01_Sigworth_Wint 2014.indd
advertisement

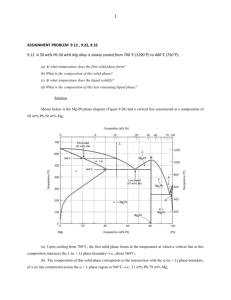
FUNDAMENTALS OF SOLIDIFICATION IN ALUMINUM CASTINGS Geoffrey K. Sigworth GKS Engineering, Dunedin, FL, USA Copyright © 2014 American Foundry Society A version of this paper was previously published in 2013 AFS Transactions. Abstract In this paper the foundation is laid for a fundamental understanding of what happens during solidification. This knowledge is then used to derive practical conclusions about commercial casting alloys. Important phase diagrams are given. These describe, in the form of easily read graphs, what phases form and the relationships between the phases. The nonequilibrium process of solidification is then discussed. The formation, growth and coarsening of dendrites is described and correlations given for secondary dendrite arm spacing (SDAS) as a function of local solidification time. Finally, segregation is considered and models which describe the segregation are reviewed. Example calculations are made for solidification paths in Al-Si casting alloys and suggestions are made to optimize casting alloy compositions. Introduction to Phase Diagrams where p is the number of phases present f is the number of degrees of freedom n is the number of components present The foundry industry is primarily concerned with the solidification process. This is essentially a phase transformation from the hot, liquid state to a colder, solid state. Phase diagrams tell us a great deal about how this transformation occurs. For example, they inform us about: 1. 2. 3. 4. what phases form; at what temperatures the phases form; the relative amounts of each phase the composition of phases, and how solute elements are distributed between the phases and 5. how difficult or easy it will be to place a specific alloying element into aluminum. These are important considerations, which affect the castability of an alloy, as well as the properties in the finished product. One starts by considering pure aluminum. If pure metal is slowly heated, it remains solid until a temperature of 660C (1220F). It then starts to melt, but remains at that temperature until all the metal is molten. Once it is fully liquid, it can be heated to higher temperatures. For a pure metal (or pure water) the number of components (n) is equal to one. When both solid and liquid are present, the number of phases (p) is equal to two. Therefore, the number of degrees of freedom (f) must be equal to one. However, in practice the pressure is fixed by the prevailing atmospheric pressure, so this ‘uses up’ the one degree of freedom. In other words, the melting temperature is not free to vary or change, as long as two phases are present in a pure material. If a pure metal was melted in a high pressure furnace in a laboratory, the melting point would increase. There is a volume increase of about seven percent when aluminum melts. Higher pressures would make it more difficult to melt metal by opposing this volume increase. Therefore, the single degree of freedom means that, as long as the pressure is fixed, the melting point is also fixed. This situation is illustrated by the phase diagram shown in Fig. 1. In this figure the temperature scale is indicated schematically by a thermometer. The situation is exactly analogous to melting ice or placing ice cubes in a glass of water. Ice and liquid water coexist only at a single temperature. This temperature is called the melting point. Only the liquid is above this temperature and below it is the solid. Now the addition of a second element to aluminum, to create a binary (two element) alloy, is considered. According to the phase rule, if there are two components there will be two degrees of freedom. This means that the melting point may change in a two component system. One way to describe the situation is to use the phase rule. The phase rule is defined by the equation: Those who live (or who have lived) in cold climates are familiar with the practice of adding salt to icy sidewalks and driveways to melt ice in the winter. Salt dissolves in water, lowering its melting point. This makes it easier to remove p+f=n+2 Eqn. 1 International Journal of Metalcasting/Volume 8, Issue 1, 2014 7 the ice, as long as the temperature is not far below the freezing point of water. The same thing happens in aluminum. Adding a second element to pure aluminum usually lowers the melting point. This effect can be illustrated by considering the Al-Si system. Silicon lowers the melting point of aluminum, but aluminum also lowers the melting point of silicon. The two curves for the melting of Al and Si meet at a eutectic—at a composition of 12.6 weight percent Si and a temperature of 577C (1071F) (Fig. 2). 