US15, CEM255, Week 1,
advertisement

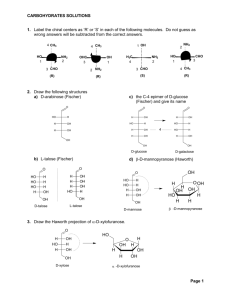
CEM 255: Organic Laboratory Spring 2016 Lecture: Thursdays 4:10 - 5:00 pm Room N130 BCC Instructors: Dr. Ardeshir Azadnia Office: 128 Chemistry Email: azadnia@chemistry.msu.edu Office hours: Mondays and Thursdays 10 - 11:30 am or by appointment Mr. Dave Voss Office: 134 Chemistry Email: voss@chemistry.msu.edu Office hours: by appointment Class Website: http:// www2.chemistry.msu.edu/courses/cem255/ Required Materials Lab Text: “CEM 255 Organic Chemistry Laboratory Manual – 7th Edition", A. Azadnia, Hayden- McNeil Publishing, Inc., 2013 Safety Supplies: Approved splash proof safety goggles (ANSI 279.1 1979) and rubber "dish washing" gloves. A rubber or plastic apron is also encouraged. Supplementary Readings: A 251-252 text, Any Organic Chemistry Text Miscellaneous: Any bound notebook with duplicate pre-numbered pages and carbon paper (comes as a bundle with the lab manual). The perforated carbon copies will be torn out and submitted at the end of each lab. Also, a ballpoint pen (not pencil) is needed for lab reports. Course Description: Material covered in lecture courses is based on experimental data accumulated over the years, which has been systematized into natural laws and theories. However, the study of chemistry (or any science) requires that you learn some laboratory techniques and methods of data acquisition. It is intended that the skills acquired in this course will be of practical value to you in your fields of interest. It is hoped that a "hands on" experience also will enable you to grasp the concepts presented in CEM 252 lecture more readily. Office Hours: Mondays & Thursdays, 10-11:30 AM or by appointment E-mail: azadnia@chemistry.msu.edu Mr. Dave Voss, room 134 Chemistry, 355-9715, X 398 Office Hours: by appointment, voss@chemistry.msu.edu Lecture Time: Thursdays 4:00–4:50 PM, Room N130 Business College Complex Grading Experiments will be graded according to the weight scale on the schedule below. Major or continual violations of safety rules will result in dismissal from lab without any points and/or reduction of grade by 50 points per occurrence. The grades will be based on accuracy and completeness of recorded observations and data as well as legibility (neatness counts). The grading procedure for each experiment will be discussed in class during the Thursday lectures (4:10-5 PM) prior to the corresponding lab. Your final grade will be based on the total points earned as follow: 965-1060 = 4.0 890-964 = 3.5 816-889 = 3.0 742-815 = 2.5 636-741 = 2.0 All CEM 255 students must get trained on the 300 MHz NMR instrument by no later than Friday, February 20, 2015. There will be a 45–point penalty for anyone who does not get trained/checked out by the due date. The sign-up sheets will be outside of room 125 Chemistry on Thursday, January 22, 2015. Students with “pacemakers” MUST not enter room 125 due to the health hazards such as DEATH, and therefore, would be excused from doing the NMR-training/check out. See me in person immediately if you have a pacemaker. Absences Because our teaching staff is being used to capacity, there will be NO MAKE-UP LABORATORY, except for special circumstances, which will be decided on an individual basis. If you are unavoidably absent from a laboratory class due to illness or family crisis, contact Dr. Azadnia as soon as possible. A grade of zero will be assigned for an experiment missed regardless of the reason. However, you may be allowed to make up the missed lab so long as you make arrangement with Dr. Azadnia within 3 days of the corresponding experiment. Students who miss three or more sessions due to illness, etc. should drop CEM 255 and re-enroll another term. Incomplete grades CANNOT be issued when three or more labs have been missed. If you drop CEM255 course, you still must be checked out of lab by your lab TA, on or before your last scheduled lab section. Failure to check out properly costs $25.00 plus breakage charges. No CEM 255 lockers will be shared. If you believe someone has access to your locker, exchange your lock at the stockroom (room 133). Write-ups