: To simujate
Materials
Onesetof çlominoes
pergroup
Stopwatch
Procedure
PARTA
iii a straightlineso that hen
1. Placetheentiresetof dominoes
youknockthefirst onedown,theotherswillfall over.
2. Tipthefirstdominoandtimehowlongit willtakefor the
entiresetto fall.
3. Repeatthisat least3 timesandtakethe averageof thethree
trials.
AverageTime-) Part A ________________
PARTB
1. Arrangetheentiresetof dominoesin a patternso thatif you
knockdownthe 1st, it willknockdown2 othertiles,whichwill
eachknockdown2 othertiles,andsoon
2. If youneedhelpdecidinghowto set upyourtiles,seeme!
3. Tipthefirstdominoandtimehowtongit willtakefor theentire
setto fall.
thisat least3 timesandtakethe averageof thethree
4. Repeat
triaIs.
AverageTime-) Part A
Created by M.Romaine
Page 1 of2
Questions:
1. W”hatis a chainreaction?
2. Explainthe processof nuclearfission.Bespecific!
to simUlatea nuclearfission
3. Explainhowwe usedthedominoes
chainreaction.
S
S
fellfaster?
4. Whichgroupof dominoes
5. Howdoesthisshowtheeffectof a chainreaction?
1%
Crealed by M.Romaine
Page 2 of 2
/
Radioactive Decay: The Half-Life of an M&M
Name________________________
INTRODUCTION:
Half-life is defined as the time required for one half of the nuclei of a radioactive isotope
to decay, or change into another element.
In this activity. M&M’s will be your “nuclei”. They are considered to be radioactive if
their M&M emblem is face up
If the M&M emblem is
the radioactiveM&M
.
has decayed and’the M&M is no longer radioactive, thus it can be removed from the
sample.
PURPOSE:
lation
To create a sin
representing radioactive decay. To graph data showing the
l mass remaining radioactive.
relationship between time elapsed ani
MATERIALS:
80 M&M’s. cup. paper plate
PROCEDURE:
1.1You are given a bag with 80 M&M’s. Toss #1 on the data table is already filled in;
the number of M&M’s still radioactive is 80. In the cohimnmarked “Prediction for Next
Toss” write the number or radioactive nuclei you think you will have with your next toss.
2. Place all the M&M’s in the cup. cover with you hand and shake them up 5 times, then
pour them into the plate.
3. Remove and eat if you would like) all M&M’s that are emblem down (thus decayed).
Count the number of M&Ms with the emblem facing up (still radioactive). Record this
number as toss 2 on the data table.
4. Return the radioactive M&M’s to your cup. Repeat steps two and three until all of the
vYs
M&
are one and you have recorded the tosses on the data table.
5. Qjgpj your data by putting “toss number” along the x axis and
radioactive” along the y axis. Title this graph “Group Results”.
—
“
ofM&M’s still
—
6. Combine class data and
but title this “Class Results”.
ji
these results in the same manner as described in step 5
Radioactive Decay: The Half-Lifeof anM&M
DATA TABLE
Toss
O
Number of
1radioactive
M&M’s
I,
Prediction for
next toss
•
80
.
:
‘
‘
Class average
radioactiv e M&Ms
:
,
.,
6
.
‘
.,
S
,
‘
]()
:
‘‘
11
ANALYSIS:
i; When vas the rate of decay faster, during the first or last trials?
S
2. \Vhat factor does the rate of decay depend upon?
3. Based on your graph. what kind of relationship exists between time that has elapsed
and the mass of M&M still radioactive?
Answer tlic klIo
questions using the analogy that each toss represents a halfving
the
half-life of an M&M is 12 seconds.
consider
life. Also.
4. What is
the number of seconds required for a radioactive M&M to pass through three
half-life periods?
5. How nlall\ rams of a 60 gram sample of M&M’s would remain after 24 seconds?
6. Ifvou started vithi 400 radioactive nuclei, how many would remain undecayed after
three half-lives?
7. Stroniiiim-90 has a half-life of 28.8 years. If ou start with a 10-gram sample of
how much will be left after 11 5.2 years? Justi
’
).
strontiun1your answer.
8. Whydid we pool the class data?
9. How m:mv hf-Iives
2 nuclei to decay to 6.25% (0.376x 1023)?
would it take for 6.02 x 1O
BubbleGum
.
Problem:Whatis the half-lifeof bubble gum?
Materials:
I piece of bubble gum per group member
Balance
Clock or stop watch
Procedure:
.
1. Obtan I piece of gum and form a group of four students. Do not chew the
.
gum just yet.
2. Have each group member place his or her gum on a balance. Be sureto
.
.
.
leave the wrapper on.
3. Using the balance, measure the total mass of the group member’s gum.
4. Record the total weight of the unchewed gum on the data chart columnA.
5. Now, have each member chew his or her piece of gum for
Accurate timing is important. Do not throw out the wrappers
.
minute.
6. Using the balance, weigh the chewed gum on the wrappers.
7. Record the total weight of the chewed gum on the data chart column B.
8. Repeat procedures # 5
-
7 until a total of 14 minutes has passed.
9. Complete the entire data chart.
10. Graph your data.
11. Compare your results to other group’s data.
12.Answer the questions that follow.
29
Questions
Could you feel a change with the gum in your mouth over time? What change
did you notice?
e?
Why did the group’s gum lose mass over tiir
What is meant by the half-lifeof a radioactiveelement?
What was the half-lifeof your group’sgum?
What representedthe radioactivedecay in the gum?
Whatdid you noticç by comparingyour results with other groups?
If your group were to continuethe experiment,wouldthe mass of the chewed
gum continueto decline? Why or Why not?
.‘
Time
minutes)
A
Total Weight
Unchewed
B
Chewed
Weight
C
Difference
I
% Decay
.
:•
.
.
1
‘1
2
,‘$
‘
,
3’,’
4
5
6’
,‘
.
‘
,
7.
,.
.,
.
,
‘
.
Page 1 AtomicSpectra
—
2t
(3
Atomic Spectra
Light,Energy, and ElectronStructure
Introduction
Sunlight passingthrough a prism produces a rainbowof colors—thevisiblespectrum. The
separation ofwhite light into its component colors occurs when light wavesof different
wavelengthsare bent by different amounts. When a pure atomic gas such as hydrogenor
helium is subjectedto a high-voltageelectrical discharge,light is producedand the gas
glows.When this light is passedthrough a diffractiongrating, however,the spectrum it pro
duces is different.Insteadof giving the familiar rainbowof colors, the light emitted by the
gas givesa series of bright, colored lines. The seriesof bright lines is calledanatomic emis
sion spectrum and is unique to each element.
Concepts
• Atomicemission spectrum
•
Quantization of energy
• Electron energy levels
•
Electron transitions
Background
The phenomenon of atomic spectra has been knownsince the mid-1800s.Their cause, how
ever,remained unexplaineduntil the structure of the atom and, in particular, its electronic
structure, was olved. Rutherford’sdiscoveryof the nucleus of the atom in 1911answered
many questions concerning the structure of the atom. It also raised many new questions,
including,where are the electrons?
In 1913NielsBohr proposeda modelof electron structure that would explainthe phenome
non of atomic spectra.Accordingto Bohr’smodel,an electron is restricted to certain specific
orbits around the nucleus of the atom. Theseorbits differin their distancefrom the nucleus
and in their energy levels.Electrons that are closerto the nucleus ae lowerin energy than
electrons that are farther awayfrom the nucleus. This ideais calledthe quantization of
energy—electronscan only occupyspecificenergy levels,they may not haveintermediate
energy levelsbetweenthese allowedstafes.The picture that is often used to describethis
idea is the rungs on a ladder.Anelectron must alwaysbe on one of the energy rungs, not
betweenthem. Anelectron ry be “excited”or promoted from a lower energy levelto a
bing
higher energy levelbyabsot
energy of the appropriatewavelength.Conversely,an elec
tron may be “relaxed”down to a lower energy levelfrom a higher energy levelby emitting
energy of the appropriatewavelength,in the form of a photon.
•
AtomicSpectra
Bohr’stheory successfullypredicted the atomic spectrum of hydrogen.Whenelectrical
energy is suppliedto hydrogenatoms in a gas dischargetube, also called a spectrum tube,
the atoms absorb energyand the electronsare promotedto excitedenergy levels.Onceexcit
ed, however,the electronshave a natural tendencyto drop backdownto a lower energy level
by emitting light of the appropriatewavelengthand energy.The emitted light for a given
transition is observedthrough a diffractiongrating as a bright line in the emission spectrum
of hydrogen.
Atomk Specirci Page2
—
Therelationshipbetweenthe energyof light andits wavelengthisshownin Equation1.
Equation 1
AEis thedifferencein energybeb
veen the two energylevelsin joules,h is Planck’sconstant
8 mlsec),andX(lambda)
=
(h 6.626 x iO Jsec), c is the speedof light (c = 2.998 x i0
is thewavelengthof light in meters.
WhenBohrcalculatedtheallowedenergylevelsfor theelectronin the hydrogenatom,he
foundthatthe resultscorrectlypredictedthe wavelengths
in the
of visiblelight observed
emissionspectrumof hydrogen(Figure1).
£6
£4
3
E
.
L
Violetindigo Blue
I
____
Red
Figure1.
fhe Bohr modelof electronstructurewasfoundto beinadequate
for atomscontainingmore
than oneelectron.However,
theideathat only certainstableelectronenergylevelsare
allowedhasendured.Thedevelopment
of quantummechanics
in the 1920sbuilt on theidea
of quantizedenergylevelsandintroducedtheideaof thewavenatureof matterto describe
the propertiesof electrons.Accordingto quantummechanics,
thelocationof an electronis
not restrictedto specificorbitsbut canonly bedefinedin termsof the probabilityof finding
an electron.A systemof atomicorbitalswasintroducedto accountfor the arrangementof
electronsaroundthenucleusof anatom.Anatomicorbitalis a regionin spacewherean
electronmaybefound.Atomicorbitalsdifferin their size,shape,andorientationin space,
andalsoin their energy.Thecharacteristicatomicemissionspectrumof an elementcanbe
of atomicorbitalenergylevelsfor its atoms,.
interpretedbasedon the uniquearrangement
In this experiment,
wewill useaspecialinstrument,calledaspectroscope,
to viewthe “bright
line”emissionspectraofdifferentelementsandto determinetheirwavelengths.
A spectro
containsa diffractiongratingthatseparates
light intoits componentwavelengths.
scope.
Experiment Overview
Thepurposeof this experimentisto recognizecontinuousversuslineemissionspectrafor
Thespectroscope
will alsobeusedto observe
varioussourcesof light usingaspectroscope.
theatomicspectraof differentelementsin spectrumtubesandto identifythe elementsthat
may.bepresentin fluorescentlights,streetlamps,novelty“neon”lamps,etc.
Flinn ChemTopic’Labs
—
Atomicand Electron Structure
Page3—AtomicSpectra
5
(3a
Pre-Lab Questions
1. Readthe Procedureand the Safety Precautions.Whathazards are associatedwith the use
of spectrum tubes?What precautions must be followedto avoidthese hazards?
2. What aspect of Bohr’soriginal modelof electronstructure is still includedin the currently
acceptedtheory of electronstructure?
3. What aspect of Bohr’soriginal modelof electron structure is no longer consideredvalid
in the currently acceptedtheory of electron structure?
4. Assumethat an atom has a total of four possibleenergy levelsand that an electron can
“jump”up or downbetweenany of these energy levels.Drawa modelof these energy lev
els similar to Figure 1and use it to predict the maximumnumber of spectral lines in the
emissionspectrum.
Materials
Colored pencilsor crayons
Diffractiongrating, such as Flinn C-spectra®,or Holographicdiffractiongrating (optional)
Gas discharge (spectrum)tubes, such as hydrogen,helium, mercury, and neon
Light sources, such as incandescentand fluorescent lightbulbs,street lamps,neon signs,
and novelty“neon”lamps
Power supply
Ringclamps,2 (optional)
Ring stand (optional)
Spectroscope
Safety Precautions
Spectrum tubes operateat very high voltages and can produce a largeelectric.hock. Do
not touch the ends of the tube when the power supply is on. Do nót touch the contacts on
the transformerwhen the power is on. Make sure the power supply is turned off before
inserting or removing the spectrum tube. Toextend the life of the tubes, do not leave the
tubes on for more than 30—45sec at a time—cycle the power on and off as needed to com
plete the observations.Spectrum tubes may get very hot. Never touch a spectrum tube
when the power is on. Afte
turning off the power,allowthe tube to coolbefore removing it
from the power iupply.
Procedure
1. Usingthe spectroscopeor a diffractiongrating, observethe continuous “rainbow”spec
trum from an incandescentlightbulb.
2. Observethe colorsof light in the visiblespectrum and the wavelengthrange for each
color band.Sketchthe spectrufrnofwhite light using coloredpencils in the appropriate
wavelengthboxesin the Spectrum Table.Notethat the units of wavelengthon the spec
troscopeare nanometers(1 nm = iO m).
AtomicSpectra
Atomic Spectra Page4
—
(yo
3. (Optional)For optimum viewingof the emission spectra of gas dischargetubes, stabilize
the spectroscopeon a ring stand. Set up a ring stand in front of the powersupplyand
attach one ring clamp. Placethe spectroscopeon the ring clamp and adjust the height of
the ring clamp so that the eyepieceon the spectroscopeis approximatelylevelwith the
middleof the gas dischargetube. Attach a second ring clampon top of the spectroscope
so that it will be held firmlyin position without moving.
4. Withthe power Oft ask the instructor to insert the hydrogenspectrum tube betweenthe
contacts on the powersupply.
5. Movethe power supplyso that the spectrum tube is about3—5cm awayfrom the
spectroscope.
6. Turn on the poweron the powersupply and observethe atomicemission spectrum of
hydrogen.Workwith a partner to note the principal featuresin the hydrogenspectrum.
7. Turn OFFthe powersupply.Recordthe followinginformationin the DataTablefor the
emissionspectrum of hydrogen:the number of lines,their colors,and their approximate
wavelengths.
8. Usingcolored pencils,sketch the atomic spectrum of hydrogenin the wavelengthboxes
in the Spectrum Table.Turn the powersupplyon and off,as necessary,to completethe
observationsin steps 7 and 8.
.9. Checkto make sure the power supply is oIl;then ask the instructor to removethe
• hydrogenspectrum tube and insert a mercury spectrum tube.
10. Observeand note the atomicemissionspectrum of mercury.
11. Turn OFFthe powersupply.Recordthe followinginformationin the DataTablefor the
emissionspectrum of mercury: the number of lines,their colors,and their approximate
wavelengths.
12. Usingcolored pencils,sketch the atomic spectrum of mercuryin the wavelengthboxesin
the Spectrum Table.Cyclethe powersupplyon and off,as necessary,to complete the
observationsin steps 11and 12.
13. Repeatsteps 9—12for any other gasspectrum tubes that are available.
14. Usinga spectroscope,observethe spectrum ofvisiblelight obtainedfrom a fluorescent’
light. Whatkind of spectrum is produced?If any bright lines are present, record the num
ber of lines, their colors,and their approximatewavelengthsin the DataTable.
15. (Optional)Usinga spectroscope,observethe emissionspectrum of other light sources,
such as neon signs, street lights,headlights,noveltylamps,etc. Whatkind of spectrum is
produced?If any bright lines are present, recordthe numberof lines, their colors, and
their approximatewavelengthsin the DataTable.
FlinnChemTopic
Labs Atomicand ElectronStructure
’
—
Page 5- AtomicSpectra
C3as•:
Name:
Class/LabPeriod:
Atomic Spectra
Data Table
LightSource
Spectrum
(Number
of Lines)
Colors(Wavelength,
nm)
Incandescent
Light
Hydrogen
SpectrumTube
,
Mercury
SpectrumTube
SpectrumTube
(optional)
.
.
Fluorescent
Light
S
S
NoveltyLamp
(optional)
S
Street Light
(optional)
s
.
Spectrum Table
S
S
S
700—650 650-600 600—550 550—500 500—450 450—400
LightSource
nm
nm
IncandescentLight
HydrogenSpectrumTube
MercurySpectrumTube
AtomicSpectra
nm
nm
nm
nm
S
:
,
S
S
S
AtomicSpectra Page6
—
,
(396
Post-Lab Calculations and Analysis
1. Accordingto Equation 1 in the Background section, the energy of light (z
E) is inversely
proportionalto its wavelength(X)—asthe wavelengthincreases,its energydecreases.
Basedon the spectrum observedfor incandescentwhite light, rank the colorsin the visi
ble spectrum from highest energy to lowestenergy.
