FEBS 29314
FEBS Letters 579 (2005) 1821–1827
Minireview
From protein networks to biological systems
Peter Uetza,1, Russell L. Finley Jr.b,*
b
a
Research Center Karlsruhe, Institute of Genetics, P.O. Box 3640, D-76021 Karlsruhe, Germany
Center for Molecular Medicine & Genetics, Wayne State University School of Medicine, 540 East Canfield Avenue, Detroit, MI 48201, USA
Accepted 31 January 2005
Available online 7 February 2005
Edited by Robert Russell and Giulio Superti-Furga
Abstract A system-level understanding of any biological process
requires a map of the relationships among the various molecules
involved. Technologies to detect and predict protein interactions
have begun to produce very large maps of protein interactions,
some including most of an organismÕs proteins. These maps can
be used to study how proteins work together to form molecular
machines and regulatory pathways. They also provide a framework for constructing predictive models of how information and
energy flow through biological networks. In many respects,
protein interaction maps are an entrée into systems biology.
Ó 2005 Federation of European Biochemical Societies. Published
by Elsevier B.V. All rights reserved.
Keywords: Interaction map; Protein network; Pathway; System
1. Introduction
Systems Biology should give us the tools to model how
genes, gene products, and other molecules work together
to mediate biological processes. Use of such tools, and indeed their very development, requires, for each biological
process, lists of the molecules involved and their interconnections. The genes and proteins predicted from genome sequences have provided a long list of parts (genes and gene
products), and new technologies have begun to define lists
of other molecules not directly encoded by the genome that
are present in cells and tissues at particular times. New computational and experimental technologies have begun to produce enormous datasets representing interactions between
the parts. For the moment, most of the interaction data
comes from technologies to detect physical or functional
interactions between genes and proteins. Here, we will review
some of the sources of these data and consider how the
quantity and quality of the available interaction data may
impact systems-level studies.
2. Protein–protein interactions
The prominent role that protein–protein interactions play in
most biological processes, combined with the fact that we
*
Corresponding author. Fax: +1 313 577 5218.
E-mail addresses: peter.uetz@itg.fzk.de (P. Uetz), rfinley@wayne.edu
(R.L. Finley Jr.).
1
Fax: +49 7247 82 3354.
know so little about the functions of most proteins, has inspired efforts to map interactions on a proteome-wide scale
(e.g., for all of the proteins encoded by a genome) [1]. To date,
most of the interactions that have been detected experimentally
have relied on one of two technologies, the yeast two-hybrid
system [2] and mass spectrometry (MS) identification of proteins that co-affinity purify (co-AP) with a bait protein [3].
The two technologies detect complementary types of interactions. Co-AP/MS identifies the constituents of multi-protein
complexes but does not reveal the individual binary contacts
that make up each complex. Without data on the constituent
binary contacts, the possible paths of energy or information
flow through the complex and its relationship to other cellular
components may not be apparent. Yeast two-hybrid data, on
the other hand, identifies likely binary interactions that may
suggest possible paths through a pathway or complex, but cannot reveal the constituents of multiprotein complexes. Thus,
both types of data will be important for understanding protein
and pathway function, and ideally both approaches would be
performed on a proteome-wide scale.
Yeast two-hybrid screens aiming to cover entire proteomes,
or at least very large numbers of proteins, have detected thousands of interactions for a few eukaryotic model organisms
(Table 1), bacteria and phage [4,5] and viruses [6]. By contrast,
proteome-wide co-AP/MS screens have been conducted only in
yeast (Table 1), where most of the proteome could be easily
affinity tagged through the use of homologous recombination.
Co-AP/MS data for other organisms is only just beginning to
emerge through the use of high throughput cloning [7] and the
expression of large sets of tagged proteins in tissue culture cells
(e.g. [8,9]). Thus, it is likely that we will begin to see protein
complex data for humans and other metazoans in similar
quantities as the yeast studies have produced.
3. How complete are current protein interaction datasets?
Despite the volumes of interaction data produced, several
independent analyses have shown that the data from the large
two-hybrid and co-AP/MS screens is far from complete.