Your Lab Manual is not to be used in the lab. You must work from your own procedures written in your lab notebook. An important laboratory skill is the making and writing down of precise observations. A well-kept notebook is an essential part of any investigation. Your notebook should be a complete description of what happened, or didn't happen, in your experiments. If someone else could repeat your experiment and get identical results using only the notebook for directions, then your notes are thorough. A well-kept set of records is an essential part of any investigation. Take notes as if you expected to repeat the work next term using only your own notes. Before entering the laboratory, you should familiarize yourself with the experiment by studying the Lab Manual and the corresponding chapter in your organic textbook. Experience has shown that those students who rewrite the procedure as numbered, single operation steps directly in their notebooks encounter fewer problems. If this is done in advance, then only observations and changes must be recorded during the experiment. Keep track of your progress by checking off the steps as you do them. Carbon copies of all prelab and in-lab write-ups are to be submitted, along with any products, as you leave the laboratory. Laboratory Safety Regulations SAFETY REGULATIONS 1. DRESS CODE Firm footwear is required (NO sandals, NO open-toed shoes) Long pants that cover the entire leg must be worn at all times (NO shorts, NO skirts) Safety Goggles must ALWAYS be worn in the lab The dress code will be strictly enforced! 2. SAFETY EQUIPMENT Every student MUST do all they can to prevent accidents in their own work and they must be prepared for accidents by knowing in advance what emergency aids are available. Laboratory Safety Regulations 2. SAFETY EQUIPMENT Every student MUST do all they can to prevent accidents in their own work and they must be prepared for accidents by knowing in advance what emergency aids are available. The laboratory is equipped with several type of safety equipment and it is essential that you become familiar with the location and the operation of these tools. a. Emergency Shower: There is an emergency shower located on the hallway. It is for use when corrosive liquids have spilled over large areas of clothes and skin, or when clothing is on fire. b. Eye Wash Stations: The laboratory is equipped with eye wash stations. These stations dispense water and provide thorough irrigation of the eyes and face in the event a person is splashed with an irritating chemical. The contaminated body part should be rinsed for a minimum of 15 minutes. c. Fire Extinguisher: The fire extinguisher is located outside the laboratory. Know its location and how to operate it. It is very effective for fires involving organic liquids and electrical fires. If a fire extinguisher is used it must be given to the laboratory coordinator for recharging. Small fires in test tubes, beakers etc can usually be smothered by covering with a watch glass. In any case, REPORT any accident to your instructor immediately Laboratory Safety Regulations 1. Absolutely NO EATING or DRINKING is permitted in the lab. 2. Waste Disposal Please place the waste materials in the appropriate containers. If you are unsure, make sure you ASK your instructor . For this lab you should expect to have both Inorganic and Organic waste, placed separately in different containers inside the last fumehood. Broken glassware belongs in the BROKEN GLASS bucket Week 1 Melting Point Apparatus Calibration Set at 3 Move to 4 Melting Point Apparatus Calibration Curve 200 Literature value (oC) 0.2 0.0 Naphthalene 79.2 80.2 Benzoic Acid 121.7 122.24 Ice Salicylic Acid 157.1 158.3 180 Literature Value (o C) Observed value (oC) - 5 points Full Credit 160 140 120 100 80 60 40 20 0 20 40 60 80 100 120 140 160 180 200 Observed Temperature (o C) Using the Calibration Curve 200 Literature Value (o C) 180 160 143.2 140 120 100 80 60 40 20 145.7 0 20 40 60 80 100 120 140 160 180 200 Observed Temperature (o C) und cal nf ion e on hat an six ree n Figure 4.11 A ring-flip in chair cyclohexane interconverts axial and