2. Doall of the colorsof light in the visiblespectrum span about the same wavelength
• “width”—thatis, do the bandsof color appear equallywide or narrow?
3. Whatcolor of light in the visiblespectrum appears brightest? Doesthis mean that it is the
•highestenergy light?
ulate
4. UsingEquation 1, cak
the energy (.E) correspondingto each line in the atomic
• emissionspectrum of hydrogen.
5. Asshown in Figure 1, the visibleemissionspectrum of hydrogenisdue to transitions
fromexcitedenergy levelsdownto the second principalenergy level(n = 2). Thus, the
• highestenergy violetline is due to the transition fromn = 6 to n = 2, and the lowest
2. Enter the energy values
energy red line is due to the transitionfromn = 3 to n
(Question4) from highestto lowestin the followingtableand fillin the missing entries.
FlinnChemlopic”Labs AtomicandElectronStructure
—
Page7—Atomic Spectra
6. Plot the energy of each line versus
1
1
—
n.
finaI
Whatdoesthe graph tell you?
on the followinggraph.
‘flitial
Emission Energy vs.
6.00 x 10_19
9
5.00xl0
4.OOx 10-19
U
3.OOx 10-19
9
2.00x10
L
9
1.00x10
0.00 x 1019
0
0.05
0.1
0.15
1
0.25
1
—
final
0.2
fl.
flitial
7. Whatis unique about the spectrum obtainedfor a fluorescent light? Whatelement is used
in fluorescept light fixtures?
8. (Optional)Discussany interesting or unique features of other typesof light sources that
the gasesused in other light sources based
’
were examined.Is it possibleto identil
on
their emissionspectra?
AtomicSpectra
Page 1-Quantum Leap Lab
Quantum Leap Lab
Probability and Electron Structure
Introduction
The picture at the right illustrates a popular viewof the elec
tron structure of the atom—electrons orbiting the nucleus
in fixedpaths. The picture is wrong! It is impossibleto know
the precise locationof an electron around the nucleus of an
atom at any given time. The location of an electron can only
be describedin terms of the total probability of findingan
electron within a region of space.
BohrModelofAtom
Concepts
• Quantum mechanics
•
Electron energy levels
• Heisenberguncertainty principle
•
Atomicorbitals
Background
Throughout the years, significant progress has been made in our knowledgeof the atom.
Atoms were originallydescribedas the smallest particles of matter. Thediscoveries,in turn,
of the electron, the proton, and the neutron destroyed the notion of the indivisibleatom.
Knowledgeof the subatomic particle make-up of the atom raised new questions—whereare
the electrons?In 1913,Niels Bohr developeda modelfor the hydrogenatom in which the
electron was assumedto move in definite orbits, called energy levels,about the atomic
nucleus. The amount of energy the electron possesseddependedon its distance from the
nucleus, with the outer electrons having the most energy.WhileBohr’stheory for the struc
ture of the hydrogenatom wasvery.successful,it failedto hold true for atoms with two or
more electrons. Hencethere wasa need for an improvedmodelof electronstructure.
•
•
•
0
0
The quantum mechanicalmodel,or quantum mechanics,was developedas awayto describe
the motion ofsmall particles(electrons)confinedto tiny regions of space.The exactposition
of an electronat any given instant is not specified;nor is the exactpath that the electron
takes about the nucleus.Theexactlocationof the electronat any given time can never be’
knownwith certainty.The Heisenberguncertainty principlestates that there is a fundamen
tal limitationto howpreciselyboth the position and the momentum of an electron can be
known. Quantummechanicsdescribesthe probabilityof findingan electronwithin a given
region of space.In other words,no longer should we think ofdefiniteorbits of electrons
around the nucleus(asin the Bohrmodel).Rather,we shouldthink of regions of space,
commonlycalledatomicorbitals or electron clouds,which represent the most probableareas
where an electron maybe found,dependingon the amount of energy that the electron
possesses.Thesizeand shape ofatomicorbitals are derivedfromcalculationsthat assume
that theelectronacts as a waverather than as a particle.
0
QuantumLeapLab
uan turn Laap Lab
-
Page2
Experiment Overview
The purpose of this activity is to investigate
byanalogy the relationship between probabil
ity and the electronic structure of an atom. A
marble will be dropped repeatedlyonto a
bull’s-eyetarget. The regions of space around
the central bull’s-eyewill be defined, as
shown on the target sheet (Areas1—6).In
each region, there will be a specificprobabili
ty of locatinga spot resulting from the impact
of the marble drop. The activitywill be car
ried out at two distancesfrom the target rep
resenting different energy levels.Asmallscaleversionof the target is shown in
Figure 1.
6
Figure 1. TargetAreas (1—6)
The pattern of spots on each target sheet will be used to imaginethe three-dimensional
properties of an atomic orbital.Thus, each spot will representa point in three-dimensional
space around the bull’s-eye(analogousto the nucleus) wherethe marble (analogousto the
electron) is capableof landing (or most likelyto be found).The region of space (analogousto
an atomic orbital) in which the marble has a high probabilityof landing will definethe size
and shape of the orbital.
Pre-Lab Questions
1. What is the fundamentaldifferencebetweenan electronorbit and an atomicorbital?
2. In this activity,the marblewillbe droppedfrom twoheights7—-knee-level
level(1.5m). Which height representsa higher energylevel?Explain.
(0.5m) and eye-
3. Form a hypothesisto predict how increasingthe energyof the marl
willaffectthe over
le
all size of the area wherethe marbleis likelyto land.Relatethis hypothesisto the energy
and size of is and 2s orbitals.
Materials
S
2-sheet set
TargetSheet, Knee-Level,9ncarb
less,
TargetSheet, Eye-Level,carbonless,2-sheet set
Marble
Meterstick
Safety Precautions
j
Although this laborator
activity is considerednonhazardous,please observeall normal
laboratorysafety guidelines.
FlinnChemlopic”Labs Atomicand ElectronStructure
Page3- QuantumLeap Lab
Procedure
Knee-Level Target (Target Distance = O.5 m)
1. Obtain a target sheet set labeled“knee-level”and a marble.The target sheet set contains
carbonless paper that will leave an imprint on the bottom sheet when an object strikes
the top sheet.
2. Tracethe bull’s-eyetarget pattern with marked areas on the top sheetof the 2-sheetset.
3. Chooseone person to be the “Dropper”and one person to be the “Catcher.”Laythe knee
level target sheet on a smooth, hard floor.
4. The “Dropper”should hold the marble in one hand and bend downon one knee over the
center of the target. The approximatedistance or height fromwhichthe marbleshould be
dropped is 0.5 m.
5. Havethe “Catcher”sit down next to the target sheet and be preparedto catch the marble
immediatelyafter the first bounce.Note: Practice bouncing the marbleon the floorfirst
to be sure the “Catcher”can catch it beforethe secondbounce.
•
6. The “Dropper”should carefullydrop (donot throw!)the marble fromthe waist(about
0.5 m), aiming for the bull’s-eye.The “Catcher”should catch the marble immediately
after the first bounceto be sure the marble doesn’tleavemore than one mark per drop on
the target heet.
7. Repeatthis procedureapproximately100times over the same target. For ease of count
ing, the “Dropper”should make a tallymark after each drop in the TallyBoxon the
Quantum LeapDataSheet. Each hit will leavea mark on the bottom “copy”sheet.
8. After100 drops,carefullyseparatethe bottom “copy”sheet from the top sheet. Notethe
pattern of marks on the copy sheet.
Eye-Level Target(Target Distance = 1.5 m)
9. Repeatsteps 1—8usingan eye-leveltarget sheet set, with the “Dropper”and the “Catcher”
switchingjobs. The “Dropper”should drop the marblewith the arm fullyextendedfrom
eye level,aiming for the bull’s-eye.Tryto drop the marblefrom a distanceofabout 1.5 m.
•
10 Carefullyseparatethe bottom “copy”sheet from the top sheet. Again,note the pattern of
marks on the copysheet.
QuantumLeapLab
Quantum Leap Lab—Page4
(3
Name:
_______________________________
Class/Lab Period:
___________________
Quantum Leap Data Sheet
Knee-Level
Eye-Level
TallyBox
ResultsTable
Knee-level
trget Distance = 0.5 m
1’
AreaNumber
Eye-level
Thrget Distance = 1.5 m
Area Number
Number of Hits
1
1
2
2
3
4
3
.
,
,
4
,
5.
+6.
Number of Hits
5
6
.
Flinn ChemTopic Labs
—
Atomicand Electron Structure
Page5- Quantum Leap Lab
(3/d.
Post-Lab Questions (Usea separatesheetof paperto answerthe followingquestions.)
1. Circleeach mark made by the marble on both target sheets. For eachsheet,count the
number of hits in each target area (1—5)by counting the number of circles.Count any
hits madeoutside areas 1—5as area 6. If a marble landed exactlyon the line betweentwo
areas, count its location as the higher number area. Recordthe results in the Results
Table.
2. Construct a bar graph for each target sheet. Labelthe horizontal axisas the area number,
and the verticalaxisas the number of hits. Space the bars evenly,makingeach the same
width. Drawthe height of each bar proportionalto the number ofhits in that area.
3. Which area on each target sheet (Areas1—6)receivedthe most hits?
4. Whydon’tall the marbles dropped from a specifiedheight land in the samespot?
5. Asthe distancefrom the bull’s-eye(nucleus)increases,what happensto the probability
distribution of findingthe marble (electron)?
6. What is the overallshape that the spots made on the target sheet?Whatdifferencescan be
seen betweenthe knee-leveltarget sheet and the eye-leveltarget sheet?
7. The two-dimensionalpattern of spots on the paper is intendedto simulate a three-dimen
sional atomicorbital’.Whathape wouldthe pattern havein three-dimensionalspace?
8. Comparethe heightsof the bars on the knee-levelgraph and the eye-levelgraph. Explain
the shift in the heights of the bars toward or awayfrom the origin (Area1).
9. Is there anyway to predict the exactlocation of any onemarble drop on the target?
Explain.
10. Describethe relationshipbetweenthe energy ofanelectron (dropheight) and its proba
ble distanceawayfromthe nucleus of an atom (bull’s-eye).
QuantumLeapLab
Teacher’sNotes
(3/i,
Quantum Leap Target Sheet
Knee-Level
6
Quantum LeapLab
Teacher’sNotes
Quantum Leap Target Sheet
Knee-Level
6
QuantumLeapLab
Teacher’sNotes
(3/3
Quantum Leap Target Sheet
EyeNLeve’
6
Flinn Chemlopic”’Labs
—
Atomicand ElectronStructure
Teacher’s Notes
C3/ó.
Quantum Leap Target Sheet
Level
Eye
6
Flinn ChemTopic
Labs
’
Atomicand ElectronStructure
Page 1 MagnesiumOxide
-
Magnesium Oxide
Percent Compositionand Empirical Formula
Introduction
There is an officialdatabasethat keeps track of the knownchemicalcompoundsthat existin
nature or havebeen synthesized in the lab.The databaseis updateddaily.Currently,over 20
million different inorganic and organic compounds havebeen recognized.Twentymillion
compounds—howis it possibleto identifyso many differentcompoundsand tell them apart?
Concepts
• Percent composition
•
Empirical formula
• Molecularformula
•
Percent yield
Background
The composition of a chemical compound—whatit is madeof—canbe describedat least
three differentways.The percent composition gives the percent by mass of each element in
the compound and is the simplest way experimentallyto describethe compositionof a sub
stance. Accordingto the lawof definite proportions, which was first formulatedin the early
1800s byJosephProust, the elements in a givencompoundare alwayspresent in the same
proportion by mass, regardless of the source of the compoundor howit is prepared. Calcium
carbonate, for example,contains cal,ciutn,carbon, and oxygen:it is present in eggshellsand
seashells,chalkand limestOne,minerals and pearls. Whether the calciumcarbonate comes
from a mineral supplementon a drugstore shelf, or from seashellsat the ocean shore, the
mass percentage of the three elements is alwaysthe same: 40% calcium, 12% carbon, and
48% oxygen.
The percent compositionof a compound tel’s us what elements are present in the compound
and their mass ratio.In terms of understanding howelements come together to make a new
compound,however,it is more interesting to knowhow manyatoms of each kind of element
are in a compound.Sinceall the atoms of a given elementin a compound havethe same
averageatomic mass, the elementsthat are present in afixed mass ratio in a compound must
alsobe present in a fixednumber ratio as well.The empiricalformula describesthe composi
tion of a compoundin terms,ofthe simplest,whole-numberratio ofatoms in a molecule or
The
formulaunit ofthe compo
ind.
The formulaof calcium carbonate,for example,is CaCO
.
3
empirical formulagivesthe ratio of atoms in a compoundand doesnot necessarilyrepresent
the actual numberof atoms in a moleculeor formulaunit. it is possible,in fact, for many
differentcompoundsto share:the same empiricalformula.
The organic compoi:indsacetyleneand benzene,for example,havethe same empiricalformula,
CH—onehydrogenatom for everycarbonatom.Thesetwocompounds,however,havediffer
ent propertiesanddifferentmolecularformulas—C
for acetyleneand C
2H
6H for benzene.
Noticethat in both casesthe molecularformulais a simplemultipleof the empiricalformula.
Themolecularformulaof a compoundtells us the actualnumberofatoms in a single mole
cule of a compound.In order to findthe molecularformulaof a compoundwhoseempirical
formulais known,the molar or molecularmass of the compoundmust also be known.
MagnesiumOxide
Magnesium Oxide Page 2
-
C3/8
Enthis experiment, the percent composition and empirical formulaof magnesiumoxide,the
main compound that is formedwhen magnesium metalcombineswith oxygenin air, willbe
determined. Heating magnesium in the presence nI air causes the metal to ignite and
burn—lots of light and heat are given offand a new compound is obtained. Accordingto the
lawof conservationof mass, the total mass of the products of a chemical reaction must equal
the mass of the reactants. In the case of the combustionof magnesium, the followingequa
tion must be true:
Massof magnesium
+
Massof oxygen = Massof magnesium oxide
If both the initial mass of magnesium and the final mass of the magnesium oxideare meas
ured, the increase in mass must correspond to the mass of oxygenthat combinedwith mag
nesium. The percent composition and empirical formulaof magnesium oxide can then be
calculated,based on the combining ratios of magnesium and oxygenin the reaction.Finally,
once the formula of magnesium oxideis known, the amount of magnesium oxidethat was
producedcan be comparedagainst the maximumamount possiblebased on 100%conver
sion of the magnesium used in the experiment. This informationcan be used to calculate
the percent yield of magnesium oxidein the reaction.
Experiment Overview
The purpose of this classic experiment is to determine the percent composition and empiri
cal formulaof magnesium oxide.
Pre-Lab Questions
A piece of iron weighing85.65 was burned in air. The mass of the iron oxide producedwas
118.37g.
1. Usethe lawof conservationof mass to calculatethe massofthcygenthat reactedwith the
iron.
2. Usethe molar mass of oxygento calculatethe numberofmolesof o,
ygen atoms in the
product.
3. Usethe molar mass of iron to convertthe mass ofiron usedto moles.
4. Usethe ratio betweenthe number of molesof iron and numberof moles of oxygenatoms
rula
to calculatethe empiricalfo
of iron oxide.Note:Fractionsof atoms do not exist.In
the case wherethe ratio of
toms results in a decimalfraction,such as 1.5:1,the ratio
shouldbe simplifiedbyconvertingit to the nearestwholenumber ratio. The ratio 1.5:1,
for example,is convertedto 3:2 bymultiplyingboth termsbytwo.
Materials
lvlagnesiumribbon, 25 cm
‘Crucibleand cruciblelid, 15-or 30-mL
Crucible tongs
‘Bunsenburner
Ring stand and ring clamp
Balance,centigram(0.O1-gprecision)
Claytriangle
Scissors
Wiregauzewith ceramiccenter
Guide (optional),
Laboratory‘Techniques
Labs MolarRelationships
FlinnChemTopic
& Stoichiometry
’
—
Page3- Magnesium Oxide
Safety Precautions
Magnesium is a flammable metal. Magnesium bums with an intense flame.Donot look
directly at burning magnesium. The light contains ultraviolet light that may hurt your
eyes. Donot inhale the smoke produced when magnesium is burned.Handlethe crucible
and its lid only with tongs. Do not touch the cruciblewith fingers or hands. Thereis a sig
nificant burn hazard associatedwith handling a crucible—rememberthat a hot crucible
looks exactly like a cold one. Always keep your face at arm’s length from the crucible.Wear
chemical splash goggles and chemical-resistant gloves and apron. Washhands thoroughly
with soap and water before leaving the laboratory.