Various authors have estimated that the roughly 6000 yeast
proteins are connected by 12 000–40 000 interactions [10–12],
yet the high throughput screens have detected only a small
fraction of those numbers (Table 1). Another clue comes from
the lack of overlap among the different datasets for a particular
proteome. For example, in Table 1, the overlap among the
large two-hybrid screens for yeast was only 6 interactions
0014-5793/$30.00 Ó 2005 Federation of European Biochemical Societies. Published by Elsevier B.V. All rights reserved.
doi:10.1016/j.febslet.2005.02.001
1822
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
Table 1
Large protein interaction screens for eukaryotes
Interactionsa
Proteins
Reference
967
4549
420
9421
3878
1004
3278
271
1665
1578
[63]
[13]
[64,65]
[66]
[67]
Yeast two-hybrid
Yeast two-hybrid
20 405
1814
7048
488
[49]
[14]
Yeast two-hybrid
4027
1926
[68]
Organism (genes)
Method
Yeast (6000)
Yeast two-hybrid
Yeast two-hybrid
Yeast two-hybrid
Co-AP/MS
Co-AP/MS
Drosophila (14 000)
Worm (20 000)
a
For two-hybrid screens, the approximate number of unique binary interactions is shown. For co-AP/MS screens, the approximate number of binary
interactions that would result if each bait protein contacted every protein that co-purified with it (the ‘‘hub and spoke’’ model) is shown. Data can be
retrieved from one of the databases cited [42–44].
[13] while the overlap between the screens for Drosophila was a
measly 28, or less than 2% of the smallest data set [14]. The coAP/MS data is not much different. For example, when results
from the two large-scale studies are compared, the number of
interactions common to both datasets is less than 9% of the total in both datasets [15]. Data from the high throughput
screens also fails to overlap significantly with published ‘‘low
throughput’’ studies, which are generally considered to be less
subject to false positives and false negatives. Such analyses
have led some authors to estimate false negative rates as high
as 85% in large yeast two-hybrid screens and 50% in co-AP/
MS screens [16,17]. These results suggest that many more
interactions could be detected by more exhaustive application
of these technologies. In addition, there is a need for improved
or new high throughput technologies to identify interactions
that may be difficult to detect with two-hybrid or co-AP/MS,
such as interactions involving membrane proteins.
4. Physical and functional interactions
Comparison of the data from yeast two-hybrid and co-AP/
MS provides an example of an important distinction between
two types of interaction data: physical interactions (A touches
B) and functional interactions (A functions with B in some biological process). A functional relationship may or may not correspond to a direct physical interaction. Thus, physical and
functional interactions are two distinct though partially overlapping types of interactions and the distinction is likely to
be important for the development of systems-level models of
protein networks and pathways. Yeast two-hybrid is an experimental approach to detect physical interactions. Co-AP/MS
detects group of proteins in stable complexes, implying that
they function together. Another example of a functional but
not necessarily physical interaction is a genetic interaction, in
which the combination of alleles of two different genes has specific phenotypic consequences. This is often taken to suggest
that the two genes function in the same or parallel pathways
affecting a particular biological process. Thus, a genetic interaction is a measured functional interaction that may or may
not correspond to a physical interaction, but that could be usefully represented as a connection between the two genes or
gene products. Ongoing large-scale screens in yeast have
mapped thousands of genetic interactions [18]. Combination
of genetic and physical interaction data is a powerful approach
to mapping pathways [18,19].
5. Predicted and experimentally measured interactions
The increasing use of computational approaches to predict
protein interactions has led to additional large datasets (e.g.