1. Hypoeutectic alloys These alloys have a silicon content less than the eutectic composition. Most of the common hypoeutectic alloys have between 5% and 10% silicon. Some examples: C355, 356, 357 and 359. These alloys are designed primarily for high strength applications where good ductility is also required. At the eutectic composition and the eutectic temperature, this phase transformation occurs during solidification: Liquid → Solid (Al) + Solid (Si) Eqn. 2 It should be noted that this transformation occurs at a single, constant temperature. This can be anticipated from the phase rule: f=n+2-p=2+2-3=1 Eqn. 3 Therefore, at constant pressure three phases can coexist in a binary (two element) system only at a single temperature (and of course, at a single composition). An appreciable amount of silicon dissolves in solid aluminum at higher temperatures. The maximum solubility is seen to be 1.65 weight percent at the eutectic temperature. However, only a negligible amount of aluminum dissolves in silicon. Figure 1. Phase diagram for pure aluminum is illustrated. Liquid aluminum and liquid silicon are completely soluble in one another and form a single phase field represented by the ‘L’ in the diagram above the eutectic temperature. Using the standard terminology for this behavior, the two liquids are said to be miscible (or mixable). At temperatures below the melting point of the pure metals, but above the eutectic temperature, there are two phase fields of solid in contact with liquid. These are labeled ‘S + L’. On the left hand side solid aluminum is in contact with liquid. On the right hand side, it is solid silicon in contact with liquid metal. At temperatures below the eutectic temperature, there is another two phase field. It contains two solids: aluminum and silicon. If one looks at phase diagrams for the Al-Si system proposed in the literature over the years, it will be found that there is disagreement as to the exact eutectic composition and to a lesser extent, the eutectic temperature. This is because the formation of the Al-Si eutectic is sensitive to small amounts of impurities, especially P, Na and other alkaline earth elements. The phase diagram given here is based on a study conducted at Alcoa.1 There are three classes of foundry alloys, which are grouped together based upon their silicon content. They are: 8 Figure 2. Phase diagram for Al-Si system is shown. International Journal of Metalcasting/Volume 8, Issue 1, 2014 2. Eutectic alloys These alloys have between 10 and 13% silicon, and consist mainly of Al-Si eutectic in the cast structure. They have a narrow freezing range, excellent fluidity and are easy to cast. They also have good wear resistance and are quite ductile when not alloyed and heat treated to high strength. Eutectic alloys, containing Cu, Mg and sometimes Ni, are used extensively for pistons. 3. Hypereutectic alloys These alloys have between 15 and 20% silicon, so their cast structure is composed of primary silicon particles imbedded in a matrix of Al-Si eutectic. These materials have remarkable wear resistance, and are used where this characteristic is desired: for pistons, liner-less engine blocks and compressor parts. They also have good high temperature strength but are difficult to machine. Diamond tools are necessary. A more detailed look at the Al-Si phase diagram provides a better understanding as to what these characteristics mean in practice. The most important portion of the Al-Si phase diagram for the foundry worker is shown in Fig. 3. As solidification continues the silicon concentration in the liquid portion of the casting increases. Silicon segregates to and accumulates in the liquid phase. This segregation during solidification is best described by a distribution coefficient: Therefore, the phase diagram tells us that, at equilibrium, the silicon content in solid aluminum is 13 percent of that found in the surrounding liquid. The other 87% remains in the liquid, where it accumulates. And as the silicon content increases in the liquid, its melting point decreases. Hence, the composition and temperature of both solid and liquid phases follow the arrows in Fig. 3. This segregation continues until the liquid contains 12.6% Si and cools to the eutectic temperature. At this point, a eutectic mixture of solid Al and Si forms. Another important factor which can be determined from the phase diagram is the depression of the melting point of aluminum. This is defined by the slope of the liquidus curve and by this equation for the Al-Si system: Now, consideration is given to the solidification of a typical hypoeutectic alloy, containing 7% silicon. The molten metal alloy is taken from a furnace held at 760C (1400F). This metal cools in the mold to a temperature of about 615C (1139F). At this temperature the first solid forms: aluminum crystals containing one percent silicon. Eqn. 4 Eqn. 5 For silicon in aluminum, m is equal to 6.6° C per weight percent Si. The last important factor is the solubility of the element in liquid aluminum at typical furnace temperatures. For silicon this maximum concentration is equal to the eutectic composition, 12.6 weight percent Si. These three factors have been tabulated for a number of important or interesting alloying elements.2 The results are shown in Table 1. The elements are listed in order of the value of the distribution coefficient (k). Several important and interesting things may be gleaned from the above tabulated values: • Figure 3. Detail from the Al-Si phase diagram is shown. International Journal of Metalcasting/Volume 8, Issue 1, 2014 Ni, Fe, Si and Cu segregate very strongly during solidification. • Zn and Mg segregate only moderately. • Mn hardly segregates at all. The concentration of Mn in solid aluminum is 94% of the liquid. This is an important factor in the improved performance of die casting alloys, where Mn replaces Fe to prevent die soldering. • The elements below Mn have a value of k greater than one. This means there is a ‘negative’ segregation—the equilibrium concentration in the solid is greater than that in the liquid. As a result, the melting point of aluminum increases. 