Procedure
1. Set up a Bunsenburneron a ring stand
beneath a ring clampholdinga clay
pipestemtriangle. (SeeFigure 1.) DoNOT
light the Bunsenburner.
2. Adjustthe height of the ring clampsothat
the bottom of a crucible sitting in the clay
triangle is about 1 cm abovethe burner. This
will ensure that the cruciblewillbe in the
hottest part of the flamewhen the Bunsen
burner is lit (step7).
3. Usingtongs to handlethe crucible (see
Figure2), measure the mass of a clean,dry
empty crucibleand its lid to the nearest
0.01 g. Recordthe mass in the data table.
Figure 1.
4. Measurea 25-cm length of magnesiumrib
bon and cut the magnesiumto this length.
5. Wearinggloves,coilthe metal ribbon
around a pencilto obtain a looseballof
metal.
6. Placethe coiledmagnesiumribbonin the
bottom of the crucibleand measure the
combinedmass ofthe crucible,cruciblelid,
and magnesium.Recordthe mass in the
data table.
7. Placethe coveredcruciblewith its lid on the
claytriangleas shownin Figure 1. Light the
Bunsenburner and brush the bottomofthe
cruciblewith the flamefor 2—3minutesto
slowlyheat the crucibleand its contents.
MagnesiumOxide
Figure 2.
Magnesium Oxide
-
Page4
8. Placethe burner on the ring stand and heat the crucihic
in the hottest part of the flame. (See Figure 3.) Notethe
approximatetime.
9. After3 minutes,usecrucibletongsto carefullylift thelid
a small amount. Thiswill allowair to enter the crucible.
Caution:Donot open the lid too far, because doing so
willallowthe metal to ignite. There will be some smoke
produced.Donot inhale the smoke!Donot lean overthe
crucible. Keepthe crucibleat arm’s length at all times.
Blueinnercone
shouldbe touching
thecrucibie.
Figure 3.
10. Replacethe lid and continue to heat the crucible.After3 minutes, again liftthe crucible
lid to allow more air to enter the crucible. Replace,thelid immediatelyif the metalstarts
to burn or the amount of smoke increases greatly.
11. Continue heating the cruciblefor a total of 15minutes.Approximatelyeverythree min
utes, lift the cruciblelid to allowair to enter.
12. After15 minutes, turn offthe gas source and removethe burner.
13. Usingtongs, removethe cruciblelid and place it on a wiregauze on the bench top. With
the tongs, removethe cruciblefrom the claytriangleandplaceit on the wire gauzeas
well. (See Figure2.),
14. Allowthe crucibleand its contents to cool completelyon the wire gauze for at !east’lO
minutes.
15. Measurethe combinedmass ofthe crucible,cruciblelid,and magnesium oxideproduct.
Recordthe mass in the data table.
16. (Optional)Iftime permits,dump the contents ofthe crucibleonto a watch glass.Usinga
arance
and consistencyof the product. Is any
spatula,break up the solidand note the app
magnesium ribbonstill present? Recordall observationsin the data table.
17. Dump the contentsof the crucibleinto the wastebasketand carefullyclean the crucible
andcrucible lid.
FlinnChemTopic°
Labs MolarRelationships& Stoichiometry
—
Page5- MagnesiumOxide
j
(3
Name:
Class/LabPeriod:
Magnesium Oxide
Data Table
Massof CrucibleandLid
,
Massof Crucible,Lid, andMg Ribbon
Massof Crucible,Lid,andProduct
Appearanceof Product
Post-Lab Calculations and Analysis
(Show all work on a separate sheet of paper.)
1. Calculatethe mass of magnesium metal and the mass of the product. Usethe lawof
conservationof mass to calculate the mass of oxygenthat combinedwith the magnesium.
2. Calculatethe percentcomposition of magnesiumoxide.
•
3. Usethe molar massesof magnesium and oxygenatoms to calculatethe number of moles
of each reactant.
4. Calculatethe ratio betweenthe number of molesof magnesiumused and the number of
molesof oxygenin the product. Whatis the empiricalformulaof magnesium oxide?;
5. Write a balancedchemical equation for the formationof magnesium oxidefrom magne
sium metaland oxygengas.
6. Usethe moleratio of magnesium oxideto magnesiumfromthe balancedchemical equa
tion and the molar mass of magnesium oxideto calculatethe theoreticalyield of product.
The theoreticalyieldof a product in a chemicalreactionis the maximum mass of product
that can be obtained,assuming 100%conversionof the reactant(s).
7. The percentyieldreflectsthe actual amount of product formedas a percentageof the
maximumthat might havebeen obtained.Usethe followingequationto calculatethe
percent yieldof magnesiumoxideproducedin this experiment.
% yield =
actual mass of product(g)
x 100%
theoretical mass(g)
8. Discusssourcesof error in this e’çperimentthat might account for a percent yield lower
than 100%.Bespe
ific.
MagnesiumOxide
Page1 FreezingPoint Depression
-
,
(32
Freezing Point Depression
How LowCan You Go?
Introduction
People who livein northern states are familiarwith winter and the snowy,icy rdads that go
with the season. Roadcrewsspread salt (sodiumchloride, calcium chlorideor a salt mix
ture) on the roads in order to lowerthe temperature at which freezingoccurs. If the road
already has ice on it, the salt helps to melt the ice, forming a solutionwith a lower freezing
point than that of pure water. Let’sinvestigatethe effectof dissolvedsolutes on the freezing
point of a solution and determine which solute has the greatest effecton the freezingpoint.
Concepts
• Freezing point
•
Freezingpoint depression
• Colligativeproperty
•
Molality
Background
n
The freezingpoint of a liquid is the temperature at which the forcesof attracti
among
molecules are just great enough to causea phase change from the liquidstate to the solid
state. Strictly speaking,the freezing(or melting) point of a substance is the temperature at
which the liquid and solid phasesare in equilibrium.
During the freezingprocess of water, for example,water molecules cometogether to form
the more orderly,crystallinepattern of ice molecules.When any solute (such as salt) is
added to a solvent(such as water), the crystallinepattern is interrupted by the presence of
the salt “impurity.”Salt and other dissolvedsolutes interferewith the ability of the solvent
to crystallize(solidify)and the solution remains liquidevenat a temperature belowthe
freezingpoint of the pure solvent.Thus a solution alwayshas a lowerfreezingpoint than its
pure solvent.This phenomenon is termed freezingpoint depression.
is definedas the differencein temperature betweenthe
Freezingpoint depression(AT
1)
of
and
that
of its pure solvent.The freezingpoint depression of the
solution
freezing point a
solution after the addition of a solute can be calculatedusing Equation 1.
1
AT
=
1 x m x i
K
Equation 1
1AT is the change in freezingpoint (the freezingpoint depression) in °C,K
1 is the freezing
point depressionconstant (1.86 °C/mfor water solutions),m is the molalityof the solution
(the solution concentration in moles of solute per kilogram of solvent), and i is the number
of particles formedwhen the solute dissolvesin the solvent.
Freezingpoint depressionis a colligativeproperty of a solution.Ideally,a colligativeproperty
dependsonlyon the number ofsolute particlesthat are formedwhen the solute dissolvesin a
givenmass ofsolvent.It doesnot dependon the size or the identity of the particles.Boiling
point elevationand osmoticpressure are other examplesof colligativepropertiesof a solution.
FreezingPointDepression
Freezing Point Depression Page2
—
’i.
L3
Certain solutes lowerthe freezingpoint more than other substances.Whena molecular
is dissolvedin waler,the moleculesdo not dissociate
compound such as sucrose )01(C
212
H
12
and remain as single particles. Ionicsolutes, on the other hand,dissociateinto ion!swhen
dissolvedin water.Thus, one formulaunit of sodium chloride(NaCI)dissociatesin water to
produce two particles—onesodium ion (Nat) and one chlorideion (Clj. One unitofcalci
when placed in water dissociatesinto three particles—onecalcium ion
urn chloride (CaCl
2)
and two chloride ions (Cl). Lookingat Equation 1, it can be seen that the freezing
21
(Ca
depressiondepends
point
on the number of particles in solution.The more particles in solu
tion, the greater the change in freezingpoint should be.
Experiment Overview
The purpose of this experimentis to measure the freezingpoint of pure water and the freez
ing point depression forvarioussolutions.The effectof the concentration and number of
dissolvedsolute particles on the freezingpoint of water willbe determined.
Pre-LabQuestions
•
•
Read the eptire procedure carefully,then answer the followingquestions.
1. What factorswill be heldconstantwhen determining the freezingpoint depressionfor
each solute in this experiment?
2. Whyis it important to keep the thermometer offthe bottomof the beaker?
te
ratur
3. Why is it necessaryto measurethe temp
assuming it to be 0.0 °C?
of the pure ice-watermixture, insteadof
Materials
Aluminumchloride 2O
30g.
36H,
hexahydrate,AlCl
Calciumchloride dihydrate, 2O
30 g
CaCl
H,
Sodium chloride, NaCl,30 g
30 g
Sucrose, ,01H
2C12
Crushed ice, 320’g
Water,tap or distilled
Balance,0.1 g
Beakers,250-mL,4
Graduatedcylinder (optional)
Thermometer
Tonguedepressorsor plasticstirrers, 4
Weighingdishes, 4
Safety Precautions
Aluminum chlorideand calciumchlorideareslightly toxic by ingestion.Sodium chlorideand
sucroseare not consideredhazardous;however,the chemicalsprovidedare for laboratoryuse
only and are not intendedforhuman consumption.Avoidcontactof all chemicalswith eyes
and skin. Wearchemicalsplashgoggles,chemical-resistantgloves,and a chemical-resistant
apron. Washhands thoroughlywith soap and water beforeleavingthe laboratory.
Flinn Chemlopic°Labs
—
SolubilityandSolutions
Page3- FreezingPoint Depression
Procedure
• Form a workinggroup with three other students and divideinto twopairs.Each pair of stu
dentswill measurethe freezingpoint ofwaterwith their thermometer (steps2—6)and then
determinethe freezingpoint depressionfor two of the solutes.Atthe conclusionof the experi
ment, both pairs ofstudents in eaëh group willshare their dataand completethe’datatable.
1. Labelfour 250-mLbeakers#1—4.
•
2. PlaceBeaker#1 on the balanceand tare the balance,if the balanceis electronic.
3. Add100 grams of ice-waterto the beaker by (a) first addingapproximately70—80grams of
crushed iceand (b)then addingenough water so the total mass of ice plus water is about
100 g. Recordthe precisemass of the ice-watermixtureto the nearest tnth of a gram in
the data table.Note: If the balancedoesnot have a capacitylargeenough,weigh the ice in
a small weighingdish and then place it in the beaker.Measurethe remainingwater using
a balanceor graduatedcylinder.
4. Stir the ice-watermixturewith a tongue depressor.
5. Carefullyinsert a thermometer into the ice-watermixture.Holdthe thermometer slight
ied
ly offthe bottomof the beaker so that the thermometer bulb is surroun
on all sidesby
the ice-watermixture.
I
’ the temperature readingto stabilize.Recordthe temperature ,f the pure ice
6. Waitfor
water mixturein °Cto the nearest tenth of a degreeon the data sheet.
Beaker # 1—Sodium Chloride
7. In a weighingdish,weighout 30.0 g ofsodium chloride.Recordthe precisemass of the
sodium chloridein the data table.
8. Addthe sodiumchlorideto the ice-watermixture in Beaker#1.
9. Stir the contents of the beakerwith a tongue depressoruntil the mixture has a slushy
appearance.
10. Carefullyinsert a thermometer into the mixtureand measure the temperature. Stir only
with the tongue depressorand not with the thermometers. Holdthe thermometer in the
beakerwith one hand to read the temperature and stir with the woodenstirrer with the
other hand.
11. Continueto stir the mixturewith the tongue depressor.Recordin the data table the low
est temperature (to the nearest tenth of a degree) that the mixture reachesbefore rising
again. This may take some time, as the salt doesnot immediatelydissolvein the icewater.
Beaker #2—Sucrose
12. Repeatsteps 2—11for Beaker#2, using 100.0grams of icewater and 30.0 grams of
sucrose. Rememberto record precisemasses and temperature readings to the nearest
tenth of a degree.
FreezingPointDepression
Freezing Point Depression Page4
-
Beaker #3—CalciumChloride
13. Repeatsteps 2—11for Beaker#3, using 100.0 grams of ce water and 30.0 grams ofcalci
um chloride.Rememberto record precisemasses and temperature readingsto the near
est tenth of a degree.
Beaker #4—Aluminum Chloride
14. Repeatsteps 2—11for Beaker#4, using 100.0grams ofice water and 30.0 grams of alu
minum chloride.Rememberto record precise massesand temperature readingsto the
nearest tenth of a degree.
15. Disposeof the solutions bypouring the mixtures downthe drain with plenty of water.
Rinsethe beakerswith tap water.
FlinnChemTopicLabs SolubilityandSolutions
—
Page 5- Freezing Point Depression
6.
C3
Name:_____________
Class/LabPeriod:
_________
Freezing Point Depression
Data Table
Freezing Point of Water
___________
Beaker
Number
Solute
1
Sodium chloride,NaCI
2
Sucrose,01C
.2H12
3
Calciumchloride,
CaC1
2O
H
4
Aluminumchloride
3
AlCl
20
6H
Mass of
Ice + WaterS
Massof
,Solute
LowestTemperature
of Mixture
.
,
____
Post-LabCalculationsand Analysis (Showall work ona separate sheet of paper.)
Fill in the results of all calculationsfo Questions#1—7in the ResultsThble.
1..Determinethe molar mass ofeach solute. Rememberto includewatersof hydration.
2. Calculatethe number of moJesof each solute using the exactmass of the solute from the
DataThbleand its molar mass.
3. Calculatethe experimentalvalueofthe freezingpoint depression,AT
1 for each solute,
using the equation below.
1 (pure solvent)
T
—
1 (solution) = 1AT (in °C)
T
4. Calculatethe AT
1 per moleofsolute.
5. For each solutethat dissociatesin water,write a balancedequation for its dissociation
reaction. Determinethe idealvalue ofI for each solute. This is the number of particles
formedwhen one formulaunit of the solute dissolvesin water.Note: Watersof hydration
are NOTincludedin the valueof i.
6. Calculatethe concentrationof each solution in molality,in, definedas the number of
molesofsolute per kilogramof solvent.
Molality = m
FreezingPointDepression
=
molesof solute
kilogramofsolvent
Freezing Point Depression Page6
—
7. Calculatethe theoreticalvalueof the freezingpoint depressionfor each solute based on
the molalityof the solution (m) and the idealnumberofsolute particles(i). UseEquation
1 from the Backgroundsection, Kf = 1.86°C/m,and the calculatedvaluesfor m and i.
8. Compare the experimentaland theoreticalvaluesof ATfforeach solute. Discuss’possible
sources of error in this experimentand their likelyeffecton the experimentalAT
1 values.
9. Whichsolute had the greatest freezingpoint depressionper mole?Whichhad the least?Is
this what wouldbe expected?Explain.
10. Preparea graph of AT
1/mole
shown by the graph.
on the y-axisversus i on the x-axis.Describethe relationship
ResultsTable
BeakerNumber
Solute
2
1
I
NaC
4
3
CaC1
2O
H
2C12
01H
36H
AlC1
2O
MolarMass (g/mole)
.
Molesof Solute
1(exp)
AT
.
,
.
.
1(tiT
exp)
mole
I (idealvalue)
m
1AT (theor)
FlinnChemTopic’
Labs SolubilityandSolutions
—
Name
Date_
______________________________
Class
___________
(328
Freezing Point Depression
withAntifreeze
Lab
45
APPLICATION
Text reference. Chapter
15
Introduction
Have you ever wondered how antifreeze lowers the freezing point of
water in the cooling systems of automobiles? In the latter part of the
nineteenth century the French chemist François Raoult noted that the
vapor pressure of a solvent was lowered by the addition of a solute
(Raoult’s Law), and that the freezing points of solutions also were
lowered.
Eventually, the following mathematical expression that related freez
ing point depression and molalitv (moles of solute per kilogram of sol
vent) was developed.
=
,m
K
In this equation, T is freezing point depression, K is the molal freezing
point constant for the solvent, and m is the molality of the particles
in solution.
As you can see from the equation. the lowering of the freezing point
depends on the concentration of dissolved particles present. In the case
of a nonelectrolyte, the molality of the nonelectrolyte and the rnolality
of particles in solution are the same (a 1:1 ratio). For electrolytes, the
molality of particles is equal to the molality of the electrolyte times the
number of ions in the chemical formula of the compound.