[20]) and to another distinction between two types of interaction data: experimentally measured and predicted. Predicted
interactions can also be classified as either physical or functional. Gene expression profiles have been used, for example,
to infer functional interactions among gene products, based
on the assumption that proteins that function together in the
same pathway or complex should be frequently expressed together; which is supported by data for stable protein complexes [21,22]. Similarly, genes whose coexpression profiles
are conserved through evolution are often functionally related
[23,24], as are genes that are co-conserved from species to species [25–27]. The functional links between proteins in each of
these cases may be direct or very general; they may suggest
roles in the same pathways, or in distinct cellular systems that
are concomitant but that have very few direct molecular connections. Genetic interactions have also been predicted based
on physical interactions, gene expression, protein localization,
and other experimental data [28,29]. Numerous methods for
predicting physical protein–protein interactions have also been
developed [22,30–35]. One very powerful approach takes
advantage of the large number of experimentally measured
interactions available for organisms like yeast and Drosophila
to predict interactions in other organisms [36]. Simply put,
the approach predicts that two proteins will interact if their
orthologs were shown to interact; such conserved interactions
have been referred to as interologs [37–39]. This approach has
been used, for example, to predict 70 000 interactions involving proteins encoded by a third of the human genes [40]. Several other studies have begun to effectively integrate genomic
and proteomic data to make increasingly accurate interaction
predictions [33,41]. The further development and use of in silico approaches to map interactions seems particularly important in light of the shortcomings of high throughout
experimental detection systems.
6. Protein interaction maps
The wealth of data from high throughput screening and
other studies has begun to be consolidated into centralized,
standardized databases. Three of the largest public database
repositories for interaction data are BIND, DIP, and IntAct
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
1823
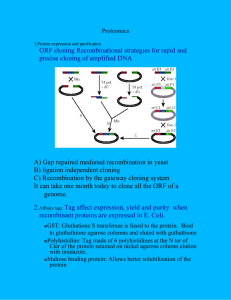
Fig. 1. Interaction maps today and tomorrow. (A) Typical representation of a protein–protein interaction map. (B) Proteins are usually shown as
nodes (e.g., circles and boxes) and interactions as edges (lines) connecting them. This organizes information so that attributes of both proteins and
interactions are easily accessible, for example, by hypertext links, but fails to capture structural information. However, additional information could
be made easily accessible through pop-up menus by clicking on the edges (like here) or the nodes. (C) and (D) Ideally, a protein interaction map
visualization tool would allow the structures of proteins and interaction interfaces to be expanded and browsed, and would also provide access to
more global interaction attributes (e.g., conditions under which a certain set of interaction can be found, experimental conditions, dissociation
constants, expression levels, etc.). Based on interactions published by Measday et al. [69], Uetz et al. [63], and reviewed in [70].
[42–44]. These allow researchers online access to browse and
download data in a standardized format [45]. Sets of interaction data can be viewed as graphs or maps in which each
gene/protein is a node and each interaction is a line connecting
two nodes (Fig. 1). The importance of this view has led to use
of the term ‘‘interaction map’’ to refer generically to interaction datasets. The map view provides not only an intuitive
interface for biologists to explore the data, but also a formal
mathematical framework for computational biologists to explore the properties of interaction networks. However, before
interaction maps can be used to represent biological networks,
their limitations must be considered.
In addition to the problem of false negatives discussed previously, most interaction maps and particularly those from
high throughput screens have false positives. Estimates of false
positive rates vary widely, in part because of the difficulty in
definitively demonstrating that any particular interaction does
not have a biological function. Because the false positive rates
may be substantial, the maps from high throughput studies
might be usefully regarded as the results from a first pass filter,
which reduces the possible search space for functionally important interactions. Thus, the question becomes how to identify
the more likely true positives. Several studies have confirmed
the general principle that interactions detected in multiple
screens and by different techniques or in different species are
more likely to be true positives than those only found once
or twice [17]. Due to the high rates of false negatives in high
throughput screens, however, there has been very little overlap
between different datasets, thus, limiting the opportunities for
such experimental cross-validation. Alternatively, a variety of
confidence scoring systems have been developed that calculate
the likelihood of an interaction being a true positive, based on
various parameters, including attributes of the proteins and the
specific assays, whether the interaction was detected by other
technologies or screens, and network topology [20,46–50].
However, thus far most of these scoring systems are specific
to particular datasets or methodologies, and no universal system has yet been effective.
Another limitation of most protein interaction maps is
that each node generally represents some generic version of a
1824
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
Galactose
Gal2
Gal7
Gal7
Galactose
Gal1-P
Gal1
Gal1
Glucose-6-P
Gal5
Gal5
Other
processes
Translation
Translation
mRNA
mRNA
Gal3
Glu1-P
Gal10
Cytoplasm
Gal3
mRNA
RNA-processing
+ export factors
Gal3
?Gal80
P?