9 For those who are curious to know the reason for the last statement, a thermodynamic explanation is available.3 Up to this point binary systems have been considered. When another element is added to a binary alloy, there is a ternary (three element) system. It is somewhat more complicated to read ternary phase diagrams, but it is often useful to consult them. Therefore one will be presented to become familiar with the procedure. Figure 4 shows the liquidus surface for aluminum-rich alloys in the ternary Al-Zn-Mg system. This diagram is similar to a topographic map, used for hiking or hunting outdoors. The contours show the temperature (°C) at which solid aluminum begins to form during solidification. A full ternary diagram is an equilateral triangle, but since the interest here is in aluminum-rich alloys, the top portion of the triangle (corresponding to Mg-rich compositions) has been removed. The key to ternary diagrams is understanding how to read the composition coordinates. There are two ternary eutectics in this diagram. They will be used for instruction in the correct procedure. Ternary eutectics are similar to the binary eutectics, discussed previously. However, since there is an additional component (3 instead of 2) there is an additional degree of freedom according to the phase rule. Thus, a ternary eutectic occurs only with this reaction: Liquid → Solid1 + Solid2 + Solid3 Eqn. 6 The formation of three solid phases in the eutectic means the reaction occurs at a fixed temperature and composition. At the top left of the phase diagram there is a ternary eutectic at 447C (837F). If a line is drawn from this point parallel to the sloping left edge, this line intersects the scale at the bottom at about 13% Zn. If a horizontal line is drawn parallel to the bottom edge, it intersects the left edge at about 31% Mg. Thus, this ternary eutectic contains 13% Zn, 31% Mg and (by difference) 56% Al. often possible to find a used copy on the internet. Another useful source for binary and ternary diagrams is the compilation by Phillips.5 For four-component and higher order systems, it becomes exceedingly difficult to represent the phase equilibria. One remarkable contribution worth noting is the study by Phragmen.6 He conducted an extremely detailed study, and presented the results for several quaternary and quinary systems. This paper is not easy reading, but would certainly be worth consulting by anyone wishing to do any serious alloy development in aluminum. Dendritic Solidification Some years ago, the author of this paper read a wonderful book on snowflakes at the Carnegie Library in Pittsburgh. This was a large, coffee table-sized book, with many photographs of individual snow flakes. In his introduction, the author claimed that each snowflake is unique, and no two crystals are alike. This claim may be true, in spite of the incredibly large number of snow flakes that form each winter. The variety of the snow flakes in this book was mind boggling. Something similar happens every time metal solidifies in the mold. The liquid-to-solid transformation involves the formation of many small, individual crystals of solid aluminum. This is a fascinating area, one which has received a great deal of study. Only a brief overview will be given here, touching on the aspects of solidification most important to aluminum foundry industry. For those who wish to study and learn more about solidification fundamentals, two excellent books are available.7,8 Table 1. Alloy Constants for Several Elements Calculated from Phase Diagrams2 There is a second ternary eutectic in the lower right hand side of the diagram, at a temperature of 475C (887F). A similar procedure shows that this eutectic contains approximately 61% Zn, 13%Mg and 26% Al. As an exercise, these results can be determined. There are a number of useful sources of phase diagrams that may be found in a good research library. Important commercial binary diagrams are also easily found on the internet. However, the ternary diagrams are more problematic. The best source found for these is the book by Mondolfo.4 It is unfortunate that this book has gone out of print. It is a fantastic reference for anyone seriously interested in the technology of aluminum and aluminum alloys. Fortunately, it is 10 International Journal of Metalcasting/Volume 8, Issue 1, 2014 The solid aluminum crystals forming during solidification are like snowflakes. The metallurgists first observing these crystals thought they resembled trees. They were therefore called dendrites, after the Greek word for tree (δενδρον or déndron). Dendrites were first observed by polishing metal samples or by etching the polished surface. More recently real time X-ray studies have observed the in situ formation of dendrites in Al-Cu alloys.9 Because the aluminum crystal contains much less copper than the surrounding liquid, they appear lighter in X-ray images. Examples are shown in Fig. 5. The formation of dendritic crystals is a curious phenomenon; so many scientists have studied them. The technical literature in this area is extensive. However, a relatively simple explanation will suffice to understand what is happening. One important clue is that pure metals do not form dendrites. But when silicon or other elements are alloyed to aluminum, dendrites appear. From the Al-Si phase diagram, only 13% of the silicon in the liquid metal remains in the first solid. This means that the silicon atoms ‘pile up’ in front of the growing solid crystals. The situation is shown schematically in Fig. 6. In keeping with the snowflake analogy, the growing aluminum grain by a snow plow is represented. The top half of the figure gives a side view; the bottom a top view. Figure 4. This diagram shows the liquidus surface for aluminum-rich alloys in the ternary Al-ZnMg system (temperatures are given in °C) Figure 5. These are dendrites found in Al-20% Cu Liquid. (Pictures were taken (a) 110, (b) 139 and (c) 360 seconds after the first grains appeared.) (Photo courtesy of Henri Nguyen-Thi, Aix-Marseille University, Marseille, France.) International Journal of Metalcasting/Volume 8, Issue 1, 2014 Figure 6. This shows a schematic view of silicon atoms in front of a moving aluminum crystal. 