Automobile manufacturers make use of the principle of freezing
point depression to protect engines from freezing in cold weather. Anti
freeze, a nonelectrolyte, is added to the water-filled radiator that cools the
engine. Under most conditions the presence of the antifreeze molecules in
the water is sufficient to keep the system from freezing.
the freezing point depres
In this investigation you will detë
rriTine
sion of antifreeze solutions by cooling them in an ice-salt bath. You will
also use this information to find the molar mass of ethylene glycol.
Pre-Lab Discussion
Readthe entirelaboratoryinvestigationand the relevantpagesof your
textbook.Thenanswerthe questionsthatfollow.
1. What is the mathematical relationship between freezing point depres
sion and molalitv? _______________________________________
2. What is a colligative property?
© Prentice-I-laH,inc
’
Freezing Point Depression with .4n:zrrt’t’:
245
Name
Lab
U
Safety
45
-
Wear your goggles and lab apron at all times during the investigation.
Commercial antifreeze is primarily ethylene glvcol, which is highly toxic.
The ice used in the investigation could become contaminated with anti
freeze by accident. Do not eat the ice. Note the caution alert symbols
here and with certain steps of the Procedure. Refer to page xi for the
specific precautions associated with each symbol.
Procedure
Put on your goggles and lab apron. Half fill a 600-mL beaker
with ice and cover it with about 20 grams of sodium chloride
(NaCI). Stir this ice-salt mixture with a stirring rod until it
reaches a constant temperature at or below 10°C. Cover the
outside of the beaker with paper toweling or aluminum foil
with the reflective side in.
2. Half fill a test tube with distilled water and place it in the ice
bath. Rinse the stirring rod and use it to stir the water gently
until ice crystals first appear. Use a thermometer to measure
the freezing point and record it in the Data Table. CAUTION:
Thermometers are fragile. Do not use the thermometerasastirring rod.
1.
I
—
jj
thermometer
papertowel
foil
600-mLbeaker
-
testtube
antifreeze-water
mixture
iceandsalt
Figure 44-1
.
3
o
.4aII
© Prentice-
Find the mass of a 250-mL beaker. Leaving the beaker on the
balance, set the balance for an additional 10.00 grams. Add
antifreeze to the beaker carefully from a 10- or 25-mL graduated
cylinder. CAUTION: Antifreeze is toxic. Do not drink it. If you
spill any, rinse it off with plenty of water and tell your teacher. When
the balance is close to equalization, add the final antifreeze with
a dropper. Now add 100 mL of distilled water to the beaker
and stir thoroughly with the stirring rod. In a similar fashion,
prepare a second solution using 20.00 grams of antifreeze. Label
the beakers Solution I and Solution 2.
Inc
Freezing Point Depression
247
Name
Lab
Critical Thinking: Analysis and Conclusions
1. Permanent antifreeze is almost 1OOethylene glycol (1, 2 ethanediol,
F.OH)
2)4(C
-1
2.
45
PL
0 I
Calculate its molar mass. (Applying concepts)
Calculate the percent error in both trials. (interpreting data,’
Solution I
Solution 2
3.
What do you think are the major sources of error in this investigation?
How might some of them be reduced? (interpreting data)
Critical Thinking: Applications
1.
Could freezing point depression
be used for substances not soluble
in water? (Making predictions)
2.
What effect on the freezing point depression of water would a 1 in
solution of the ionic substance (NH
have? (Making predictions)
4)1PO
3.
What assumption
investigation?
4.
is made about the density of distilled water in this
(Making inferences’ ______________________________
Would this method of molar mass determination
other substances
Preriiice-Hafl. inc
be practical for
soluble in water? (.4ppliiing concepts)
FreezingPoint Depressionwith Antifree:e
249
Page1 ClassicTitratiOn
•
—
ClassicTitration
pH. Curves and an Unknown
Introduction
One of the most commonquestions chemists haveto answer is howmuch of something is
present in a sample or a product. Ifthe product contains an acid or base, these questions are,
titrations can be used to measure the concentration
usually answeredbytitration. Acid—base
of an acid or base in solution and to calculatethe formula (molar)mass of an unknownacid
or base.
Concepts
Acidsand bases
• Neutralizationreaction
•
Titration
•
Equivalencepoint
• Titration curve
Background
Titration is a methodofvolumetric analysis—the use ofvolumemeasurementsto analyze
an unknown. In acid—base
chemistry,titration is most often used to analyzethe amount of
acid or base in a sampleor solution. Considera solution containing an unknown amount of
hydrochloricacid.In a titration experiment,a knownvolumeof the hydrochloricacid solu
tion would be “titrated”by slowlyaddingdropwisea standard solution of a strong basesuch
as sodium hydroxide.(Astandard solution is one vhose concentration is accuratelyknown.)
The titrant, sodium hydroxidein this case, reactswith and consumes the acidvia a neutral
ization reaction (Equation1). The exactvolumeof base neededto react completelywith the
acid is measured.Thisis calledthe equivalencepoint of the titration—the point at which
stoichiometric amounts of the acidand base havecombined.
HCI(aq) + NaOH(aq)
—
NaCl(aq) + H
20(l)
Equation 1
Knowingthe exactconcentrationand volumeaddedof the titrant gives the number of moles
ofsodium hydroxide.The latter, in turn, is related bystoichiometryto the number of moles
of hydrochloricacid initiallypresent in the unknown.
Indicators are usuallyadded to acid—base
titrations to detect the equivalencepoint. The end
point of the titration is the point at which the indicator changes color and signals that the
equivalencepoint has indeedbeen reached.For example,in the case of the neutralization
reaction shownin Equation 1, the pH of the solutionwouldbe acidic (< 7) beforethe equiv
alence point and basic (> 7) after the equivalencepoint. The pH at the equivalencepoint
should be exactly7, correspondingto the neutral products (sodium chloride and water). An
indicatorthat changescolor around pH 7 is therefore a suitable indicator for the titration of
a strong acidwith a strong base.
The progress of an acid—base
titration can also be followedby measuring the pH of the solu
tion beinganalyzedas a function of the volumeof titrant added.A plot of the resulting data
is called a pH curveor titration curve. Titration curves allowa precise determination of the
equivalencepoint ofthe titration without the use of an indicator.
ClassicTitration
ClassicTitration Page2
—
Experiment Overview
The purpose of this experiment is to analyze the shape of the titration curve for neutraliza
tion of hydrochloric acidwith sodium hydroxideand to determine the concentrationof an
unknown hydrochloric acid solution. In Part A,pH measurements may be madeelectroni
callyas a function of time using a pH sensor attached to a computer or calculator-based
interface system, or manuallyas a function of volumeadded using a pH meter. In Part B, the
titration will be repeated with the use of an indicator to detect the endpoint.
Pre-Lab Questions
The approximateconcentration of a hydrochloricacid solution is 0.5 M.The exactconcentra
tion of this solution is to be determined by titration with 0.215Msodium hydroxidesolution.
1. A 10.00-mLsampleof the HCIsolutiOnwas transferred bypipet to an Erlenmeyérflask
and then diluted byaddingabout 40 mL of distilledwater.Whatis the approximateH
3O
concentrationand pH of the solution in the flaskbeforethe titration begins?
2. Phenolphthaleinindicatorwas added,and the solutionin the flaskwas titrated with 0.215M
The exactvolumeofNaOHrequired
NaOHuntil the indicatorjust turned pink (pH 8—9).
was22.75mL.Usethe followingequationto calculatethe concentrationof HCIin the origi
nal 10.00-mLsample.
Mb >( Vb
•
=
nM
X
V
Mb = molarity of standard basesolution Ma = unknown molarity of acidsolution
Vb = volumeof baseadded
Va = initialvolumeof acid solution
of
mole
ratio
molesof
basethat
(number
reactwith one moleof acid)
n
3. Onestudent accidentally“overshot”the endpointand added23.90mLof 0.215MNaOH.Is
the calculatedconcentrationofHCIlikelyto be too high or too lowas a result ofthis error?
Materials
Hydrochloricacid solution, HCI,unknownconcentration, 40 mL
Phenolphthalein indicator solution, 0.5%, 1 mL
Sodium hydroxide(standard)solution, NaOH,approximately0.1 M, 75 mL*
Water,distilled or deionized
Beakers,250-mL,2
pH Sensor or pH meter
Buret, 50-mL
Computer interface system (LabPro)t
Erlenmeyerflask, 125-mL
Computeror calculator for data collectiont
Pipet and pipet bulb or filler,10-mL
Datacollectionsoftware(LoggerPro)t
Stirrer and stirring bar (optional)
Washbottle
*Consultyour teacher to obtain the preciseconcentration ofsodium hydroxide.
tOptional
FlinnChemTopic®Labs
—
AcidsandBases
Page3 ClasskTitratioh
Safety Precautions
Dilute hydrochtoricacidand sodium hydroxide silutions are irritating to skin and eyes.
Notify your teacherand clean up all spills immediately with largeamount. of water.
Phenolphthalein is an alcohol-basedsolution and is flammable.It is moderately toxic by
ingestion. Keep away from flames and other ignitIonsources.Avoid contact of all chemicals
with eyes and skin and wash hands thorOughlywith soap and water beforeleaving the labo
ratory. Wearchemicalsplashgoggles and chemical-resistantgloves and apron.
Procedure
S
Part A. Measuring a Titration Curie
1. Usinga volumetricpipet, transfer 10.00mLofthe “unknown”hydrochloricacidsolution
to a 250-mLbeaker.
2. Adddistilledwaterto the solution in the beakeruntil the liquidvolumeis at about the
150-mL mark.
3. Carefullyadd‘astirrin bar to the solution and‘placethe beaker on a magnetic stirrer.
4. ObtainabOut75 mLofstandardsodium hydroxidesolution and record the precisemolar
ity of the solutionin DataTableA.
5. Obtain aclean50-mLburet and rinse itiwith two 5-mL portions of standardsodium
hydroxidesolution.
6. Clampthe buret to a ring stand and placea “waste”beaker,under the buret. Fill the buret
to abovethe zero markwith sodium hydroxidesolution.Openthe stopcockto allowany
air bubblesto escapefrom the tip. Closethe stopcockwhen the liquid levelin the buret is
betweenthe 0- and 1O-mLmark.
7. Recordthe preciselevel (initialvolume) ofthe solutionin the buret. Note:Volumesare
read from the top downin a buret. Alwaysreadfromthe bottom of the meniscusand
nber
remei
to includethe appropriatenumber of significantfigures.(SeeFigure 1.)
8. Positionthe buret over the beaker so that the tip of the buret is within the beakerbut at
least 2 cm abovethe liquid surface.
9. If using a pH sensor and computeror calculator-interfacesystem, proceedto step 10.If
usinga pH meter,proceedto step 22.
mL
(not
•24.55)
Figure 1. How to read a buret volume.
ClassicTitration
Figure 2. Setup.
ClassicTitration Page4
—
-
.Computer- or calculator-based data collection:
10. Insert a pH sensor into the beaker.Adjust the levelof the sensor so that the bottohi 2 cm
of the sensor is submerged in liquid. Do not allowthe sensor to come in contact with the
stirring bar. Clamp the sensor in position. (SeeFigure 2.)
ii. Plug the pH sensor into the interface system.
•
12. Open andformat a graph in the data collectionsoftwareso that the y-axis reads pH on a
scaleof 0—14.
13. Set the x-axisfor time in seconds.Set the minimum and maximumtime valuesat 0 and
•300 sec, re
pectively.
• 14. Set the time intervalto take a reading everysecond.
15. Waitabout 30 secondsto allowthe pH’sensorto adjust to the solution pH, then press
start to begincollectingdata.
•
16. Immedi
tely
open the stopcockon the buret and beginaddingsodium hydroxidesolution
slowlyto the flask.Adjustthe drip rate from the buret to addabout one drop per second.
17. Trynot to change the drip rate during the course of the titration.
18. The system should automaticallyrecord data for the allottedtime (300sec), then stop.
•19. Closethe stopcockon the buret when data colleFtionhas ended.Removethe sensor from
the beakerand rinse it with distilledwater.
20. Obtain a printout of the titration graph from the cOmputer.
.’e the sodifimhydroxidesolution in the
21. Note: If proceedingto Part Bat this time, sa
buret. Skipsteps 1—3in Part Band proceedimmediatelyto step 4.
Alternative procedure using a pH meter:
22. Insert the pH electrodefrom a pH meter into the beaker.Adjustthe levelof the electrode
so that the bottom 2 cm of the electrodeis submerged in liquid.Donot allowthe elec
trode to come in contactwith the stir bar. (SeeFigure2.)
23. Whenthe pH reading has stabilized,record the initial pH of the solution in DataTableA.
24. Addabout 1 mLof sodium hydroxidesolution to the beaker.Recordthe exact buret read
ing in DataTableA.
25. Recordthe pH of the solutionin DataTableA.
26. Addanother 1-mLincrement ofsodium hydroxidesolution.Recordboth the buret read
ing and the pH in DataTableA.
27. Continue adding sodium hydroxidein 1-mLportions. Recordboth the buret reading and
the pH after each addition.
28. Whenthe pH beginsto increaseby more than 0.3 pH units after an addition, decreasethe
portions of sodium hydroxideaddedto about 0.2 mL.
29. Continue adding sodium hydroxidein about 0.2 mL increments.Recordboth the buret
readingand the pH after each addition.
FlinnChemlopic®
Labs AcidsandBases
—
Page5—ClassicTitration
LA3S
30. Whenthe pH change is again about 0.3 pH units, resume adding the sodium hydroxide
in 1-mLincrements.Continue to record both.theburet reading and the pH after each
addition.
31. Stop the titration when the pH of the solution is greater than 12.Recordthe finalvolume
of solution in the buret and the final pH.
32. Note: If proceedingto Part Bat this time, savethe sodium hydroxidesolution in the
buret. Skipsteps 1—3in Part B and proceedimmediatelyto step 4. It may be necessaryto
refillthe buret at this point.
Part B. Analyzing an Unknown
1. Obtainabout 75 mLof standard sodium hydroxidesOlutionand record the precisemolar
ity of the solutionin DataTableB.
2. Rinse a clean50-mLburet with two 5-mL portionsofthe sodium hydroxidesolution.
3. Clamp the buret to a ring stand and placea “waste”beakerunder the buret. Fillthe buret
to abovethe zero mark with sodium hydroxidesolution. Open the stopcock to,allowany
air bubblesto escapefrom the tip. Closethe stopcockwhen the liquid levelin the buret is
betweenthe 0- and 1O-mLmark.
4. Recordthe preciselevel (initialvolume) ofthe solutionin the buret in DataTableB.
Note: Volumesare read from the top downin a buret. Alwaysread from the bottom of the
meniscusand rememberto includethe appropriatenumber of significantfigures.(See
Figure 1.)
5. Usinga pipetor graduatedcylinder,transfer 10.00mL of the unknown hydrochloricacid
solution to a 125-mLErlenmeyerflask.
6. Addabout 20 mLof distilledwater to the flask,followedby 2 drops ofphenolphthalein
indicator.
7. Positionthe flaskunder the buret so that the tip ofthe buret is insidethe mouth of the
flask.Placea pieceofwhite paper under the flaskto make it easier to detect the color
change ofthe indicatorat the endpoint.
8. Open the stopcockto allowapproximately5—8mLofthe sodium hydroxidesolution to flow
into the flaskwhilecontinuouslyswirlingthe flask.Observethe color changesoccurring.
9. Continueto addsodium hydroxideslowly,drop-by-drop,while swirling the flask.Usea
wash bottle to rinsethe sidesof the flaskwith distilledwater during the titration.
10. When a faint pink color appears and persists for 10secondsor more while swirling the
flask,the endpointhas been reached.Closethe stopcockand record the finalburet read
ing for TrialI in DataTableB.
11. Pour the solutionout of the flaskinto the sinkand rinse the flaskwith distilledwater.
12. Repeatthe titration (steps4—11)with a secondsampleof hydrochloric acid. Recordall
data for Trial2 in DataTableB.
13. (Optional)11time permits, completea third trial.
ClassicTitration
ClassicTitration Page6
—
336.
Name:
-
b
C1ass/L
Period:
ClassicTitration Lab
Data Table A. Measuring a Titration Curve
Standard NaOH Concentration
Initial Buret Reading
•
.
Initial pH
,
Buret
,
Buret
Reading
pH
•
Reading
pH
(continued)
Data Table B. Analyzing an Unknown
Standard NaOH Concentration
,
Initial
Buret
Reading
Final
Buret
Reading
Trial1
Trial2
,
Trial3 (optional)
Flinn ChemTopic®Labs
—
AcidsandBases
Page7—ClassicTitration
Post-LabQueshons (Usea separatesheet of paper to answerthe followingquestions.)
