NMR Spectroscopy Intro Organic
advertisement

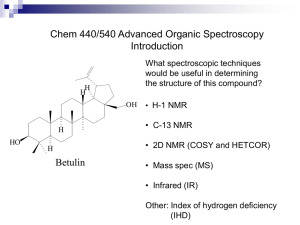
Index of Hydrogen Deficiency (IHD) & Intro To NMR Spectroscopy Index of Hydrogen Deficiency IHD The sum of the number of rings and pi bonds in a molecule • To determine IHD: • compare the number of hydrogens in an “unknown” compound with the number in a reference hydrocarbon of the same number of carbons with no rings or pi bonds (the corresponding alkane) • The molecular formula of the reference hydrocarbon is • CnH2n+2 (for a straight chain alkane) What about odd-ball atoms? IHD Correction Table From Fully Saturated Straight Chain Hydrocarbon Group V Group VI Group VII -1 No Change +1 N O F P S Cl Br I (H reference IDH = - H unknown ) 2 H unkown = molecule you may not know the structure of, or it is not an aliphatic alkane Add correction term to this value IHD ALKANE Alkanes Alkenes Alkynes CnH2n+2 CnH2n CnH2n-2 IHD = 0 IHD = 1 IHD = 2 cyclic / ring alkane IHD = Acyclic / open chain CH3 H3C Alkane H 14 Compound - 12 ______ C6H14 C6H12 CnH2n+2 CnH2n IHD = 0 CH3 H3C Alkane H 14 Compound - 10 ______ Alkane H 14 Compound - 12 ______ C6H12 2H IHD = IHD = CnH2n C6H10 2H 2/2 = IHD of (1) 4H CnH2n-2 4/2 = IHD of (2) 2/2 = IHD of (1) Lacks 1 mol of Hydrogen Lacks 1 mol of Hydrogen Lacks 2 mol of Hydrogen IHD = O CH3 H3C C6H12O IHD = Alkane H 14 Compound - 12 ______ 2H CnH2n 2/2 = IHD of (1) lacks 1 mol of Hydrogen O O - C6H8O _________ 4H C6H10O CnH2n-2 C6H14 Alkane H 14 Compound - 10 ______ 6H 4/2 = IHD of (2) Lacks 2 mol Hydrogen 1 IHD ring 1 IHD carbonyl 6/2 = IHD of 3 1 IHD ring 1 IHD alkene 1 IHD carbonyl Index of Hydrogen Deficiency Problem: Our “unknown” is isopentyl acetate. It has a molecular formula of C7H14O2. Calculate its IHD Solve: Reference hydrocarbon C7H16 (CnH2n+2) O O Isopentyl acetate IHD = (16-14)/2 = 1 Problem: calculate the IHD for “unknown” niacin, molecular formula C6H6N2O Solve: Reference hydrocarbon C6H14 (CnH2n+2) IHD = (14 – (6-2))/2 (14 – 4)/2 = 10/2 = 5 O NH2 N Niacin Structure Determination NMR All hail … NMR !!! Rf • NMR is the most powerful tool available for organic structure determination It is used to study a wide variety of nuclei: 1H, 13C, 15N, ,19F, 31P Sensitive for only one particular isotope of one particular element Nuclei with an odd mass or atomic number These Nuclei have a spin quantum number of (1/2) Nuclear Spin • A nucleus with an odd atomic number or an odd mass number has a nuclear spin. ( +1/2 ↑) or ( -1/2 ↓) • The spinning charged nucleus generates a magnetic field. External Magnetic Field When placed in an external field, spinning protons act like bar magnets. Magnetic Resonance A spinning gyroscope in a gravitational field Magnetic Shielding • If all protons absorbed the same amount of energy in a given magnetic field, not much information could be obtained. How useless is this!? • But protons are surrounded by electrons that shield them from the external field. CH3CH2OH 5 4 3 PPM 2 1 0 Much Better!!! • Circulating electrons create an induced magnetic field that opposes the external magnetic field. Shielding of Protons NMR Spectrometers 750 MHz 400 MHz CH3 H3C Si CH3 CH3 Tetramethylsilane • TMS is added to the sample. • Since silicon is less electronegative than carbon, TMS protons are highly shielded. Signal defined as zero. • Organic protons absorb downfield (to the left) of the TMS signal. Three types of Hydrogens ~ we see 3 different signals Sample Preparation 2 H Deuterium = NMR Invisible Solvent O D3C CD3 D2O D3C C N Deuterated Water d6-Acetone d3-Acetonitrile CD2Cl2 D D D D D Deuterated Methylene Chloride D3C D O S CD3 CDCl3 d6-Benzene Deuterated Chloroform d6-DMSO And Many More ...... 