11 The author of this paper grew up in Jamestown, New York. Winter storms often left 30 to 40 in. (0.75 to 1 m) of snow on the ground. So, a lot of shoveling was experienced. When a shovel is pushed, the snow quickly piles up in front, so one can go no further. When the author was a child the city plowed our sidewalks. A man came by in the night, or early morning, with a horse-drawn plow. This plow had a “V” shaped blade the width of the sidewalk. This blade easily cut through the snow, pushing it to the sides of the walkway. This is shown schematically in Fig. 7. Dendrites act much like this “V” shaped plow. Therefore, growing aluminum crystals adapt a dendritic shape as a response to the alloy composition. Growing solid crystals adapt a planar or a non-planar (dendritic) shape depending on the interaction of two factors. • • result, the dendrites in the final casting are thicker. he spacing between arms also becomes larger.10 It has long been known that the spacing of arms of the dendrite in the casting depends on the solidification time. One of the first detailed studies was published by Alcoa researchers in 1963,11 who related dendrite cell size to the solidification time. Many of the early papers reported cell size in their studies. However, it is now known that a better measure is the secondary dendrite arm spacing (SDAS). The easiest way to measure SDAS is to use the linear intercept method. This is illustrated in Fig. 9 for a modified Al-7% Si alloy. Lines are drawn on a micrograph where well defined The growth rate of the crystal. This is usually defined as the velocity of motion of the solid/liquid interface, in microns per second (R), and is controlled by the thermal gradient in front of the crystal (G). The rate at which the ‘piled up’ solute elements can be removed, by diffusion, from the solidifying front. For a binary system, the relevant factor for non-planar crystal growth has been shown to be:7 Eqn. 6 Figure 7. Schematic view of silicon atoms in front of a growing dendrite tip is shown. The factors m and k are the slope of the liquidus and the distribution coefficient, as determined from the phase diagram and given in Table 1. C is the concentration of the solute. DL is the diffusion coefficient of the solute in liquid. So the shape of the solidifying aluminum depends on the amount and type of solute dissolved in the alloy. The grain size is also influenced by the presence of growthrestricting solutes, like Si and Cu. This may be seen by comparing the grains of different Al-Cu alloys in Fig. 8. These alloys were solidified at an average cooling rate of 1ºC (1.8ºF) per second. All four figures are shown at the same magnification. It is instructive to compare the crystals shown in Fig. 5 and in the bottom half of Fig. 8. In Fig. 5, the crystals are just forming and are ‘new.’ The arms on the branches of the dendrite are fine, much like needle-shaped leaves on a Christmas tree. Also, the dendrites are growing freely into liquid metal. They are still largely unimpeded by neighboring grains. At some point, however, the ‘trunks’ of the dendrites come in contact with neighboring grains. (This time of contact is called dendrite coherency.) After this time any further solidification (and growth of dendrites) can occur only by thickening of the leaves and branches on the dendrite. As a 12 Figure 8. These micrographs show grain morphology in a) Al-5% Cu; b) Al-9.6 %Cu; c) Al-16.2 %Cu and d) Al-25 %Cu. (Micrograph courtesy of David St. John, University of Queensland, Brisbane, Australia.) International Journal of Metalcasting/Volume 8, Issue 1, 2014 dendrite arms can be observed and the average spacing between the centers of adjoining arms is measured. Typically, a number of measurements are made and the results averaged. The SDAS can be used to determine the local solidification time at any point in a casting. The results of many commercial and laboratory measurements on Al-Cu alloys have been reviewed.10 The author of this paper has developed correlations of his own based on measurements of SDAS and the local solidification time measured by thermocouples in the casting. These measurements were in commercial castings and in thermal analysis samples cast from 356 and 319 alloys. The results are plotted in Fig. 10. The curve for Al-Cu alloys in this figure was taken from reference 10. It can be seen that, for a given freezing rate, the copper-containing 319 alloy has a somewhat smaller SDAS than 356 alloy. The correlation for most other foundry alloys would probably lie somewhere between these two curves. Segregation When a metal is liquid, it is homogeneous. That is, the metal properties (especially composition) are the same everywhere. However, there is a different situation in the solid casting. During freezing there is a redistribution of