Part A. Measuring a Titration Curve
1. Obtain a graph ofthe pH curve from Part A.Note:Ifthe pH data was collectedmanually
using a pH metei draw a graph of pH (y-axis)versusvolumeof NaOHadded(x-axis).
Labelthe axes,including the appropriateunits.
• 2. Describein wordshowthe pH changesduring the course of the titration. Bespecific.
3. Whatis the theoretical equivalencepoint for the titration of a strong acidwith a strong
base?What happensto the pH of the solution as it approachesthe equivalencepoint?
4. Drawa horizontalline across fromthe pHat the equivalencepoint to the titration curve,
followedbya verticalline downfrom this point on the titration curve to the x-axis.
5. Phenolphthaleinis often used to detect the endpoint in the titration ofa strong acidwith
a strong base.It changes color fromcolorlessto pink at about pH 9. Drawa horizontal
line across from pH 9 to the titration curve, followedbya vertical line downfom this
point on the titration curve to the x-axis.Is there a large differencebetweenthe volume
of NaOHaddedat the equivalencepoint versus the indicatorendpoint?Whyor why not?
Part B. Analyzing an Unknown
6. Calculatethe unknown molarityof the HCIsolution (Ma) for trials 1 and 2, as well as the
averagemolarity.Note: SeePrelabQuestion#2.
7. Considerthe followingpotential sources of error in the titration. Fill in the blank:
“H”ifthe error wouldhavecausedthe calculatedmolarityof HCIto come out too high.
I to come out too low.
“L”if the error wouldhavecausedthe calculatedmolarityof HC
“N”if the error wouldhavehad no effecton the the calculatedmolarity of HCI.
a. Therewasa littledistilledwaterin the Erlenmeyerflaskbeforethe titration began.
b. There wasa little HCIin the Erlenmeyerflaskbeforethe titration began.
c. There wasa little distilledwaterin the buret beforeyou began and you forgotto rinse
it out with NaOH.
_____
d. Youadded3 drops of phenolphthaleininsteadof2 drop&
e. SomeNaOHsolutiondrippedinto the Erlenmeyerflaskbeforethe initialNaOHvolume
was measured.
______
f. Whileyou weretitrating, some NaOHdrippedout onto the table instead of into the
Erlenmeyerflask.
_____
8. Completethe followingsentenéewith a creativebut appropriateanalogy:Performinga
titration withou an indicatoris like
__________________
: I;ss
Ii I i;i
Page 1 Measuring Acid Strength
—
Measuring Acid Strength
Ka Values of Weak
Acids
Introduction
Acidsvary greatly in their strength—-theirabilityto ionize or produce ionswhen dissolvedin
water.What factorsdetermine the strength of an acid?In this experiment,the strength of
acidswill be measured bydetermining the equilibrium constants for their ionization reac
tions in water.
•
Concepts
•
•
WeakacId
•
Conjugate base
Equilibrium constant
•
Neutralizationreaction
Background
•
The modern Brønsteddefinitionof an acid relies on its abilityto donate hydrogen ions to
other substances.When an acid dissolvesin water, it donates hydrogen ions to water mole
cules to form H
3O ions. The general form’ofthis reaction, called an ionization reaction, is
shown in Equation 1, where HAis the acid and A- its conjugate baseafter loss of a hydrogen
ion. The double arrowsrepresent a reversiblereaction.
I
HA(aq) + H
20(l)
A(aq)
+
Equation 1
(aq)
3O
H
The equilibriumconstantexpression(Ka)forthe reversibleionizationof an acidis givenin
Equation2.,The squarebracketsreferto the molar concentrationsofthe reactantsand products.
[A-J[H 0j
[HA
Ka
Equation 2
Not all acids, of course, are created equal. The strength of an acid dependson the value of its
equilibrium constant Ka for Equation 1. Strong acids ionizecompletelyin aqueous solution.
The valueof Ka for a strong acid is extremelylarge and Equation 1 essentiallygoes to com
pletion—onlyH
3O and A- are present in solution. Weakacids, in contrast, ionize only par
tially in aqueous solution. The value of Ka for a weakacid is much lessthan one and
and H
Equation 1 is reversible—allspecies(HA,A—,
are present at equilibrium.
)
3O
Polyproticacidscontain more than one ionizablehydrogen. Ionization of a polyproticacid
occurs in a stepwisemanner, where each step is characterized by its own equilibrium con
stant (Kai,Ka
2 etc.). The second reaction (removalof the second acidic hydrogen)always
occurs to a much smaller extent than the first reaction, and so Ka2 is alwayssignificantly
smaller than Kai• Sulfuricacid 4)(H
and phosphoric acid 4)(H
are examplesof
2S0
3P0
polyproticacids.
MeasuringAcidStrength
2A(aq)
H
+
20(J)
H
HAiaq)
+
70(l)
H
t
HAiaq)
2iaq)
A
+
+
(aq)
3O
H
(aq)
3O
H
•
1
K
=
=
MeasuringAcid Strength Page 2
-
33C
The ionizationconstant of a weakacid can be determined experimentallybymeasuring the
3O concentration in a dilute aqueous solution of the weakacid.This procedure is most
H
ofthe weakacid and its cqnjugate
ts
accurate when the solution contains equal molar amour
base.Consideracetic acid as an example.Aceticacid (CH
3COOI-l) and the acetate anion
The
acid—base
equilibriumconstant expressionfor
p
air.
represent
conjugate
a
3COOj
(CH
ionizationof acetic acid is shown in Equation 3. If the concentrationsof acetic acid and
acetate ion are equal, then these two terms cancelout in the equilibriumconstant expres
sion, and Equation 3 reduces to Equation 4.
Ka
=
COO-]1H
jECH
3O
3COOHJ’,
[CH
Equation 3
0h1
3
Ka =[H
Equation4
In this experiment, solutionswillbe prepared in which the molarconcentrations of an
unknown acid and its conjugate base are equal. The pH of thesesolutionswill then equal the
PKafor the acid.The definitionof PKais closelyrelated to that of pH. Thus, pH =
and PKa = ogK
The substancesthat willbe tested are salts of polyptotic,
30’i
—log[H
.
acids that still contain an ionizablehydrogen. Sodium bisulfate,for example,is a weakacid
ion is a weakacid—the equilibrium cdn
ions. The HS0
salt; it contains Na and HSO
4
4stant for ionizationof HSO
correspondsto Ka
42 for sulfuricacid.
2Saq)
4(H
0
+
20(l)
H
4(aq)
HS0
+
20(l)
H
t
4(aq)
HS0
42aq)
SO
+
+
(aq)
3O
H
Kai
(aq)
3O
H
2
Ka
=
____________
4
j-iso
Experiment Overview
The purpose of this experimentis to measure the pKavaluefor ionizationof an unknown
weakacid. Solutions containingequal molar amounts ofthe weakacid and its conjugate
basewill be preparedby “half-neutralization”of the acid.Their pH valueswill then be meas
ured and used to calculatethe PKavaluefor the unknownand determine its identity.
Pre-Lab Questions(Usea separate sheet of paper to answer the followingquestions.)
Phosphoricacid is a triprotic acid (three ionizablehydrogens).Thevalues of its stepwiseion
.
,
and Ka
izationconstants are Kai = 7.5 x iO
,
3 ‘= 4.2 x iO’
2 = 6.2 x 1O
Ka
1. Writethe chemicalequation for the first ionizationofphosphoricacid with water.
2. Writethe equilibriumconstant expression(Kai) forthis reaction.
3. Whatwouldbe the pH of a solutionwhen 41[H
3P0
Note: pH = —log
J.
3O
H
2P?0
411H
FlinnChemTopic®
Labs
—
AcidsandBases
Page3—MeasuringAcid Strength
•
O.
c3
•
4. (Optional) Phenolphthaleinwouldnot be an appropriateindicatorto use to determineKai,
•
for phosphoricacid. Whynot? Choosea suitableindicatorfrom the followingcolorchart.
pH
Indicator
1
2
F
3
5
Colorless
Phenolphthalein
•
MethylRed
Red
,
OrangeIV
Orange
Peach
6
.
8
7
9’
10
Pink
,
j
11
Red
Yellow
Orange
Yellow,
Materials
Unknownweakacids, about 0.5 g each
Phenolphthaleinsolution, 0.5%, 1 mL
Sodium hydroxidesolution, NaOH,0.1 M, 15 mL
Washbottle and distilled or deionizedwater
pH Meter
Weighingdishes,2
Balance,centigram (0.01 g precision)
Beaker,150-mL,I
Erlenmeyerflask,125-mL,
Graduatedcylinder,50- ‘or100-mL
Pipets,Béral-type,2
Stirring rod
•,
,
Safety Precautions
Acids and basesare skin and eye irritants. Avoid contact of all chemical with eyes and skin.
Inform the teacher and clean up all acid and base spills immediately. Phenolphthalein is an
alcohol-based solution and is flammable. Keep the solution away from flames. Wear chemical
splash goggles and chemical-resistant gloves and apron. Wash hands thoroughly with soap
and water before leaving the laboratory.
Procedure
I. Labeltwo weighingdishes#1 and #2.
2. Obtainan unknownweakacidand record the unknownnumber in the data table.
3. Measureout a small quantity (0.15—0.20
g) ofthe unknowninto each weighing dish.
Note: It is not necessaryto knowthe exact mass of each sample.
4. Usinga graduatedcylinder,measure precisely50.0mL of distilledwater into a 150-mL
beaker.
5. Transfersample#1 to the water in the beakerand stir to dissolve.
6. Usinga graduatedcylinder,transfer precisely25.0mL of this acid solution into an
Erlenmeyerflask.
7. Add3 drops of phenoiphthaleirisolution to the acidsolution in the Erlenmeyerflask.
8. Usinga Beral-typepipet, add sodium hydroxidesolutiondropwiseto the flask.Gently
swirlthe flaskwhile adding the sodium hydroxidesolution.
MeasuringAcidStrength
Measuring Acid Strength
-
Page4
(34/.
9. Continue adding sodium hydroxidedropwiseand swirling the solutionuntil a faint pink
color persiststhroughout the solution forat least5 seconds.This is called he endpoint.
Note:A pink color developsimmediatelywhen the baseis added. hut fadesquicklyonce
the solution is swirled.When nearing the endpoint, the pink color beginst fademore
slowly.Proceedcautiouslywhen nearing the endpoint.SO as not to”overshoot”it.
At this point the solution in the beakercontains exactlyone-halfof the originalamount of
IA. The Erlenmeyerflaskcontains an equal
acid, essentiallyall ofwhich is in the acid form, ibaseA
obtained
neutralization.
amount of the conjugate
by
10. Pour the contents of the flaskbackinto the beaker.Pour the solution backand forth a few
times to mix.Note:It is important to transfer the solution as completelyas possiblefrom
the flaskbackinto the beaker.
11.,Usinga pH meter,measure the pH of the resulting solution in the beaker,whichcontains
equal molar amounts of the acidand its conjugatebase.Recordthe pH in thedatatable.
12. D
spose of thebeakercontentsdownthedrainandrinseboththebeakerandthe
Erlenmeyerflaskwith distilledwater.Dry the beakerwith a paper towel.
ple
13. Repeatsteps 4—12using sari
#2.
F’linnChemTopic®
Labs Acidsand Bases
—
Page5- MeasuringAcid Strength
i
(31
Name:
___________________
Class/LabPeriod:
_________
Measuring Acid Strength
Data Table.
UnknownLabel
Thai
pH
Sample#1
Sample#2
.
Post-Lab Questions (Usea separatesheetof paperto anstherthe followingquestions.)
1. Averagethe pH readings for each trial (samples#1 and 2) to calculatetheaverage PKa
value for your unknownweakacid.
2. Comment on the precision(reproducibility)of the PKa determinations.Describesources
of experimentalerror and their likelyeffecton the measured PKa(pH)values.
3. The followingtable lists the identitiesof thepossible unknowns in this experiment.
valuefor each acid.Note: PKa =
Completethe table bycalculating the
4
Acid
Formula
Potassiumdihydrogenphosphate
2PO
4KH
Potassiumhydrogensulfate
4
KHSO
P
Ka
K
of 4H
3P0
=
6.2 x 10
of 4H
2S0
=
1.2 x 10-2
Potassiumhydrogenphthalate
4O
8H
KHC
K
of H
840
2C
=
3.9 x 1O
Potassiumhydrogentartrate
6
O
4H
KHC
K
of H
406
2C
=
4.6 x iO
.
4. Comparethe Picavaluefor your unknownwith the informationin the table. Determine
the probableidentityof the unknown.
5. Write separateequations for the unknown dissolvingin water and for the ionization reac
tion of the weakacidanion that this salt contains.
6. Whywas it not necessaryto knowthe exact mass of each acid sample?
7. Whywas it not necessaryto knowthe exactconcentrationofthe sodium hydroxidesolution?
8. Whywas it necessaryto measure the exactvolume of distilledwater used to dissolvethe
acid,as well as the exactvolume,of solutiontransferredfrom the beakerto the
Erlenmeyerflask?
1;isiirii
.AciFSI lL(
ii
Page 1 Heats of Reaction and Hess’sLaw
-
3.
L3
Heats of Reaction and Hess’sLaw
Small-Scale Calorimetry
Introduction
The reaction of magnesium metal with air in a Bunsenburner flameprovidesa dazzling
demonstration of a combustion reaction. Magnesiumburns with an intense flamethat
produces a blinding white light. This reaction was utilizedin the earlydays of photography
as the source of “flashpowder”and later in flashbulbs.It is still used today in flaresand fire
works. Howmuch heat is produced when magnesium burns?
Concepts
• Heat of reaction
•
Heat of formation
• Hess’sLaw
•
Calorimetry
Background
Magnesiumreacts with oxygenin air to form magnesiumoxide,according to Equation 1.
Mg(s) + ½0
2(g)
—
MgO(s) + heat
Equation 1
Asmentioned above,a great deal of heat and light are produced—thetemperature of the
flame can reach as high as 2400 °C.The amount of heat energy produced in this reaction
cannot be measured directly in the high school lab.It is possible,however,to determine the
amount of heat producedby an indirect method, using Hess’sLaw.
The heat or enthalpychange for a chemical reaction is calle
the heat of reaction (H
i
).
The enthalpychange—definedas the differencein enthalpybetweenthe productsand
reactants—is equalto the amount of heat transferredat constant pressure and dies not
depend on howthe transformation occurs. This definitionof enthaipy makes it Øossibleto
determine the heats’of reaction for reactions that cannotbe measured directly.Accordingto
Hess’sLaw,if the same overallreaction is achievedin a seriesof steps, rather than in one
step, the enthalpychange for the overallreaction is equalto the sum of the enthalpy
changes for each step in the reactionseries. There are tvo basic rules for calculating the
enthalpy change for a reaction using Hess’sLaw.
Equations can be “multiplied”bymultiplyingeachstoichiometriccoefficientin the bal
anced chemicalequationby the same factor.Theheat of reaction (All) is proportionalto
the amount of reactant. Thus, if an equation is multipliedby a factor of two to increase
the number of molesof product produced,then the heat of reactionmust also be multi
pliedbya factoroftwO.
•
Equations can be “subtracted”b reversingthe reactants and products in the balanced
chemical equation.The heat of raction (AH)for the reversereaction is equal in magni
Iu(I(’
Heatsof Reactiti
I Ie’.
hut oppositen sign’to that of the forwardreaction.
I.. v
Heats of Reactionand Hess’s Law Page 2
-
(34#
Considerthe followingthree reactions:
Mg(s) + 21-ICI(aq)
9(aq)
MgCI
—*
MgO(s)
9(g)
l-1
+
+
21-ICI(aq)
9(g)
½0
—
—
9(aq)
MgCI
Equation A
9(g)
I-1
+
+
Equation B
0(!)
9
F1
Equation C
20(l)
H
It is possibleto express the combustion of magnesium (Equation 1) as an algebraicsum of
EquationsA,B, and C. ApplyingHess’sLaw,therefore, it should also be possibleto deter
mine the heat of reaction for Equation 1 by combining the heats of reaction for Equations
A—C
in the same algebraicmanner. Note: Chemical equations may be combined byaddition,
subtraction, multiplication, and division.
Experiment Overview
The purpose of this experimentis to use Hess’sLawto determine the heat of reactionfor the
combustionof magnesium(Equation 1). The heats of reactionfor EquationsAand Bwillbe
measured by calOrimetry.The heats of reactionfor these tworeactionswill then be combined
algebraicallywith the heat of formationofwater (EquationC)to calculatethe heat of reaction
for the combustionof magnesium.