equatorial positions. What is axial in cial to determining its properties and biological behavior. the starting structure becomes equatorial in the ring-flipped structure, and what is equatorial in the starting We know from Section 1.5 that ! bonds are cylindrically symmetrical. In structure is axial after ring-flip. other words, the intersection of a plane cutting through a carbon–carbon single-bond orbital looks like a circle. Because of this cylindrical symmetry, rotation is possible around carbon–carbon bonds in open-chain molecules. In ethane, for instance, rotation around the C ! C bond occurs4.11, freely, constantly As shown in Figure a chair cyclohexane can be ring-flipped by keeping changing the spatial relationships between the hydrogens on one carbon and the middle four carbon atoms in place while folding the two end carbons in those on the other (Figure 3.5). opposite directions. In so doing, an axial substituent in one chair form becomes an equatorial substituent in the ring-flipped chair form and vice versa. For H example, axial bromocyclohexane becomes equatorial bromocyclohexane after H ring-flip. Since the energy barrier to chair–chair interconversion is only about C C H H H 45 kJ/mol (10.8 kcal/mol), H the process is rapid at room temperature and we see Rotate H what appears to be a single structure rather than distinct axial and equatorial H H C C isomers. H H Molecular Models: Conformational Isomers H The different arrangements of atoms that result from bond rotation are called conformations, and molecules that have different arrangements are called conformational isomers, or conformers. Unlike constitutional isomers, however, different conformers often can’t be isolated because they interconvert too rapidly. Conformational isomers are represented in two ways, as shown in Figure 3.6. A sawhorse representation views the carbon–carbon bond from an oblique angle and indicates spatial orientation by showing all C ! H bonds. A Newman projection views the carbon–carbon bond directly end-on and represents the two carbon atoms by a circle. Bonds attached to the front carbon are Ring-flip represented by lines to the center of the circle, and bonds attached to the rear carbon are represented by lines to the edge of the circle. Br Back carbon H H H H C H C H H H H Axial bromocyclohexane H H H Front carbon Br Equatorial bromocyclohexane molecule so that the fourth-ranked group ( " H) is oriented toward the rear, away from the observer. Since a curved arrow from 1 ( " OH) to 2 ( " CO2H) to 3 ( " CH3) is clockwise (right turn of the steering wheel), (!)-lactic acid has the R configuration. Applying the same procedure to (#)-lactic acid leads to the opposite assignment. Molecular Models: Stereoisomers (a) Figure 5.8 Assigning configuration (b) to (a) (R)-(!)-lactic acid and (b) (S)-(#)lactic acid. H H3C C HO H CO2H HO2C 2 1 H CO2H HO C CH3 3 R configuration (–)-Lactic acid 2 HO2C H C C CH3 OH 1 OH CH3 3 S configuration (+)-Lactic acid Further examples are provided by naturally occurring (!)-glyceraldehyde and (#)-alanine, which both have the S configuration as shown in Molecular Models: Stereoisomers 5.10 | Chirality at Nitrogen, Phosphorus, an Stereoisomers (Section 4.2) are compounds whose atoms are connected in the same order but with a different spatial arrangement. Among the kinds of stereoisomers we’ve seen are enantiomers, diastereomers, and cis–trans isomers of cycloalkanes. Actually, cis–trans isomers are just a subclass of diastereomers because they are non–mirror-image stereoisomers: Enantiomers (nonsuperimposable mirror-image stereoisomers) CO2H H3C H C HO2C HO OH (R)-Lactic acid Diastereomers (nonsuperimposable non–mirror-image stereoisomers) H H Configurational diastereomers Cis–trans diastereomers (substituents on same H HO OH CH3 CO2H NH2 C C H CH3 2R,3R-2-Amino-3hydroxybutanoic acid H3C H CH3 (S)-Lactic acid CO2H NH2 C C C H 2R,3S-2-Amino-3hydroxybutanoic acid H3C CH3 Molecular Models: Meso Compounds 5.2 | The Reason for Handedness in Molecules: Chirality 145 Figure 5.4 and is achiral, while lactic acid, CH3CH(OH)CO2H, has no plane of 160 CHAPTER 5 | Stereochemistry at