alloying elements and impurities. This redistribution is called segregation. Microsegregation There are two types of segregation. Microsegregation is the variation in composition on a very small scale: between dendrites and dendrite arms. Al-Cu alloy system will be used to illustrate how segregation occurs. This alloy system has been examined in great detail by researchers. It also forms the basis for the high strength family of 2xx casting alloys. The ability to measure SDAS and the correlations, shown in Fig. 10, represents a useful tool. It can help in learning about the thermal history of a sample from an ‘unknown’ casting (e.g., a competitor’s product) or from one’s own castings. It may not always be convenient to place thermocouples in the mold, but the solidification time at various points in the casting from the SDAS can be estimated. The dendritic structure is often visible if you look carefully into pores on the fracture surface of tensile bars. An example is shown in Fig. 11. The rounded ends of the secondary dendrite arms are sticking out from the left hand side of this picture. The SDAS in the sample appears to be between 40 and 50 microns, which corresponds to a local solidification time of about two minutes (for A356 alloy). Figure 9. Measuring SDAS by linear intercept is shown. (Micrograph courtesy of Reza Ghomashchi, University of Adelaide, Adelaide, Australia.) International Journal of Metalcasting/Volume 8, Issue 1, 2014 Figure 10. SDAS versus solidification time in aluminum casting alloys is graphed. Figure 11. This is a SEM micrograph of secondary dendrite arms in a large pore (A356 alloy). 13 Figure 12 shows the complete Al-Cu phase diagram. This is a complicated system. A number of intermetallic compounds form. Some of the phases in this system are: q-Al2Cu, h-AlCu, g-AlCu2 and b-AlCu4. In the aluminum foundry industry there is a concern with the aluminum-rich portion of the figure and the formation of the q (Al2Cu) phase. The relevant portion of the figure is shown in Fig. 13. This diagram is based on data given in a recent study of the system.12 If an alloy containing 4.5% copper is cast and is held near the eutectic temperature, the casting will be in the single phase field corresponding to solid aluminum. This is indicated by the red box in Fig. 13. According to the phase diagram, this alloy should be a single phase—aluminum with copper in solid solution. Looking at a sample from a casting of this alloy, it is found to contain a significant amount of eutectic Al2Cu phase. This eutectic should not be present, according to the phase diagram. So, it is called a non-equilibrium eutectic. Also, if the distribution of copper in a sample of the casting is studied by microprobe analysis in a SEM, the finding is that the copper content in the aluminum phase varies. In the center of dendrite arms, which corresponds to the first solid, the copper content is low. Moving towards the outside of the arms which corresponds to metal freezing later, the copper content increases. An example of this type of measurement is shown in Fig. 14. (This figure was constructed from Gungor’s data.13) Solidification scientists have studied this phenomenon for many years, and have offered models to explain microsegregation. The first and most well-known was given by Scheil.14 This model is based on the following assumptions: • • • There is a local equilibrium at the solid-liquid interface. The compositions of the solid and liquid at this interface as a function of local temperature are described by the binary phase diagram. There is no diffusion of solute in the solid phase. Because diffusion coefficients are much larger in the liquid, it is assumed that the liquid composition is uniform. Figure 12. This is the Al-Cu phase diagram. Figure 13. Detail of the Al-Cu phase diagram is shown. 14 Figure 14. This diagram shows the distribution of Cu in Al-4-5% Cu alloy (local solidification time is 750 seconds). International Journal of Metalcasting/Volume 8, Issue 1, 2014 Based on these assumptions, one can derive this relationship for the concentration of solute in the solidifying metal: Cs = kCo (1– fs)k–1 Eqn. 7 where CS is the instantaneous concentration of solute element in the solid phase, at the solid/liquid interface; k is the distribution coefficient of solute (wt % in solid divided by weight % in the liquid); Co is the original nominal composition of solute in the alloy and fS is the fraction solidified, on a weight basis. Equation 7 has been used to calculate the lower (red) curve in Fig. 15. The Scheil equation predicts the major features of the results, but the actual copper content in the solid is higher and the amount of non-equilibrium eutectic lower than predicted by the Scheil equation. ticularly thorough study was conducted by Sarreal and Abbaschian.16 They measured the amount of non-equilibrium eutectic forming in Al-Cu alloys solidifying at cooling rates over six orders of magnitude. Their experimental results are shown in Fig. 17. The amount of eutectic is plotted as a normalized value. When this value is equal to one, the amount of eutectic is equal to the amount predicted by the Scheil equation. The approximate range of cooling rates found in commercial casting processes is also indicated in this figure. For the slower freezing rates, the eutectic is 70-80 % of the value predicted by the Scheil equation. But as the cooling rate increases, the Scheil equation is a better approximation to the results