5 mg Compound 1.5ml solvent CH3 H3C Si CH3 CH3 TMS - Tetramethylsilane Very Common Solvent and Reference Standard Inside the NMR Spectrometer Liquid Helium (Bp = 4K or Liquid Nitrogen (Bp = 77K -269ºC) or -196ºC) Location Is Everything ↑ Increasing B field Protons require Less Magnetic Protons require Higher Magnetic Field to resonate / flip Field to resonate / flip NMR Signals • The number of signals shows how many different kinds of protons are present. • The location of the signals shows how shielded or deshielded the proton is. • The intensity of the signal shows the number of protons of that type. • Signal splitting shows the number of protons on adjacent atoms. Location of Signals • More electronegative atoms deshield more and give larger shift values. • Effect decreases with distance. • Additional electronegative atoms cause increase in chemical shift. Chemical Shift Values Chemical Shift Read From Left to Right NMR Spectrum • 1H-NMR spectrum of methyl acetate • Downfield: the shift of an NMR signal to the left on the chart paper • Upfield: the shift of an NMR signal to the right on the chart paper Two signals = Two different kinds of protons Number of Signals • Equivalent hydrogens: have the same chemical environment • a molecule with 1 set of equivalent hydrogens gives 1 NMR signal O CH3 CCH3 ClCH 2 CH2 Cl C H3 C Propanone (Acetone) Number of Signals CH3 H3 C 1,2-Dichloroethane Cyclopentane C CH3 2,3-Dimethyl2-butene Number of Signals Different Hydrogens: Different Signals • a molecule with 2 or more sets of equivalent hydrogens gives a different NMR signal for each set Cl CH3 CHCl 1,1-Dichloroethane (2 signals) Number of Signals Cl O Cyclopentanone (2 signals) CH3 C C H H (Z)-1-Chloropropene (3 signals) Cyclohexene (3 signals) Number of Signals – Simple Cases Splitting of NMR signals The N + 1 Rule If a signal is split by N equivalent protons, it is split into N + 1 peaks. 1 Neighboring Proton (N+1) Peaks (1+1) = 2 doublet 2 Neighboring Protons (N+1) Peaks (2+1) = 3 triplet Splitting of NMR signals Darn Neighbors! RH N + 1 = Split Signal No (s) H Neighbors R H = Singlet R R R N + 1 = 1, (singlet) N=0 R No Neighbors One (d) H Neighbor H R R R N + 1 = 2, (doublet) N=1 R One Neighbor Two (t) H Neighbors H = Doublet H H R R R H = Triplet N+1=3 N=2 Two Neighbors Splitting of NMR signals Peak Splitting N+1=1 Singlet (s) N = 0 Neighbors 1 peak Doublet (d) N+1=2 N = 1 Neighbor 2 peaks Triplet (t) 3 peaks N+1=3 N = 2 Neighbos Quartet (q) 4 peaks N+1=4 N = 3 Neighbors Quintet / Pentet (p) 5 peaks Hextet (h) 4 Neighbors 6 peaks 5 Neighbors Septet (sept) 7 peaks 6 Neighbors Octet (oct) 8 peaks 7 Neighbors (m) Multiplet Splitting of NMR signals Coupling Constants J1 J3 R C C J2 J J N+1=3 N = 2 Neighbors Triplet (t) 3 peaks R J3 J2 C J4 J3 R C R RH Protons are coupled to Neighboring Protons that are no more than 3 Bonds away "J3 coupling" Typical J values are 7 Hz R Hc Hb Ha Hx Ha is coupled to Hx Ha is also coupled to Hb Ha is NOT coupled to Hc R The doublet-septet pattern Isopropyl Group Nuclear Magnetic Resonance Spectroscopy 1H NMR—Number of Signals • In comparing two H atoms on a ring or double bond, two protons are equivalent only if they are cis (or trans) to the same groups. 33 Integrations: Relative Numbers of Protons O H3C CH3 O 3 CH3 CH3 1 Methyl Propanoate O H3C CH3 O Molecular Formula IHD = 1 3 - signals O H3C a O 4 RECAP Location c O CH3 b Number of Types of Hydrogens H3C 3.67 3 O O 3H 1.14 CH3 H3C 2.29 2 PPM O 3H CH3 2H 1 Splitting O H3C s O t CH3 q 0 CH3 O FT-IR = DANCE (bond vibrations/bends/stretches) O GC-MS = DESTRUCTION (bond cleavage) CH3 NMR = SPIN FLIP C10H12O2 8 7 180 160 6 140 5 4 PPM 120 100 PPM 3 80 2 60 1 40 20 0 0