Tetrahedral Centers symmetry in any conformation and is chiral. Symmetry plane 146 Not symmetry plane Figure 5.4exists The achiral tartaric acid inpropanoic three acid stereoisomeric forms: two enantiomers a molecule versus the chiral lactic acid meso form. molecule. Propanoic acid has a plane of symmetry that makes one side of the through the C2–C3 bond of mesomolecule a mirror image of the other acid the molecule side. Lactic acid has noHsuch symmetry CHmakes | 3 CHAPTER 5CH Stereochemistrytartaric at Tetrahedral Centers 3 CO2H HO achiral. plane. H C H H C OH C Symmetry plane 3 and 5. Thus, the C6–C5–C4 “substituent” is equivalent to the C2–C3–C4 subCO2H Figure 5.11 A symmetry plane CO2H C reaching the same stituent, and methylcyclohexane is achiral. Another way of CO H HO conclusion is to realize that methylcyclohexane has a symmetry2 plane, which H passes through OH the methyl group and through C1 and C4 of the ring. The situation is different for 2-methylcyclohexanone. 2-MethylcycloCH3CH2CO2H CH3CHCO2H hexanone has no symmetry plane and is chiral because C2 is bonded to four Propanoic acid Lactic acid different groups: a ! CH3 group, an ! H atom, a ! COCH2 ! ring bond (C1), and (achiral) a ! CH2CH2(chiral) ! ring bond (C3). Some physical properties of the three stereoisomers are listed in Ta The (!)- and (")-tartaric acids have identical melting points, solubilit densities, but they differ in the sign of their rotation of plane-polarize Symmetry The most common, although not the only, cause of chirality in an organic The meso isomer, by contrast, is diastereomeric with the (!) and (") f molecule is the presence of a tetrahedralplane carbon atom bonded to four different has no mirror-image relationship to (!)- and (")-tartaric acids, is a d groups—for example, the central carbon atom in lactic acid. Such carbons are compound altogether, and has different physical properties. referred to as chirality centers, although other terms such as stereocenter, asymmetric center, and stereogenic center have also been used. Note that chirality is a H CH CH3 Properties of the Stereoisomers of Tartaric Acid property of the entire molecule, whereas a chirality center is 3the cause Table of 5.3HSome 1 2 * chirality. O Melting Density Solubility at 20 6 2 3 Detecting a chirality center in a complex molecule takes practice because it’s 1 3 ° Stereoisomer point ( C) [!]D (g/cm ) (g/100 mL H2O not always immediately apparent that four different groups are 3bonded to a 4 5 6 168–170 1.7598 139.0 (!) !12 given carbon. The differences don’t necessarily appear right next to the chiral4 5 ity center. For example, 5-bromodecane is a chiral molecule because four differ168–170 1.7598 139.0 (") "12 ent groups are bonded to C5, the chirality center (marked with an asterisk). A Meso 146–148 0 1.6660 125.0 butyl substituent is similar to a pentyl substituent, but it isn’t identical. The difference isn’t apparent until four carbon atoms away from the chirality cenMethylcyclohexane 2-Methylcyclohexanone ter, but there’s still a difference. (achiral) (chiral) Molecular Models: Newman Projection The extra 12 kJ/mol of energy present in the eclipsed conformation of ethane is called torsional strain. Its cause has been the subject of controversy, but the major factor is an interaction between C ! H bonding orbitals on one carbon with antibonding orbitals on the adjacent carbon, which stabilizes the staggered conformation relative to the eclipsed one. Because the total strain of 12 kJ/mol arises from three equal hydrogen–hydrogen eclipsing interactions, we can assign a R R R R H R The value of approximately 4.0 kJ/mol (1.0 kcal/mol) to each single interaction. R H H H R to rotation R barrier that results can be represented on a graph of potential energy H R H H H H versus degree of rotation in which the angle between CH! H bonds and H on front H H H H H R H H back carbons H H as viewed end-on (the dihedral angle) goes full circle from 0 to 360°. Staggered conformations, Staggered and energy maxima Eclipsedoccur at Energy minima occur at staggered Anti eclipsed conformations, asGauche shown in Figure 3.7. Figure 3.7 A graph of po