Gal4
RNA-processing
factors
Gal80 Gal3
Gal80 Gal3
Gal80 Gal3
Gal80 Gal3
Gal4
Gal4
Gal4
Gal4
Gal4
Gal4
DNA
Gal1
Gal10
Gal10
Gal7
Gal7
X
P
P
P
P
Gal80
Srb10 Srb11
Gal11
RNA pol II holoenzyme
General TAFs Gal11
Nucleus
Nucleus
X
Chromatin
Chromatin
TRENDS in Cell Biology
YJL036W
Gal1
YFL036W
YDL113C
YLR294C
YMR228W
YCR045C
YMR057C
YGL174W
Gal3
Gal80
YIR005W
YBR020W
YBL025W
YML043C
YDR532C
YDR142C
YMR297W
YMR214W
YLR055C YBR081C
YML051W
YLR191W
YNR003C
YGL153W
YGL145W
YBL014C
YJR083C
YLR315W
YHL042W
YJL084C YOR110W
YPL248C
YOL051W
YJL034W
YGR252W
YCL020W
YOR298CA
YGL013C
YOL148C
YBL010C
YPL019C
YKR022C
YEL062W
YLR447C
YLR150W
YFL002WA
YJR122W
YOR254C
YPL082C
YKR062W
YDR277C
YNL257C
YGR258C
YLR015W
YDR392W
YJR072C
YLR424W
YLR292C
YER148W
YIL143C
YCR042C
YDR422C
YGL208W
YML042W
YCR088W
YDL194W
YER171W
YGR274C
YBR198C
YKL028W
YMR181C
YGR268C
YJL137C
YHR128W
YLR273C
YML114C
YGL180W
YPR072W
YLR243W YOR250C
YCR093W
YNL122C
YPR049C
YKL061W
YGR167W
YDR469W
YGR253C
YLR005W
YDL135C
YDR129C
YGR032W
YGL126W
YLR371W
YLR249W
YJL089W
YDR477W
YML109W
YDR329C
YBR140C
YAL003W
YPR165W
YDR309C
YDR388W
YNL138W
YDL138W
YFR042W
YBL101W-A
YPR159W
YGR123C
YBR171W
YKL020C
YJL019W
YBR118W
YNL039W
YGR047C
YLR342W
YDL089W
YKL129C
YER027C
YOR062C
YGR160W
YPR185W
YGR178C
YOR047C
YLR258W
YBR196C YAL028W
YCR035C
YNR025C
YDR167W
YER040W
YPR040W
YNR032W
YGL134W
YOR181W
YDL165W
YHR016C
YBR170C
YPR180W
YPR080W
YER177W
YNL126W
YLR049C
YJL005W
YDR482C
YHR161C
YPL222W
YGL028C
YDL070W
YGR048W
YPL161C
YBL105C
YDL179W
YNL098C
YBR274W
YPL211W
YNL086W
YBL007C
YIL045W
YLR233C
YJL211C
YER165W
YNL229C
YGR162W
YDR390C
YMR311C
YDR028C
YLR277C
YGL073W
YGL024W
YHR100C
YOR315W
YNL116W
YER133W
YLR117C
YAL047C
YDR130C YPR107C
YDL101C
YHR135C
YER079WYER054C
YCR011C
YMR267W
YJL013C YHR185C
YHR014WYPR008W
YPL218W
YIL004CYGR188C
YNL154C YOR026W
YKR030W
YGL025C
YDR195W
YLR268W YGR172CYNL146W
YER031C
YDR416W
YBR188CYBR205W
YPL022W
YGL049C
YIL072W
YLR078C
YGL212W
YPL070W
YNL188W
YLR229C
YLR212C
YGL154C
YPL192C
YOL123W
YPR070W
YLR263W
YDR174W
YKR026C
YCL054W
YGL122C
YNR023W
YER032W
YDR335W
YPR048W
YML103C
YFR002W
YNL112W
YAL030W
YGL201C
YAL034WA
YGR003W
YLL026W
YNL090W
YGL254W YNL271C
YBR200W
YJL157C
YER114C
YOR180C
YLR284C
YGL192W
YCL055W
YBR057C