Pre-Lab Questions
1. Reviewthe Background section. ArrangeEquationsA—C
in such a way that they add up to
Equation 1.
2. UseHess’sLawt&expressthe heat of reactionfor Equation 1 as the appropriatealgebraic
surn of the heats of reactionfor EquationsA—C.
.
The heat of reactionfor EquationC is equalto the standardheat of formationofwater.The
heat offormationof a compoundis definedas the enthalpychange forthe preparationof
one moleof a compoundfromits respectiveelementsin their standardstates at 25 °C.
Chemicalreferencesourcescontaintablesof heats offormationfor many compounds.
Lookup the heat offormationofwaterin your textbookor in a chemicalreferencesource
such as the CRCHandbookof Chemistryand Physics.
Materials
HydrochloricAcid,HCI,1 M, 60 mL
Magnesiumribbon,Mg,7-cmstrip
Magnesiumoxide,MgO,0.40 g
Balance,centigram (0.01g precision)
Calorimeter,small-scale
Digital thermometer or temperaturesensor
Forceps
Graduatedcylinder,25- or 50-mL
Metric ruler, markedin mm
Scissors
Spatula
Stirring rod
Washbottle and water
Weighingdish
Precautions
i
Safet
Hydrochloricacid is toxic by ingestion and inhalationand is corrosiveto skin and eyes.
Magnesium metal is a flammablesolid. Keepaway from flames.Do not handle magnesium
metal with bare hands;Weqrchemicalsplash gogglesand chemical-resistantgloves and
apron. Washhands thoroughlywith soap and water beforeleavingthe lab.
FlinnChemTopic
Labs Thermochemistry
’
—
Page3 Heats of Reactionand Hess’sLaw
-
(343:
Procedure
Recordall data for PartsA and B in the Data Table.
Part A. Reaction of Magnesium with Hydrochiork Acid
1. Obtain a 7-cm strip of magnesium ribbon and cut it into two piecesof unequal length,
roughly 3- and 4-cm each.Note: Handlethe magnesium ribbon using forceps.
2. Measurethe exactlength of each piece of magnesium ribbon to the nearest 0.1 cm.
3. Multiplythe length of each piece of Mg ribbon by the conversionfactor (g/cm)provided
by your teacher to obtain the mass of each piece of Mg.
4. Massa clean,dry calorimeter to the nearest 0.01 g.
5. Usinga graduatedcylinder,add 15 mL of 1 M hydrochloricacidto the calorimeterand
measure the combinedmass of the calorimeterand acid.
6. Usinga digital thermometer or a temperature sensor, measure the initial temperature of
the hydrochloricacidsolution to the nearest 0.1 °C.
7. Addthe first (shorter)piece of magnesium ribbonto the acidand stir the solution until
the magnesiumhas dissolvedand the temperature of the solutionremains constant.
8. Recordthe final temperature of the solution to the nearest 0.1 °C.
9. Rinsethe contents of he calorimeterdownthe drainwith excesswater.
10. Dry the calorimeterand mass it again to the nearest0.01 g.
11. Repeatsteps 5—9using the second (larger)piece of magnesiumribbon.
Part B. Reaction of Magnesium oxide with Hydrochloric Acid
12. Massa clean,dry calorimeterto the nearest 0.01 g.
13. Usinga graduatedcylinder,add 15mLof 1 MHCIto the calorimeterand measure the
combinedmass ofthe calorimete and hydrochloricacid.
14. Tarea small weighing’dishand addabout 0.20 g of magnesiumoxide.Measurethe exact
mass of magnesiumoxideto the nearest 0.01 g.
15. Usinga digitalthermometer or a temperature sensor,measure the initial temperature of
the hydrochloricacidsolutionto the nearest 0.1°C.
16. Usinga spatula,addthe magnesiumoxideto the acid.Stir the reaction mixture until the
temperature remainsconstant forseveralfive-secondintervals.Recordthe final tempera
ture ofthe solutionto the nearest 0.1 °C.
17. Pour the reactionmixturedownthe drainwith excesswater.Rinseand dry the calorimeter.
18. Repeatsteps 12—16
usinga secondsampleof magnesiumoxide.
19. Washthe contentsofthe calorimeterdownthe drainwith excesswater.
HeatsofReactionandHess’sLaw
Heats of Reaction and Hess’sLaw
-
Page 4
(346.
Name:
Class/LabPeriod:
Heats of Reaction and Hess’sLaw
Data Table
ReactionA (Mg + HC
I)
Trial 1
.
Trial2
ReactionB (MgO + HC1)
Trial2
Trial1
Massof Calorimeter(g)
Massof Calorimeter+
HC1Solution (g)
Massof Mg(ReactionA) or
MgO(ReactionB) (g)
InitialTemperature(°C)
FinalTemperature(°C)
Post-Lab Calculationsand Analysis (Show all work on a separatesheet of paper.)
Construct a ResultsTableto summarize the results of all calculations.For each reaction and
trial, calculate the:
1. Massof hydrochloricacidsolution.
2. Totalmass of the reactants.
3. Change in temperature, AT =
—
Tinjtiai•
4. Heat (q) absorbedby the solutionin the calorimeter.Note: q = m x s x AT,where S is
the specificheat of the solutionin J/g°C. Usethe total massof reactants for the mass (m)
and assume the specificheat is the sameas that of water,namely,4.18 J/g
°C.
5. Numberof molesof magnesiumand magnesiumoxidein ReactionsAand B, respectively.
6. Enthalpychange for each reactionin units of kilojoulesper mole (Id/mole).
7. Averageenthalpychange(heatof reaction,)AH forReactionsAandB.Note: Theenthalpy
changeis positivefor an endothermicreaction,negativeforan exothermicreaction.
8. UseHess’sLawto calculatethe heat of reactionfor Equation1.Hint: See your answer to
PreLabQuestion#2.
9.’The heat of reactionfor Equation1 is equalto the heat offormationof solid magnesium
oxide.
(a) Lookup the heat of(ormationof magnesiumoxidein your textbookor a chemical
referencesource.
(b) Calculatethe percenterror in your experimentaldeterminationof the heat of reaction
for Equation 1.
FlinnChemlopic”Labs Thermochemistry
—
Page 1 The EquilibriumConstant
—
(317
The Equilibriumconstant
Complex Ion Formation
Introduction
For any reversiblechemicalreactionat equilibrium,the concentrationsofall reactants and
products are constant or stable.Therewillbe no further net change in the amounts of reac
tants and products unlessthe reactionmixture is disturbed in someway.The equilibrium
of the position of equiIibriu for any reversible
constant providesa mathematical descrip
iôn
chemical reaction.Whatis the equilibr
constart and how can it be determined?,
un
Concepts
Chemical equilibrium
•
Equilibriumconstant
Complex-ionreaction
•
Colorimetry
Background
Any reversiblereactionwill eventuallyreach a position of chemical equilibrium. In some
cases, euilibrium favorsproducts and it appears that the reaction proceedsessentiallyto
completion. The amount of reactants remaining under these conditionswill be very small.
In other cases, equilibrium favorsreactants and it appears that the reaction occurs only to a
slight extent. Underthese conditions, the amount of products present at equilibrium will be
very small.
These ideas can be expressedmathematicallyin the form of the equilibriumconstant.
Considerthe followinggeneral equation for a reversiblechemical reaction:
aA + bB t
cC
+
dD
Equation 1
The equilibriumconstant Keq forthis general reactionis givenby Equation2, wherethe
square bracketsreferto the molar concentrationsofthe reactants and products at equilibrium.
Equation 2
Keq =
The equilibriumconstant gets its name from the fact that for any reversiblechemical
reaction, the valueofKeqis a constant at a particular temperature. The concentrations of
reactants and productsat equilibrium will vary,dependingon the initial amounts of
materials present. The specialratio of reactants and products describedby Keqwill always
be the same, however,as long as the system has reached equilibrium and the temperature
does not change. Thevalueof Keqcan be calculatedif the concentrations of reactants and
products at equilibriumare known.
The reversiblechemicalreaction of iron(IIl) ions (Fe
3
with thiocyanate ions (SCN—)
pro
vides a convenientexampleto determine the equilibrium constant for a reaction.Asshown
3 and SCN ions combine to form a special type of combined or “complex”
in Equation3, Fe
2.
ion having the formulaFeSCN
+ SCN1aq)
3aFe
q)
Paleyellow Colorless
TheEquilibriumConstant
2(FeSCN
aq)
Blood-red
Equation 3
The Equilibrium Constant Page2
—
The equilibrium constant expression or thi
Keq
‘
i’
lu
i
un 4.
Equation4
=
I
Ky n
3
Iiiuwn cuncentrations of Fe
The value of Keqcan be determined experiment
xii
iIlv
and SCN—
ions and measuring the concentration ul l’cSCN uns equilibrium. Asroted in
Equation3, the reactant ions are paleyellowand colurless. t’csptcl ively,while the product
2
ions are blood-red.The concentration of FeSCN
complex iuiis equilihriunrwill be pro
portional to the intensity of the red color.
A specialsensor or instrument called a colorimeter cai be used to measure the absorbance
of light by the red ions. The more intense the red color,the greater the absorbancewill be.
The wavelengthof light absorbed by the red ions is about470 rim. Noneof the other ions
present in solution absorb light at this wavelength.Aslongas the same size container is
used to measure the absorbanceof each solution, the absorbancewill be directly proportion
2
al to the concentration of F’eSCN
ions.
Experiment Overview
The purpose of this experiment is to calculatethe equilibriumconstant for the reaction of
iron(1II)ions with thiocyanate ions. The reaction will be tested under different copditionsto
determine if the equilibriumconstant alwayshas the same numericalvalue. There are two
parts to the experiment.
0
In Part A,a referencesolution and a series of test solutionswillbe prepared. The’
3 ions with a known
referencesolution will be preparedbymixinga large excessof Fe
amount of SCN-ions. Accordingto LeChâtelier’sPrinciple,the large excess of iron(III) ions
2
should effectivelyconvert all of the thiocyanate ions to the blood-redFeSCN
complexions.
2
The concentration of FeSCN
complexions in the referencesolution will essentiallybe
equalto the initial concentration of SCN-ions. The test solutionswill be prepared by mixing
3 ions with differentconcentrationsof SCN—
ions. Thesesolu
a constant concentration of Fe
2
FeSCN
ionsat equilibrium.
tions willcontain unknownconcentrationsof
In Part B, the absorbariceofboth the referencesolutionandthe test solutionswillbe measured
2
in the test solutionswillbe calculated
bycolorimetry.The unknownconcentrationsof FeSCN
the
absorbanceofthe
referencesolution.
their
absorbance
to
bycomparing
readings
Labs
FlinnChemlopic
’
—
Equilibrium
Page 3 The EquilibriumConstant
-
‘I
Pre-Lab Questions (Show all work on a separate sheet of paper.)
1. The referencesolution in Part Awillbe prep
red
by mixing 9.00mL of0.200M Fe(NO
)3
3’
solution and 1.00mL of 0.00
MKSCNsolution.The concentration pfFe
ions in the
0
ence
efe
before any reaction occurscan be calculatedusing the so-called
solution (M
2)
“dilution equation,”as shown below.
1V
M
1N
V
2
V
=
=
=
=
Dilution Equation
2V
M
concentrationof solution before mixing = 0.200M Fe(N0
3)
volumeofsolution before mixing = 9.00mL
finalvolumeof referencesolution after mixing = 9.00 + 1.00mL
M
1V
2
V
=
(0.200 M)(9.00mL
)
‘(10.00‘mL)
=
=
10.00mL
0.180M
Usethe dilution equation to calculatethe concentrationof SCN ions in the reference
solution beforeany reaction occurs.
2
2. “The equilibriumconcentrationof FeSCN
ions in the referencesolution is essentially
concentrationof
the
SCN
ions
in
solutionbefore
to
I
equ
any reactionoccurs.”Use
LeChâtelier’sPrincipleto explainwhy this statement is true.
3 and SCN-stocksolutionsthat willbe
3. The followingtablesummarizesthe volumes of Fe
mixedtogetherto prepare the test solutions in Part A.Usethe dilution equation to calcu
3’
late the concentrationsof Fe
and SCN ions in each test solution beforeany reaction
in scientificnotation in the DataTable.
s
occurs. Enter the results of these.calculatio
of
Hint: The finalvolume (V
each test solutionis 10.00mL.
2)
I
Volumeof 0.0020 M
Solution
3)
Fe(N0
Volumeof 0.0020 M
KSCNSolution
Testsolution #1.
5.00mL
1.00mL
4.00mL
Testsolution#2
5.00mL
2.00mL
3.00mL
Testsolution #3
5.00mL
Sample
Test solution #4
,
3.00mL
5.00mL
4.00mL
5.00mL
5.00mL
,
Testsolution #5
TheEquilibriumConstant
Volumeof Distilled
WaterAdded
‘
,
2.00mL
1.00mL
0.00mL
The EquilibriumConstant Page4
-
MateriaIs_.
0.0020M,30 mLt
0.200M, 10 mLt
Iron(HI)nitrate, Fe(N0
3),
Potassium yariate,
KSCN,0.0020M, 1 mL
thio
Water,distilled or deionzied
Colorimeter sensor or spectrophotometer
Computer interface system (LabPro),15*
Computeror calculator for data collection, 15*
Datacollectionsoftware (LoggerPro)*
*Not required if spectrophotometer is used
tContajns 1 Mnitric acid as the so!vent.
Irón(III) nitrate, Fe(N0
3),
rs
ik&
I3e
6
ftst tuhes,50—m
L,
Cuvetleswith uk,
c
nr l;r
rii
.Labeliii r n
pen
5- or 10-mL,3
il-tvpe,
Pipets, serulngic
Pipet bulb or pipct filler
Stirring rod
Tissuesor lens paper, lint-free
Thermometer
Washbottle
Safety Precautions
Iron(III) ni
trate solution contains I N nitric acid and is d corrosiveliquid;it willstain skin
and clothing.Notify the teacherqnd clean up all spills immediately.Potassiumthiocyanate
is toxic by ingestion; it an generatepoisonous hydrogen cyanidegas if heated strongly.
Avoid contact of all chemicalswith eyes and skin. Wearchemical splashgoggles and
chemical-resistantgloves and apron. Washhands thoroughly with soap and water before
leavingthe laboratory.
Procedure
I
Part A. Preparing the Solutions
1. Obtainsix50-mLbeakersor large test tubes and labelthem #1—6for the test solutions
and referencesolution.
2. Usinga separatepipet for each reagent to be added,combinethe followingvolumesof
reagents to prepare the test solutions.Note:There are two differentiron “stock”solu
Readthe labelscarefullybeforeuse!
tions, 0.0020Mand 0.200M Fe(N0
3).
Reagents
•
Sample
0.0020 M Fe(N0
3)
0.0020 f4 KSCN
DistilledWater
Testsolution#1
5.00mL
1.00mL
4.00 mL
Testsolution#2
5.00mL
2.00 mL
3.00mL
Testsolution#3
5.00mL
3.00mL
2.00mL
Testsolution#4
5.00mL
4.00 mL
1.00mL
Testsolution#5
5.00mL
5.00mL
0.00mL
FlinnChemTopic”
Labs
—
Equilibrium
Page 5- The EquilibriumConstant
U57
3. Prepare the referencesolution #6 bymixing 9.00mL of 0.200M Fe(N0
and 1.00mL of
3)
0.0020M KSCNin beaker#6.Note: Usethe same pipets that were used in step 2.
4. Mixeach solution using a stirring rod. Rinsethe stirring rod and dry it betweensolutions.
5. Measurethe temperature of one of the solutionsand record it in the data tab’e.This will
be assumedto be the equilibriumtemperature for all of the solutions.
Part B. Colorimetry Measurements
6. Fillsix cuvettesabout ¾-fullwith the solutions from Part Aand arrange them in order on
a labeledsheet of paper to keep track of the solutions.Do not write on the cuvettes.
7. Handlethe cuvettesby their ribbedsides or their tops to avoidgetting fingerprintson the
surface.Wipethe cuvetteswith lint-freetissue paper or lens paper.
8. Connectthe interfacesystem to the computer or calculatorand plug the calorimeter sen
sor into the interface.
9. Select Setup and Sensors from the main screen and choose“Colorimeter.”
Note: Manynewer sensors have an automatic calibration feature that automatically
calibrates the colorimeter before use. If the sensor has the autocalibration feature, set
the wavelengthon the calorimeter to 470 nm (blue), press the autocalibration key,and
proceed to step 14. If the sensor does not have the autocalibration feature, followsteps
10—13to calibrate the calorimeter with a “blank”cuvette containing only distilled
water.