found. At very fast cooling rates, found in splat cooling or other special processes, a different situation occurs. The amount of the eutectic decreases as the cooling rate increases. Various explanations have been offered for this behavior. (For Brody and Flemings offered a correction to the Scheil equation, which accounted for diffusion of solute into the solid phase during solidification.15 A simple equation described the liquid composition during solidification, but to calculate the final composition of the copper in the dendrites, it was necessary to make numerical calculations. These calculations were made for an Al-4.5% alloy and their results are plotted in Fig. 16, together with the experimental results13 and the Scheil curve. The calculations which include solid diffusion are much closer to the actual microsegregation observed in castings. Other people have studied microsegregation by measuring the amount of non-equilibrium eutectic that forms. A par- Figure 16. The experimental distribution of Cu in Al-4-5% Cu alloy compared with two models is shown. Figure 15. This plot shows the experimental and theoretical distribution of Cu in Al-4-5% Cu alloy. International Journal of Metalcasting/Volume 8, Issue 1, 2014 Figure 17. This plot shows the non-equilibrium eutectic in Al-Cu alloys. 15 example, the discussions by Kurum, et al.12 and Sarreal and Abbaschian16 should be read). It appears that local equilibrium no longer applies at very fast freezing rates. It will be instructive now to make some simple calculations. A slightly different form of the Scheil equation is used: Cl = Co (1– fs)k–1 Eqn. 8 and the equation derived by Brody and Flemings: Eqn. 9 where Cl is the composition of the liquid phase, as a function of fraction solid (fS) The term ak in the denominator of Equation 9 is a correction factor to the Scheil equation, which accounts for loss of solute from the liquid by diffusion into the solid. The model proposed by Brody and Flemings (B-F) can be seen to work extremely well for Al-Cu alloys made by conventional casting methods. Unfortunately, this model involved some approximation which may cause problems in some alloys, as noted by others.16, 18 When the value of ak becomes larger than about 0.5, the B-F model produces erroneous results. Consequently, it cannot describe the segregation of elements having a high diffusivity, such as carbon in iron-carbon alloys. For these cases, it is necessary to apply a ‘fix’ to the BF model.18 One may also consider an alternative formulation.19 However, for the elements commonly found in aluminum castings, and for all but extremely slow freezing rates, the B-F model appears to offer a perfectly satisfactory description of what happens in the castings. Therefore, it will be used subsequently by calculating solidification paths for Al-Si casting alloys. Many models have been offered to account for the microsegregation observed during solidification. The review by Kraft and Chang tabulated more than sixty different mod- The factor alpha is defined by:7, 15 a = 4Dsts/d2 where DS is the diffusion coefficient of solute in solid aluminum (cm2/sec), tS is the local solidification time (seconds) and d is the SDAS (in centimeters, not microns). Once the local solidification time is known, the dendrite arm spacing can be calculated from the correlations given in Fig. 10. Also, the diffusion coefficient needs to be known. Fortunately, a critical review has been made of the diffusion coefficients in solid and liquid aluminum.17 The values for the most important alloying elements in aluminum casting alloys are shown in Fig. 18. Another important element is iron. However, the diffusion coefficient of iron in solid aluminum is two orders of magnitude smaller than the values for the shown elements. Also, the distribution coefficient for iron is very small. This means the factor ak in Equation 9 is essentially zero and can be ignored. It also means the Scheil equation will be valid for iron segregation. For these reasons, the diffusion coefficient of iron is not plotted in Fig. 18. One can now calculate the amount of non-equilibrium eutectic according to the model proposed by Brody and Flemings (B-F). Equation 9 is used to calculate the composition of liquid during solidification, and the fraction of solid at which the eutectic forms (that is, when Cl is equal to 5.65%). Comparing the results of the B-F model to the amount predicted by the Scheil equation produces the red curve shown in Fig. 19. (The conversion from cooling rate to local solidification time has been made for the expected freezing range of the Al-4.5% Cu alloy—100C [212F]). 16 Figure 18. Diffusion coefficients (cm2/sec) for elements dissolved in solid aluminum are plotted. Figure 19. This plot shows the non-equilibrium eutectic in Al-Cu alloys. (Red curve is from Equation 9.) International Journal of Metalcasting/Volume 8, Issue 1, 2014 els proposed in the literature.20 For most aluminum castings, however, the situation is relatively simple. The Brody and Flemings model for segregation and diffusion of solute in the solid describes the microsegregation. Calculation of Solidification Paths Macrosegregation Now that the equations for microsegregation during solidification have been established, it is possible to calculate solidification paths for casting alloys. An example calculation for the commercially important Al-Si based alloys will be discussed. Now consideration is given to the redistribution of solute