YDL226C
YPL242C
YOR231W
YAL041W
YJL085W
YIL118W
YOR373W
YGR068C YBL102W
YGL019W
YIL035C
YLR147C
YGL015C
YER029C
YNR029C
YBR109C
YLL021W
YKL042W
YOR326W
YNL225C
YPL124W
YOL016C
YML057W
YER149C
YJL064W
YOR061W
YKL112W
YJL065C
YHR140W
YER124C
YOR039W
YLR362W
YHR198C
YDL012C
YIL047C
YDR151C
YLR319C
YKL161C
YPL140C
YOR115C
YMR129W
YHR030C
YHL046C
YEL009C
YPR025C
YFR047C
YLL046C
YOR303W
YOR151C
YJR109C
YMR095C
YHR032W
YPL089C
YLR313C
YDR026C
YLL019C
YFR034C
YGR009C
YMR183C
YHR215W
YGR152C
YKL178C
YDR264C YJL095W
YOR212W
YJR086W
YKL008C
YLR363C
YHR005C
YHR038W
YJR056C
YOR355W
YBL085W
YNL243W
YDR356W
YDR103W
YML064C YDL097C
YOL133W
YJL141C
YPL204W YOR327C
YDL229W
YKR055W
YMR032W
YHR061C
YMR055C
YIL159W
YHR172W
YOR122C
YGR153W
YER143W
YIL001W
YCL059C
YLR127C
YPL240C
YJL200C
YLL024C
YKL023W
YLR291C
YLR166C
YMR071C
YOR257W
YMR109W
YMR039C
YNL335W
YPL031C
YNL309W
YOR276W
YOR101W YMR186W YMR102C
YDR382W
YLR305CYNL201C
YOL139C
YHR200W
YJR094C
YGR233C
YOL082W
YGL044C
YLR071C
YGL240W
YKL072W
YFR036W
YDR169C
YOL004W YHR197W
YLR216C
YBL075C
YHR178W
YLR423C
YPR115W
YDR207C
YOR178C
YIL061C
YNL330C YBL084C
YMR061W
YHR166C
YPR086W
YMR255W
YMR180C
YLR310C
YDR214W
YJR032W
YML015C
YGL116W
YOR027W
YDR122W
YKL022C
YKL103C
YFL056C
YDR146C
YGR014W
YDL008W
YDL020C YBR006W
YKL117W
YDR206W
YOL001W
YKL095W
YAR018C
YDL215C
YBR045C
YOR329C YKL204W
YDL106C
YJL030W
YGL112C
YGL086W
YIL007C
YJL047C
YKL193C
YJL151C
YOR292C
YNL020C
YHR102W
YDL127W
YOR353C
YIL050W
YOR275C
YMR009W
YGL215W
YBR155W
YNL218W
YMR139W
YJR093C
YKR002W
YMR053C
YOL130W
YAR014C
YLR045C
YLL016W
YGR120C
YOR346W
YGL229C
YIR037W
YMR019W
YBR050C
YBR244W
YBR190W
YGR080W
YNL289W
YLR432W
YOR105W
YFL039C
YMR316CB
YDR311W YPL219W
YDL065C
YDR463W
YLR337C
YOL144W
YIL163C
YNL298W
YHL007C
YHR071W
YDR228C
YJL042W
YKL081W
YOR127W
YJL178C
YGL206C
YCR005C
YDR389W
YDR099W
YIL123WYML038C
YIR006C
YHR206W
YBL043W
YGR241C
YOL101C
YMR092C
YLR324W
YDR017C YDL178W
YLL050C
YER059W
YJL162C
YOL012C
YDR216W
YIL038C
YDR376W
YJL185C
YBR108W
YCR009C
YML110C
YNL042W
YHR158C
YER068W
YIR024C
YLR102C
YCL046W
YGL115W
YLR190W YGR246C
YDR448W
YOR138C
YOR262W
YMR232W
YDR244W
YJL114W
YDL214C
YBR123C
YOR331C
YGR238C
YPR105C
YHR060W
YDR176W
YDL161W
YDL078C YMR291W
YOR164C
YMR270C
Gal4
YNL084C
YHR160C