10. SelectCalibrateandPerformNow from the Experimentmenu on the main screen.
11. Fill a cuvetteabout¾-fullwith distilledwater.Wipethe cuvette with a lint-free tissue,
then placethe cuvettein the calorimeter compartment.
12. Set the wavelengthknob on the calorimeter to 0%T—theonscreen boxshould read zero.
Press Keep when the voltage is steady.
13. Turn the wavelengthknob on the calorimeter to 470 nm (blue)—theonscreen boxshould
read 100.PressKeep when the voltage is steady.
14. Return to the main screen and set up a livereadout and data table that will record
absorbance(as a function of time).
15. Select Setup followedbyData Collection.Clickon SelectedEvents to set the computer
for manual sampling.
16. Removethe “blank”cuvette from the calorimeter compartment and replace it with the
cuvette containingtest solution #1.
17. Press Collecton the main screen to begin absorbancemeasurements.
TheEquilibriumConstant
record
The Equilibrium Constant Page 6
—
,.
c35
matically
18.Whentheabsorbancé
readingstabilizes,pressKeep on themainscreento aut
Note: Theabsorbance
theabsorbance
measurement.
measurement
ild
sho
appear
tablewill
alsocontaina time reading,whichmay
Theonscreen
in a datatableonscreen.
beignored.
andreplaceit with thecuvette
19.Removethecuvettefromthecolorimetercompartment
containingtestsolution#2.
20. Whentheabsorbance
readingstabilizes,pressKeep on themainscreento automatically
recordtheabsorbance
measurement.
andwith the referen
e
2.1.Repeatsteps19and20with theothertestsojutions#3—5
solution#6.
22. PressStopon th mainscreento e
d
printout of thedatatable.
thedatacollection,
If possible,obtaina
process.
in theDataTable.
23. Recordtheabsorbance
datafor solutions#1—6
24. Disposeof thecontentsof thecuvettesand,ofthe remainingtestsolutionsasdirectedby
your instructor.Followyourinstructor’sdirectionsfor rinsinganddryingthecuvettes.
Flinn Chemlopic” Labs
—
Equilibrium
Page7- The EquilibriumConstant
3.
L35
Name:
Class/Lab Period:
The EquihbriumConstant
Data Table
Temperature
Sample
341*
IFe
Absorbance
ESCN1*
0
,
Testsolution#1
Testsolution#2
Testsolution#3
..
Testsolution #4
Testsolution #5
•1
Referencesolution #6
.
*These are the concentrations of ions in solution immediatel
after mixing and before any reaction has
’
occurred. See the Pre-LabQuestionsfor calculations.
Post-Lab Calculations and Analysis
(Usea separate sheet of paper to answer the followingquestions.)
1. Asdiscussedin the Background section and the Pre
Lab
Questions, it is assumedthat
essentiallyall of the thiocyanate ions present in the reference solution will be converted
2
to product. What is the concentration of FeSCN
ions in the reference solution?
For Questions 2—7,construct a Results Tableto summarize the results of the calculations.
2. For each test solution, the absorbance (An,where n = 1—5)should be directly propor
2
2
tional to the equilibrium concentration of FeSCN
ions. The concentration of FeSCN
ions can be calculated by comparing its absorbance versus that of the reference solution
2
(Aref).Usethe followingequation to calculatethe equilibrium concentration of FeSCN
ions in each test solution#1—5.Enter the results in the Results Table.
2]
IFeSCN
21ref
(An/Aei) x (FeSCN
=
3 ions in each test solution #1—5:subtract
3. Calculatethe equilibrium concentration of Fe
3 ions
2
the equilibriumconcentration of FeSCN
ionsfromthe initial concentration of Fe
(see the DataTable).Enter the rsults in the Results Table.
+l
3
IFe
TheEquilibriumConstant
eq, n
—
—
3’
Fe
‘initial
‘
—
2
FeSCN
The EquilibriumConstant Page8
-
(.354,
4. Calculatethe equilibrium concentration o?SCN ions iii each test solution #1—5:subtract
2
the equilibriumconcentration of FeSCN
ions from the initial concentratiod of SCN
ions (seethe Data Table).Enter the results in the Results ‘lihlc’.
Jeq
lSCN
•
n
=
Tiinitiai
‘lS
-
2’
[FeSCN
i1
5. UseEquation 4 in the Background section to calculatethe valueof the equilibrium con
stant Keq for each test solution#1—5.Enter the results in the Results Table.
6. Calculatethe mean (averagevalue) of the equilibriumconstant for the fivetest solutions.
•
7. Calculatethe averagedeviation for Keq:Find the absoiutevalueof the differencebetween
each individualvalueof the equilibriumconstant and the mean. The averageof these dif
ferencesfor solutions#1—5is equal tà the averagedeviation.
8. The averagedeviationdescribesthe precision of the results. Doesthe precision indicate
that the equilibriumconstant is indeed a “constant”for this reaction? Explain.
•
9. ribe
Desc the possiblesources of error in this experimentand their likelyeffecton the
results.
•
Fllnn Chemlopic° Labs
—
Equilibrium
(353,
iftia. & rz.re.
Materials
60n112% Polyvinyl alcohol
•5m1,IOml, or .15m1012% Sodium Tetraborate (see your teacher)
Craftstick/spoon
Large StyrOfoam cup or 250m1glass beaker
Plastic pipette
Food coloring (various colors)
Powder-free/non-latex gloves
Paper towels,
Sthall plastic bags (ZI
,loc)
lOOm!graduated cylinder
lOml graduated cylinder
Procedure
1. Measure 6Oml ,f polyvinyl alcohol into the large Styrofoam cup or 250mi beaker. Take a
look at it and give it a stir or two. Notice the iocation of any bubbles that have formed.
Add 2 drops of food coloring to the polyvinyl alcohol and mix. This will add color to your
slime.
••
2. Measure about 5m1,lOmi,or 15mJ of sodium tetraborate solution (see your teacher for the
in a lOmi graduated cylinder’and pour it into he cup containing the
)
specific amount
polyvinyl alcohol. Check out the location of the bubbles. Stir quickly with the craft stick
to produce the slime.
3. Examine the properties of the cross-linked polymer (i.e., play with the stuff). Scoop it out
of the cup and let it flow between your fingers. Is it a solid or a liquid? Is it elastic? Does
it bounce? Compare the viscosity of your slime with the two solutions used to create the
slime the polyvinyl alcohol and sodium tetraborate. Write down your observation
relating to the physical properties of your slime on the back of this page. How does this
lab relate to the concept of chemical bonding?
—
4. When you’re all done, you can save the slime for a few days by putting it in a plastic bag.
What’s happening
When you mix polyvinyl alcohol with a bit of water, you make a substance that is
known as a polymer and that the sodium tei.raborate solution is a cross-linking’ substance that
binds the polymer chains together to make the solution thicker. So, as the polymer chains get
more ‘bound-together, it gets harder for them to move around, and your slime starts to be
.
more like Silly-puny
-S6.
(3
Name
#i
Lab
-
Measuring Liquid Volume
With a Graduated Cylinder
Purpose
I,
Materials
Red, blue and yellow food coloring
Water
3 100-mIbeakers
6 paper cups
50-mI graduated cylinder
66
Marker
I
Procedure
1. Label each paper cup with the markerA, B, C, D, E, andF.
2. FIll threebeakers with 50 ml of water. Add a few drops of food colonng to each
beaker so that you have one beakerof red water, one of blue water, and oneofyeHow
water.
3. Into paper cup A measure 19-mi of red water.
4. into paper cup C measure 18-mi of yellow water.
5. Into paper cup E measure 18-mi of blue water.
6. From paper cup C measure 4-mi and pour the 4-mi into paper cup D.
7. From paper cup E measure 7-mi and add it to papercup D. Mix gently.
8. Fromthe beaker of blue waler measure 4.-miand pour it into paper cup F. Then from
the beakerof red watermeasure 7-mi and add it to paper cup F. Mix gently.
9. From paper cup A measure 8-mi of and pour it into paper cup B. From papercup C
measure 3-mi and add it to paper cup B. Mix gently.
10. Complete datatable that follows.
II. Clean up.
II
L35T
andConclusions
Qbservations
I
Complete the data table below by listingthe final colors in each paper cup. Giv the total
amount of water in each paper cup in millilitersml).
• Paper Cup
Color of Water
Total Amount of Water
(ml)
,
:
A
,
B
C
••
D
$
,,
F
Checkyourchart with your teacher.
Go backandfill inthe purpose of this lab.
What weresome sources of error?
I.,
2.
,
Name
—
Date
I
I
F’—-’ F—in this activity You will compare the rates of two different reactions.
Materials (per group)
2 test tubes
I arttadd tablet
water
vinegar
test tube rack
Procedu.re
1. Put on your goggles and lab apron.
2. Fill one test tube half full with water and the other test tube half full with vinegar.
3. Break the antacid tablet into two equal parts.
4. oDp one piece of the antacid tablet into the test tube with water and the other into the test
tube with vinegar.
5. Compare the reaction in the two tubes.
I
Questions
1. What differences did you observe between the reactions rn the two test tubes?
2. Does the chemical reaction occur more rapidly in one of the test tubes than in the other? How
do you know?
3. Why do you think there is a difference between the rates of the chemical reactions that occur
in each of the two solutions?
9.
aw
N
N
rro,se
-
e
ke
co
1i
Q
+\- s ceac4
c,
‘
--
7•
5
(3
Name:
Bäypon-BluePOintHS: Che
iistry
Date:
______________________________________
bapt
--
___________________
ers
SIiIf
Pre-LabDiscussion:
andtheShape
Shifters
inthisactivityaremadeof
Commercial
Shrinky-Dinks®
Depending
polystyrene,
acommonpolymer.
uponhowtheyaremanufactured,
have
the abilityto shrinkwhenheated.
andcertain
other
polymers
can
polystyrene
Whentheshrinkable
isheatedat lowtemperatures,
it doesnot
polystyrene
themolecules
form
to
return
to theiroriginal
newproducts;
merely
decompose
Thismeansthatnochangein massis expected..
(notstretched)
configurations.
theirresponse
Oneclassification
scheme
of polymers
involves
to heating.
soften
unaltered
in thisheating
Thermoplastic
polymers
onheating,are
chemically
and canbeheatedand meltedanynumberoftimes.Thermosetting
process,
andcannotb
polymers
aresetintothe desiredformduringpolymerization
themto degrade
extensive
reformed;
heatingcancause
ordecompose.
Polystyrene
isathermoplastic
inthatit softensduringheating.Butwhatcauses
the shrinking?
-
issomewhat
unusual.
Theshrinking
Mostcommonsolids,
abilityof polystyrene
whenheated,
eitherexpandbeforetheymelt intoliquids(forexample,
metals)
or
andothershrinkable
woodintocharcOal).
(forexample,
Polystyrene
decompose
exhibittheirshrinkingnaturedueto thewaytheyaremanufactured.
As
plastics
these
stretched
heated,
outintoafilm,thenquickly
theyareproduced, plasticsare
tl
themolecules
ofthe polymerin their
cooled.
Thesuddencoolingufreezes
thisprocess,
stretched-out
Tovisualize
configuration.
imaginehowa personmight
askedto freezewhilein themiddleof doingjumpingjacks.
appearif suddenly
withinthemarereleased
Whenthe plastics
areheatedonceagain,themolecules
fromtheir“frozen”configurations;
theyreturnto theiroriginaldimensions,
i
ntheobserved
shrinkage.
resulting
1. Rinse and dry one clear piece of polystyrene. This is labeled with a recycle
“Code 6” on the bottom.
a. It must be completely dust free!
2. Using the permanent markers, draw a design on your piece of plastic.
3. Weigh the plastic (after coloring).
4. Trace the piece of plastic in the observation section.
5. Place the plastic on the tray covered with aluminum foil and put into the
toaster oven.
6. Heat at 325°.
7. The piece is ready after the edges have curled up
8. Carefully remove from oven and allow to tool.
9. Weigh the piece after shrinking and record the mass.
10. Trace the piece after shrinking.
.
Observations:
I
Weight before:
Weight after:
Difference:
FORLB
______________
_______________
include your design (in COLORED PENCIL-not marker)
AFTER- include your design (in COLORED PENCIL- not marker)
.
Post lab questions:
r; DiliñeheaL
2. Was this a physical or a chemical change? Explain!’
3. What effect did heating the plastic have on the motion of the particles of the
• plastic? Explain!
4. What happened o the color on the plastic? Did you notice any changes?
5. Describe the process for preparing polystyrene for commercial use. This is
found in the prelab discussion.
.
--.--
.
.
$1
II
Name:______________________________________
FLOATING CANDI
-4!S
Studentswill observea combustionreactionanddeducethe components
necessaryfor the reactionto occur.They will also see the relationshipbetween
pressure,volume, and numberof molecu!esfor gasses.
Substitutions
Materials
votive candle
2 small jars with diameter largerthan votive candle
2 400-mLbeakers
candletper
I aluminum pie pan
I large petndish
water (withoptionalfood coloring)
matches
beakerwith water
jar with watqr
,
.
•
Procedure
1. Place a votive candlein the centerof a petn dish or an aluminumpie pan
and light it.
2. Carefully, pour somewater into the dish or pan untilthe candleis
floating.
3. Quickly,place the invertedjar over the candleand allow it to rest on the
bottomof the pan. Reportthe results.
e votive candle again and repeatsteps 1-3. Makesure you dry
4. Light tt
the invertedjar eachtime you repeat.
5. LISt the sequenceof events and the reasonfor each. Repeatsteps 1-3,
as desired.
Data and Observations
Sequencethe eventsthat you observe.
2.
3.
4.
5.
(J63.
Disposal
All solidmaterialsmaybe placedinthetrashcan,andthe liquidsmaybe
pouredintothesink.
Questions
We will review your data & observations as a class BEFORE you
answer the questions.
Useyourknowledge
of the gaslawsandthe principlesof
combustion
to explaineachof theobservations
listedabove.
1.
2.
4.
5.
‘ame_
Date
How Many Drops :Ca y
PiIé.on a Penny?
H
4O
S
3
A
LICATIO
Tex: Refererice Chapt
’r
j
Introduction
Have you ever seen how water striderscan walk across the surtaceof
a pond. or how a mosquito can land on a puddle of water without
sinking? Although the strider’s weight pushes down on the water, forces
within the water create a “skin” that supports the strider on the waters
surface. This phenomenon is called surface tension.
The forces within water that are responsible for surface tension origi
nate at the molecular level. As ‘ou knqw, a water molecule is polar. The
oxygen atom has a partial negative charge. and each hydrogen atom has
a partial positive charge. As a result, electrical attractions occur between
the oxygen atom of one molecule and the hydrogen atom of another
molecule, as illustrated in Figure 40—1.These intermolecular attractive
forcesare called hydrogen bonds (H-bonds).
H
H
1•
:b
H
e
watermoiecu
Figure
...
O—H
0—H
H
H
rlyaroQenbonairigbetween
watermoiecuies
40—1
Surface tension can be affected by substances dissolved in water. If
substance interferes with hydrogen bonding, the surface tension of
water decreases. If a substance enhances hydrogen bonding. the surface
tension of the water increases, in this invesbga
on,
you will observe the
effects of several solutes in water. The ionic or molecular structures of
the solutes you will be using are illustrated in Figure 4.0-2 or. the next
page. You will use your knowledge of molecular geometry and the
behavior of molecular dipoles to make predictions about the effects of
these solutes on the surface tension of water.
In Part A. you will investigate the surface tension of water by seeing
how many drops of water can be piled on top of a penny. You will
observe the shape and behavior of the water as surface tension holds
the drops of water together. in Part B. you will design your own experi
merit to determine the effects of three substances on the surface tension
of water: liquid detergent. sodium chloride (NaCI), and sodiu.m carbon
3).
CO
ate (Na
2
)
renlice.P1aH.Inc
J
How Many Drops Can You Pile on a Penny?
217
4O
Name
T
(36
sooiurncnionoe(NaC1)
-
Sothurn
cartonate(Na,C
3’
-
Na
2Na
aetergentmolecule
Na
H
H ‘H’HHH
\/•
\/
H’’
N
/
/C
/N
H
\
H
/\
C
C
C
C
C
C
/,•\
‘/\
/\H/\
\
HH 1HH HH 3H
H HH
•
,
‘
:0:
C—c
1/
\/‘
/C\
H
/
nonpolarend
/
C—S—O:
:0:,
H
polar,chargecena
I,
Figure 40-2
b
Pre-L
ssion
Disci
Read.the entirelaboratoryinvestigationand the relevantpages of your
textbook.Thenanswerthe questionsthat follow.