elements on a larger scale. In net-shaped castings this is caused by the motion of fluid inside the casting and is usually related to feeding. Macrosegregation is most easily seen in copper-containing alloys. An example is shown in Fig. 20. This X-ray shows a section of a cylinder head cast in 319 alloy. Two risers are visible at the top. Both show shrinkage cavities. Underneath the risers are dark bands in the casting, where copper-rich liquid has been ‘sucked into’ the casting. The easiest way to proceed is to assume the distribution coefficients for the binary alloy systems can be used in ternary, alloy systems. There may be some error associated with this, but it usually is not serious. There are sophisticated thermodynamic data bases and programs that can be used to calculate phase relations in multi-component systems but these are not foundry tools. Besides, the goal here is not to exactly simulate what happens, but to provide a simple model that one may use to understand what is occurring. Risers are placed in the mold to ‘feed’ liquid to the solidifying metal, whose volume shrinks by 5-7% during solidification. When the risers are not sufficiently large, they will feed the casting late in their solidification when the remaining liquid is enriched in copper (and other solute elements). The starting point is the aluminum rich corner of the Al-SiFe ternary. This was reviewed in detail in the compilation by Phillips.21 The liquidus surface, shown in Fig. 21, is based on his compilation. It should be noted that a rectilinear diagram is used to present the data, even though this is a ternary system. This is useful, since it is easier to read compositions on this plot. Macrosegregation of this sort can also develop in Al-Si casting alloys, but it is more difficult to see this in X-rays of castings. (The density difference between Al and Si is much smaller than between Al and Cu.) A metallographic examination is usually needed to see macrosegregation in copperfree alloys. The Al-Si-Fe system has also been considered in a recent review.22 Eleven different intermetallic compounds have been identified. Four of them occur in the aluminum-rich portion of the ternary presented in Fig. 21. They are: • FeAl3, which is found in the Al-Fe binary and in alloys low in silicon; • a-FeSiAl, which has a composition close to Fe2SiAl8; • b-FeSiAl, which is usually represented by the composition FeSiAl5 and • d-FeSiAl, which has the composition FeSi2Al3. The compound which is of most concern here is the b phase. This is the intermetallic compound normally observed in commercial castings. Now the solidification paths for an AA309 alloy having 5% Si and various iron contents are calculated. (This alloy also has 1.2% Cu and 0.5% Mg, but this is ignored in the calculation.) To simplify presentation of the results, the liquidus curves are not shown, only the phase boundaries are shown. Also a detailed section from Fig. 21 is taken. The result is shown in Fig. 22 for alloy iron contents of 0.3 and 0.6 percent. Two segregation curves are given for each case. The lower (red) curve is for a solidification time of 10 seconds. The upper (blue curve) is for a longer freezing time of 1,000 seconds. Figure 20. Radiograph shows the macrosegregation in a cylinder head casting. (Radiograph courtesy of J. Fred Major, Rio Tinto Alcan, Kingston, Ontario.) International Journal of Metalcasting/Volume 8, Issue 1, 2014 From this result, it can be seen that 0.3% Fe is a borderline case for this alloy. In rapidly solidified parts of a casting 17 there should be no primary b phase, only a ternary eutectic according to this reaction: Liquid → Al(solid) + Si(solid) + b At slower solidification rates, however, primary b should form. At higher iron contents (e.g., 0.6%) primary b forms before any Al-Si eutectic regardless of the freezing rate. This alloy was studied by Center for Advanced Solidification Technologies (CAST) researchers.23 They found that casting defects were associated with iron contents that produced primary b. However, when they switched to a higher silicon version of the same alloy, the defects went away. The reason for this behavior may be seen by considering Fig. 23. Similar calculations are made for the same two iron contents. In this alloy the higher iron content (0.6%) becomes the borderline case. Therefore, 9% silicon alloy can tolerate twice the iron content of the 5% silicon alloy. In exactly this manner, the CAST researchers calculated solidification paths for numerous silicon contents. In this way they derived a map of ‘safe’ iron contents for their casting (Fig. 24). In conclusion, the results of related research, conducted by Caceres and co-workers,24-25 should be considered. They produced castings in a number of alloy compositions and mea- sured mechanical properties. Some of their results are shown in Fig. 25. The tensile strengths for castings heat treated to the T6 temper are shown on a quality plot (Ultimate Tensile Strength [UTS] versus the log of elongation). The red lines show constant values of quality index (in MPa). The blue arrows indicate the change in alloy composition. For example, iron was added to alloy 1 to obtain alloy 2. The result was a significant loss in casting quality—about 120 Mpa according to the quality index. Silicon was added to alloy 2 to obtain alloy 3. Nearly all of the lost quality was regained by increasing the silicon content from 4.5 to 9 percent. A similar result was found going from alloys 1 → 6 → 7, except in this case copper was added along with the iron. The loss in quality with the combined addition of Fe and Cu was larger—about 200 Mpa—but that loss was regained by increasing the silicon content. By contrast, when copper was added by itself; in the alloys 1 → 4 → 5, there was only a small loss of quality found in alloy 4. It is hoped that this illustration will make clear the importance of microsegregation during segregation and how higher silicon contents may be used to advantage in Al-Si based alloy castings. Figure 21. The ternary phase diagram for the Al-Si-Fe system is shown. Figure 22. Solidification paths for Al-5%Si alloys are plotted. 