Eclipsed conformations energy versus bond rotatio The staggered conformatio 12 kJ/mol lower in energy t eclipsed conformations. Energy 12 kJ/mol H H H H H H 0° H H H H H H H 60° H H H H H 120° H H H H H H H 180° H H H H H 240° H H H H H H H 300° H H H H 360° H Molecular Models: Newman Projection R R H R H H H H H H R R R H H H R H H H R Staggered Gauche Staggered Anti H H R H H H R H H Eclipsed 4.5 | Conformations of Cyclohexan (a) (b) H H H 3 H H 4 H 2 H 5 H 1 H (c) H 6 H H H 2 H Observer Figure 4.7 The strain-free chair conformation of cyclohexane. All C ! C ! C bond angles are 111.5°, close to the ideal 109.5° tetrahedral angle, and all neighboring C ! H bonds are staggered. The easiest way to visualize chair cyclohexane is to build a molecular model. CH2 1 H H 6 3 H 5 4 H H CH2 H H C O H OH H O H O H O Molecular Models: Fischer Projection Depicting Carbohydrate Stereochemistry: Fischer Projections H CH2OH 25.2 | 1003 D-Glyceraldehyde A tetrahedral carbon atom is represented in a Fischer projection by two crossed lines. The horizontal lines represent bonds coming out of the page, and the vertical lines represent bonds going into the page. Press flat W C Y Z W C X X W Y Fischer projection For example, (R)-glyceraldehyde, the simplest monosaccharide, can be drawn as in Figure 25.1. C CH2OH Bonds out of page CHO = H D-Ribos carbonyl group has the same configuration as (R projections. Z Z Y H HO CH2O Figure 25.2 Some naturally occurring d sugar X CHO [(R)-(+)-glyceraldehyde] C O C OH In contrast with D sugars, L su chirality center, with the bottom projections. Thus, an L sugar is t sponding D sugar and has the op chirality centers. H CHO = H CH2OH OH CH2OH (R)-Glyceraldehyde (Fischer projection) Figure 25.1 A Fischer projection of (R)-glyceraldehyde. Bonds into page HO C O H CH2OH L-Glyceraldehyde [(S)-(–)-glyceraldehyde] CH2OH CH2OH Molecular Models: Figurein 25.2 Some naturally occurring d sugars. The # OH group at the chirality center farthest from the Fischer Projection A tetrahedral carbon atom atom is represented a Fischer projection by two A tetrahedral carbon is represented in a Fischer projection by two | Depicting D-Ribose D-Glucose D-Fructose 25.2 25.2 Carbohydrate Stereochemistry: FischerFischer Projections 1003 1003 | Depicting Carbohydrate Stereochemistry: Projections carbonyl group has the same configuration as (R)-(!)-glyceraldehyde and points toward the right in Fischer crossed lines. lines. The horizontal lines represent bonds coming out of the page, and crossed The horizontal lines represent projections.bonds coming out of the page, and the vertical lines represent bondsbonds goinggoing into the the vertical lines represent intopage. the page. Press flat Press flat W Y C X W Z Y C In contrast with D sugars, L sugars have an S configuration at the lowest chirality center, with the bottom # OH group pointing to the left in Fischer Z projections. Thus, an LZsugar Z is the mirror image (enantiomer) of the correZ W sponding C WX C XD sugar and configuration from the D sugar at all W has X W Xthe opposite centers. Ychirality Y Y X Z Y FischerFischer projection projection Mirror H O can becan O For example, (R)-glyceraldehyde, the simplest monosaccharide, drawn For example, (R)-glyceraldehyde, the simplest monosaccharide, be drawn H C C as in Figure 25.1. 25.1. as in Figure HO CHO CHO H HO C CHO CHO H Bonds Bonds CH2OH out of page out of page CHO CHO L-Glyceraldehyde = H = C HOHC =OH C HCH2OHCH OH 2 CH2OHCH OH HO 2 = H [(S)-(–)-glyceraldehyde] HOH OH Bonds Bonds into page into page CH2OHCH OH 2 (R)-Glyceraldehyde (R)-Glyceraldehyde (Fischer projection) (Fischer projection) HO H H OH O H HO C H OH H HO H H OH HO H H OH CH2OH L-Glucose CH2OH D-Glucose (not naturally occurring) FigureFigure 25.1 A 25.1 Fischer of (R)-glyceraldehyde. A projection Fischer projection of (R)-glyceraldehyde. Note that the D and L notations have no relation to the direction in which a given sugar rotates plane-polarized light. A D sugar can be either dextrorotatory Because a given chiralchiral molecule can becan drawn in many ways, ways, it’s sometimes Because a given molecule be drawn in many it’s sometimes