1. Explain how the intermolecular forces between water molecules act
e
to create the “skir” or ‘surfa
tension upon which water striders
cart walk.
2. What causes water molecules to be polar?
3.’ Which part of a detergent molecule aUows it to dissolve in water?
Which part of a detergent molecule allows it to dissolve in cilv
substances?
________________________________________________
4. Predict whetherthe nature of deteigent molecules would increase or
decrease hydrogen bonding in water. Explain.
—
(T,
218
0 Pvennce-I-4aft.
Inc.
Date
C’ass
(366.
110W
$
f0:
fl
11I
fl
C
?
oll pvon penn
fi
The ii
te onof water allows it to bead up on many surfaces. In this
MithLab, you will compete to see who can deposit the most drops of water
arid the most drops of an aqueous detergent solution on a penny.
CHENLRB
RKC
MINILAS
SHEE1E
WDR
MINILA8
13.1
PROCEDURE
1. Lay a penny flat on your lab table.
2. Fill a microtip pipet with tap water, and
count the number of drops you can
deposit on the penny before water spills
over the edge. Record the number of
drops.
3. Fill another microtip pinet with a deter
gent solution prepared by your teacher,
and repeat the process. Record the num
ber of drops.
NDL
VSIS
ID
1. How is surface tension demonstrated in
this experiment?
2. Which has the lower surface tension: the
water or the aqueous detergent solution?
\\That accounts for this fact?
L
C
C
C
C
Chemistry:Concepts and Appkcations
MiniLab13.1
87
Bubble Making Lab
•
i&rstand
Purpàse: Toi
Lab#
H,
___
the role of a hydroscopic substancein a solution
Hypothesis:
Saftey:
Materials:
Procedure: In this lab.you will be working off of your desks Clean the desktops we1l
Next
..
you will obtain a beaker and add 50 ml of water to it. Now add a teaspoon of dish detergent (Joy,
Palmolive) to the beaker. After you mix the solution well, poor a small amount onto the desktop.
Then take a straw and try to, blowa bubble. If successful, measure the bubble ring once it pops.
Determine which detergent makes the largest bubble. Finally, all of the groups will be given an
assignment. Each group will stall with 100 ml of water in a beaker. Then they will each be
assigned a specific number of drops of glycerol The goal is to have each group measure the size
of this bubble. Each group will share their data on the blackboard and chart the data foi-mthis
portion ‘ofthe lab.
Data:
Drops of Glycerol
•5drops
—
10 drops
15’drops
2odrops
25 drops
30 drops
35 drops
40 drops
45 drops
50 drops
55 drops
60 drops
‘65drops
70 drops
Size of bubble
.,
‘
.
.
Observation(s):
Conclusion / Theory: Define4 Sw-facetension, Cohesiveness, Adbesiveness,
and hydroscopic substance.
Cover these points: 1) Why can’t we blow bubbles with plain water?
2] What is the role of the detergent?
3] How does cohesiveness come into play in the water molecule?
4] Surface tension relates to cohesiveness. What effect on surfacetension
should the substance added have on the water?
Lab: MakingMedievalMesses
The yearis 1450. You are the chemistfor a smalltownin Europe.Oneof the,
villagershas broughtyoutwoflasks; one containsan acid,andthe other
containsa base. The problemis thatthe labelshavefallenoffof the flasks,and
hedoesn’tknowwhichflask containswhatsolution.
As townchemist,yourjob is to determinewhichof theflaskscontainsacid
andwhichcontainsbase. Becauseof yourlimitedtechnology,you haveonlythe
followingmaterials:
Acidicsolution
Basicsolution
Extractof cabbage
Testtubes
Goggles(Theseyou mustuse,no matterhowyou solvethe problem!)
Good luck!
© 2000 CavalcadePublishing
For chemistryhelp, visit www.chemfiesfacom
Name______________________________
Date____________ Class
7—2Explore
Bending Water
In this activity you will explore the effect of a negatively charged comb on streams of different
liquids.
Materials (per pair)
plastic or nylon comb
35-mL syringe with plunger removed
200-mL plastic cup
distilled water
ethanol
mir)eral oil or vegetable oil
paper towel
-
Procedure
1. Put on your goggles and lab apron.
hold the syringe tube upright, plug up the small hole at the bottom with a
tr1er
2. Have one pa
finger, and fill the tube with distilled water.
3. The other partner should comb his or her hair vigorously to build up a negative charge on the
comb.
4. The first partner should uncover the hole in the syringe and let the water drain in a thin stream
into the cup. The second partner should hold the charged comb about 10 cm below the bottom
of the syringe and move it close to the flowing stream of water. Do not move the comb into
the stream of water. CAUTION: Clean up any spilled liquids immediately.
5. Note any changes in the flow as you bring the comb closer to the stream of water.
6. Clean and dry out the syringe with a paper towel. Repeat the activity with the alcohol and the
mineral oil. Write your observations in the chart below.
Liquid
Effectof chargedcombon
the liquid
distilledwater
ethylalcohol
mineraloil
.
Questions
1. Compare the effect of the charged comb on the three liquids.
2. Why do you think the negatively charged comb did not have the same effect on all three
liquids?
3. Predict how a stream of water would respond to an object with a positive charge.
S
Chapter7 11
4.2Heat Transfer
377
4.2.2 CONDUCTIONAND COMBUSTION
kindlingpoint,heat conductivity, combustion.
Concepts to Investigate: Conduction,
Materials: Insulated copper wire, candle, matches.
Safety: Wear goggles in this activity and whenever using an open flame.
.‘
Principles and Erocedüres: The ability to use fire for heating, light, cooking and metal
ons.
Modern civilizations con
\smelting was essential for the development of early civilizati
tinue tci rely upon fire to meet ever growing energy demands. Coal, natural gas and oil are
burned to produce energy necessary for transportation, industry and homes. Three criteria are
required to sustain fire: fuel, oxygen and sufficient temperature. A tire will be extinguished
if the fuel is consumed, the oxygen is depleted or the temperature drops below the kindling
point (the minimal temperature at which combustion may be sustained). Water is effective in
extinguishing many types of fires because it reduces the temperature of the fuel source below
the kindling point. (Water has a high specific heat and a high heat of vaporization, and there
fore.absorbs a great amount of energy as it is heated and boiled.) Is it possible to extinguish
o prevent fire using metal rather than water?
Strip the insulation from a 30-cm section of a 40-cm length of copper wire. Wind the
lower the coil’over
‘bare wire into a coil as shown in Figure C. To extinguish the cndleflame,
a lit candle. Re-light the candle, and lower,the coil over the flame slowly so that the coil is
heated by the flame as it is lowered. Is it possible to lower the coil slowly enough that the
flame is not extinguished? Allow
the coil to cool, and re-light the
candle. Heat the coil in the flame
of a laboratory burner and then
lower it over the candle flame
(Figure D). Is the flame extin
guished as before? Why or why
not?
D
C
Table 1: Average Thermal and Electrical Conductivities at Room Temperature
copper
aluminum
zinc
brass
iron
nickel
steel
Thermal Conductivity
2
s’cm
cal
Electrical Conductivity
ohm’cm’
0.92
0.50
0.27
0.26
0.16
0.14
0.12
0.59
0.38
0.17
0.13
0.10
0.15
0.03
rs’
m
-a
L371.
378
Unit Four: Thermodynamic and Kinetics
Questions
i
of copper wire is lowered Over it?
(I) Why is the flame extinguished when a cpol cdi
flame
in
such
the
coil
the
over
(2) Is it possible to lower
a manner that the candle remains
lit? Explain.
(3) Would it be easier to extinguish the candle if you used iron wire rather than copper
wire? Explain (see Table 1).
I,
L
(3
364
Unit Four: Thermodynamicsand Kinetics
4.1.8 ENTROPYANDENTHALPY
Concepts to in vestigate: Entropy, entha]py, free energy, Gibbs free energy equation, sponta
neous processes.
Materials: Wide rubber bands, goggles, ring stand, paper clips, weights, hair dryer.
Safer)’: Wear goggles.
Principles and Procedures:
Part 1: Put on goggles! Hold a strong, wide rubberband tightly across your forehead. Quickly
stretch the rubber band while keeping it pressed to your skin. Does the temperature of the rub
ber band increase or decrease? After approximately.20seconds, relax the rubber band. Does
the temperature rise or fall when the rubber band is relaxed?
The stretching of the band is not a spontaneousprocess because it will no occt
with
r
from
We
band
is
the
outside.
know
that
the
exothermic
out energy
stretching
J1)
jez
(negati’
since heat is released and felt by thermoreceptors in the skin. An analysis of the Gibbs free
energy equation indicates that if z’iGis positive, and H is negative, then the change in enS) must be negative if the equation is
tropy (z
to be balanced:
-C.
\G=ziH-ThS
Thus, the arrangement of molecules in a
stretched rubber band must be less random
(lower entropy) than when relaxed.
Part 2: Place a wide rubber band over a
lateral post on a ringstand. Using a paper clip,
hang weights (fishing weights, washers, bal
ance weights, etc.) from the rubber band un
til it is fully stretched as shown in Figure N.
Measure the length of the rubber band, and
then heat it with a hair drier. Does the rubber
band shorten or lengthen? Is this what you
expected? Record the percentage change in
length.
Questions
(I) Is the relaxation of the rubber band a spontaneous process? Explain. Is the iG value
of the rubber band relaxation process positive or negative? Explain.
Is
(2)
stretching the band exocherrnic or endothermic? Is the contraction of the band
exoihermic or endothermic? Explain.
Did
the rubber band shrink or expand when heated?
(3)
(4) Is the arrangement of molecules more orderly when stretched or relaxed? Is the en
tropy of the band greater when stretched or relaxed? Explain.
(5) What is the driving force that causes a stretched band to contract (relax)?
(6) Why does healing the stretched rubber band cause it to contract?
c.-
Li
‘O here’s the mission,youare goingto balanceequationsor Ms
K
wayyou wouldlike to lookal it.
Step One:
whichever
‘.,
••
Assigneachelementin your problema different color MaM.
Step Two:
Placethe numberof M&M’s for eachelementonyour paper.
StepThree:
Choosea coloror an elementandsee if there is anevenamounton either.
side.of the..orrow.If NOT..,go tb step four...’
Step Four:
Match up the nuthberof M&M s on either side by changingthe coefficient
or numberin front of the compoundor element. Try to usethe smallest
coefficient possible.
Step 5: List the elementsunderthe equationandthe numberof them on either
side of the arrow.’
PRACTICE:Fill in the blanks.
1.
CO
,
•
___
C):
9
2
2CC)
:.-
2.
3.
•
__
.
3
___NH
•
__
0:
02
9
.
3
___HNO
’
0
2
__H
0
2
___H
—
-
—
For each Equation set up your M&M’sto determine the nurnber’of elements on
either side of the equation.
*TitIe your paperand put your names
on it..
..
AndAwaywe 6©©©©©©©©©©©.
1. __N
2.
CuO +
3. __NH
3
4.
4
AIPO
+
3
__NH
__
4
__Cu:
+
02
+
__NO
+
2
_H
+
+
+
4
2
__Ca(OH)
__
O
H
3
AI(OH)
+“,
-
Good Luck they do get harder as you 90 along don’t be afraid to coh
pare
numbers right away for either side.
(’P’
3
.Co
—
the
*5ometimes it is easier to odd up either side and find commondenominators
between them.
ton’t
forget, some time.s there is more than 1 occuronce for each element on
the same side of the arrow.
0
KEEPPO5IiIVE... AN!) KEEPTRYING YOU WILL SUCCEEb.
As Albert Einsteinsaid,
Imagination is MORE important than knowled9e.”
.
Using the Scientific Method
Introduction
You will be given four common substances which when
mixed
wi-ilproduce a number of cheth±calreactions. The two solid
are sodium.bicarbonate ( baking soda) and calcium chlor
.de
ice melting compound ) while the two liquids are water and•
phenol red ( swimming pool indicator
The object of this experiment is to mix the four chemicals
and to carefully observe the results. Next you will be
expected to set up a series of experiments to determine which
substances are responsible for each of the observed changes.
Safety
1.
Wear protective goggles throughout the experiment.
2.
Leave a small opening in the ziplock bag when the four
chemicals are mixed
3.
The ziplock bag can be placed in the waste basket a
d
the microscale reaction plates can be rinsed in the sink.
Procedure
Part One
Observations
1.
Describe the properties of each substance which you,will
be using during the experiment.
2.
Mix two tablespoons of calcium chloride and one
tablespoon of sodium bicarbonate in a freezer type
ziplock bag. Shake and list your observations.
3.
Mix 10 ml of water with 10 ml of phenol red and report
your observations.
4.
Now pour the liquid mixture into the ziplock bag
containing the two solids. Squeeze out as much air as
possible from the ziplock bag and seal it. Make complete
and detailed observations.
Part Two
Student Experimentation
1.
Which combinations caused the observed changes?
2.
Using a microscale reaction plate, a microthermometer and
small amounts of the four chemical, conduct a series of
experiments to determine which substances are responsib’e
for each of the observed changes.
Data Analysis
1.
Prepare a chart indicating the experiments performed and
their results
2.
From your data, indicate which combinations of chemical
are required to produce each of the observed changes.
$
‘.15Clues found from Colby Nyc’s Murder
1. A janitor found Mr. Colby Nye’s lifeless body behind his desk at 8:00pm.
2. Jack.Mackenzie is Colby’s business partner and Robin’s older brother.
3. Colby has been married to Robin for 9 years.
4. Jack Mackenzie was at his country estate at the time of the murder.
5. Colby left his office at 6:20pm to mail Jack a self-addrçssed envelope with some
contracts for him to sign. Colby returned to his,office at 630pm.
6. Colby told Robin the day before the murder that he wanted a divorce because he was
in love with Lynda.
7. Lynda & Colby were having an affair for 3 months.
8. Lynda is Jack Mackenzie’s‘wife& a close friend of Robin.
9. Jack was forced into th partnership with Colby after Jack’s father died & left Colby
half of the busines in order to provide for Robin.
10. Robin showed up at Colby’s office at 7:00pm. They had a few drinks & argued.
11. Lynda showed up at Colby’s office at,7:15pm & saw Robin running out of the office.
1’2.Lynda told Colby she would never forgive him for tdlin Robin they were having an
affair & wished him dead.
13. Lynda left the office at 7:20pm.
14. Colby Nye was killed by excessive amounts of potassium cyanide, which was ingested
& found in his stQmach.
15. No traces of potassium cyanide were found in either food or liquid tested at the scene
of the crime.
Based on these clues try & answer these questions.
1. Who killed Colby Nye?
2. How was the poisoned administered?
3. What was the killer’s motive?
L3T
The murderofColbyNye
1. Who killed ColbyNye?
JackMackenzie
2. How was the poisonedadministered?
Thecyanide was placed on the self-addressed envelope provided by Jack
Mackenzie.
3. What was the killer’s motive?
To have total control of the business.
Explanation
Jack Mackenzie hated ColbyNye fromthe minute he laid eyes on him; and that was before
he was forcedinto partnershipth
him. So after workingwith the 1 man he despisedmoreth
n’
anyoneelse, his levelof hatred roseto an all newhigh. Jack new nothingabout his wife’s affair
with Colby;all he knewwas that he needed to get Colbyout of thç business& his family’s life.
Insteadof trying to buy him out & waste evenmore moneyon his lazy brother-in-law,Jack would
simplymurder Colby& all his problemswouldbe solved.
If his partner was to meet with an untimely death, Jackhad to make sure that he was
nowherethe officeat the timeof the murder. Jack wouldgo away to his countryestate the same
weekthat their secretarywouldbe on vacation. WithJack & their secretaryaway fromthe office
that wouldleaveColbyall aloneto handle everydetail.
He wouldhave no otherchoicethan to handle all the annoying office choreshimself such as
sealing & mailing envelopesas well as answeringthe phones.
Before Jack left town, he placeda contract & a self addressed stamped envelopeon
Colby’sdesk. He thenleft a note with Colby, insisting that he sign the contract & mail it back to
Jack at his country estate. Unfortunatelyfor Colby,the envelope’sadhesivewas laced with
potassiumcyanide(KCN). As soonas he lickedthe envelope, it wouldonly be a matter of time
beforethe poisontook effect. Colby mailedout the letters& went back to his office& eventually
died. A few dayslater,whilethe murder investigationwas wellunder way, Jack receivedthe letter
in the mail & destroyedthe envelope. The murder weaponwas neverdiscovered,& the murderer
of ColbyNye was neverbroughtto justice.