18 Figure 23. Solidification paths for Al-9%Si alloys are plotted. International Journal of Metalcasting/Volume 8, Issue 1, 2014 Figure 24. Solidification ‘map’ to avoid primary β is shown. Figure 25. The plot shows the properties of heat treated (T6) castings for seven different alloy compositions: 1) 4.5Si-1Cu-0.1Mg-0.2Fe; 2) 4.5Si-1Cu-0.1Mg-0.5Fe0.25Mn; 3) 9Si-1Cu-0.1Mg-0.5Fe-0.25Mn; 4) 4.5Si-4Cu0.1Mg-0.2Fe; 5) 9Si-4Cu-0.1Mg-0.2Fe;6) 4.5Si-4Cu-0.1Mg0.5Fe-0.25Mn and 7) 9Si-4Cu-0.1Mg-0.5Fe-0.25Mn In this paper, the segregation of hydrogen was not considered. The diffusivity of H is 100,000 times faster than the dissolved elements: Fe, Cu, and Si. Consequently, the segregation mechanism for hydrogen is very different. In this case, one will have an equilibrium distribution of hydrogen as given by the phase diagram.26 REFERENCES 1. Wright, E.H., Willey, L.A., “Aluminum Binary Equilibrium Diagrams,” Alcoa Research Laboratories Technical Paper No. 15, Alcoa, Pittsburgh, PA (1960). 2. Backerud, L., Johnsson, M., Sigworth, G., “Method for the Optimization of the Grain Refinement of Aluminum Alloys,” U.S. Patent 6,073,677 (June 13, 2000). 3. Sigworth, G.K., “The Grain Refining of Aluminum and Phase Relationships in the Al-Ti-B System,” Met. Trans., vol. 15A, pp. 277-282 (1984). 4. Mondolfo, L.F., Aluminum Alloys: Structure and Properties, Butterworths (1976). International Journal of Metalcasting/Volume 8, Issue 1, 2014 5. Phillips, H.W.L., Annotated Equilibrium Diagrams of Some Aluminum Alloy Systems, Monograph and Report Series, no. 25, Institute of Metals, London (1959). 6. Phragmen, G., “On the Phases Occurring in the Alloys of Aluminum with Copper, Magnesium, Manganese, Iron and Silicon,” J. Inst, Metals, , vol. 77, pp. 489552, London (1950). 7. Flemings, M.C., Solidification Processing, McGrawHill, New York (1974). 8. Kurz, W., Fischer, D.J., Fundamentals of Solidification, Trans Tech Publications, Switzerland (1986). 9. Mathiesen, R.H., Arnberg, L., Nguyen-Thi, H., Billia, B., “In-Situ Video X-ray Microscopy as a Tool in Solidification Science,” J. Metals, vol. 64(1), pp. 76-82 (2012). 10. Flemings, M.C., Kattamis, T., Bardes, B.P., “Dendrite Arm Spacing in Aluminum Alloys,” AFS Transactions, vol. 99, pp. 501-506 (1991). 11. Spear, R.E., Gardner, G.R., “Dendrite Cell Size,” AFS Transactions, vol. 71, pp. 209-215 (1963). 12. Kurum, E.C., Dong, H.B., Hunt, J.D., “Microsegregation in Al-Cu Alloys,” Metal. Mater. Trans. A, vol. 36A, pp. 3103-3110. 13. M.N. Gungor, M.N., “A Statistically Significant Experimental Technique for Investigating Microsegregation in Cast Alloys,” Metall. Trans., vol. 20A, pp. 2529-2533 (1989). 14. E. Scheil, E., Z. Metallkunde, vol. 34, p. 70 (1942). 15. Brody, H.D., Flemings, M.C., “Solute Redistribution in Dendritic Solidification,” Trans. AIME, vol. 236, pp. 615-624 (1966). 16. Sarreal, J.A., Abbaschian, G.J., “The Effect of Solidification Rate on Microsegregation,” Metallurgical Transactions A, vol. 17A, pp. 2063-2073 (1986). 17. Du, Y., Chang, Y.A., co-workers, “Diffusion Coefficients of Some Solutes in FCC and Liquid Al: Critical Evaluation and Correlation,” Mater. Sci. and Engineering, vol. A 363, pp. 140-151 (2003). 18. Cline, T.W., Kurz, W., “Solute Redistribution during Solidification with Rapid Solid State Diffusion,” Metallurgical Transactions A, vol. 12A, pp. 965-971 (1981). 19. Voller, V.R., Beckermann, C.R., “A Unified Model of Coarsening and Microsegregation,” Metall. Mater. Metall. Trans. A, vol. 30A, pp. 2183-2189 (1999). 20. Kraft, T., Chang, Y.A., “Predicting Microstructure and Microsegregation in Multicomponent Alloys,” Journal of Metals, vol. 49(12), pp. 20-28 (1997). 21. Phillips, H.W.L., Annotated Equilibrium Diagrams of Some Aluminum Alloy Systems, Monograph and Report Series, No. 25, Institute of Metals, London (1959). 22. Krendelsberger, N., Weitzer, F., Schuster, J.C., “On the Reaction Scheme and the Liquidus Surface in the Al-Si-Fe System,” Metall. Mater. Trans. A, vol. 38A, pp. 1681-1691 (2007). 19 23. Taylor, J.A., Schaffer, G.B. St. John, D.H., “The Role of Iron in the Formation of Porosity in Al-Si-Cu–Based Casting Alloys: Part II. A Phase-Diagram Approach,” Metall. Mater. Trans. A, vol. 30A, pp. 1651-1655 (1999). 24. Caceres, C.H., Svensson, I.L., Taylor, J.A., “StrengthDuctility Behaviour of Al-Si-Cu-Mg Casting Alloys in the T6 Temper,” Internat. J. Cast Metals Res., vol. 15, pp. 531-543 (2003). 20 25. Caceres, C.H., Taylor, J.A., “Enhanced Ductility in Al-Si-Cu-Mg Casting Alloys with High Silicon Content,” Shape Casting: The John Campbell Symposium, pp. 245-254, TMS, Warrendale, PA (2005). 26. Poirier, D.R., Sung, D.K., “Thermodynamics of Hydrogen in Al-Si Alloys,” Metall. Mater. Trans. A., vol. 33A, pp. 3874-3876 (2002). International Journal of Metalcasting/Volume 8, Issue 1, 2014