YPR190C
YDR383C
YHR079C
Gal11
YOL105C
YDR498C
YJL025W
YDR009W
YNL151C
YOR348C
YIL172C
YNL027W
YCR087W
YDR110W
YHR145C
YIL113W
YLR433C
YDL159W
YKR068C
YDR472W
YNL333W
YBR254C
YMR096W
YDL108W
YFL059W
YNL251C
YGL130W
YDR460W
YMR322C
YPL228W
YGR046W
YJL057C
YDR246W
YML077W
YGL150C YDL002C
YFL060C
YER161C
YDL246C
YKL190W
YLR384C YDL140C
YMR211W
YNL091W
YER132C
YNL164C YNL288W
YBR236C
YGL043W
YER092W
YBL016W
YPL038W
YPR187W
YJL128C
YKR034W
YOR208W YKL075C
YPL229W
YER118C
YMR153W
YOR284W
YDR253C
YGR040W
YOR341W
YOL149W
YDR323C
YJL110C
YBL050W
YCL032W
YNL047C
YJL058C YJR131W
YOL129W
YJR077C
YLR269C
YGR155W
YJR159W
YDL056W
YPR041W
YER179W
YER047C
YHL006C
YOR034C
YMR201C
YNL312W
YLL036C
YLR046C YNL021W
YBR080C YAL040C
YGL104C YJR050W YML001W
YDL154W
YER075C
YIL144W
YER100W
YOR361C
YBR270C
YMR309C
YPR103W
YHR152W
YER112W
YNL118C
YGL175C
YDR412W
YNL244C YER111C
YBR133C
YJL092W
YDR078C
YCR086W
Y
EL1
YPL260W
YLR113W
YOR089C
YGL158W
YEL015W
YPL049C
YGL178W YNL103W
YMR213W
YMR094WYIL150C
YMR087W
M
YER012W
YHR039C
YMR012W
YLL053C
YDL203C
YOR264W
YJR133W
YNL135C YBR079C
YOR157C YML097C
YDR128W
YDR357C
YPR019W
YML032C
YDR106W
YFL003C
YJL187C
YOL106W
YPL238C
YLL039C
YGR058W YPL059W
YPL075W YMR146C
YER136W
YGR136W
YOR167C
YPR182W
YOR229W
YHR129C
YDR480W
YOL020W
YLR208W
YIR017C
YAR007C YJL173C
YDR464W
YDR378C
YPR101W
YCR067C
YDR054C
YOL059W
YML031W
YER052C
YOR372C
YPL174C YIL046W
YHL031C
YOR230W
YKL012W
YLR275W
YGL170C
YNL233W
YBR138C
YER095W
YPL256C
YPL151CYFL005W
YBR023C
YNL147W
YJL184W
YDR369C
YBR017C
YIL105C
YJR132W
YGR179C
YDR326C
YDL195W
YPR011C
YBR202W
YDL088C
YBL079W
YMR068W
YMR117C
YML046W YDR439W
YJR022W
YPL213W
YBL061C YKL145W
YGR119C
YPR163C YDR429C
YPR017C
YDR070C
YER007CA
YOR362C
YLR026C
YPL085W
YFL017C
YGL238W
YMR224C
YLR264W
YKL203C
YJR060W
YGR140W
YHR084W
YBR094W
YDR032C
YBL026W
YPL018W
YDR201W
YMR233W
YEL032W
YJL124C
YHR083W
YIL109C
YEL037C
YMR168C
YNL092W
YOR160W
YDL043C
YKL074C
YDL064W
YDR076W
YNL250W
YGL172WYKL068W
YPR119W
YOR259C
YDR510W
YOR191W
YLR082C
YPR181C
YPR010CYML049C
YNR022C
YKL049C
YOL034W
YOR159C
YDL160C
YNL307C
YKL089W
YOL006C
YKR010C
YGL048C
YER018C
YPL268W
YCR004C YDR189W
YMR199W
YHR170W
YLR167W
YGL197W
YER162C
YER102W YLR175W
YAL038W
YLR103C
YGL092W
YER110C
YHR165C
YLR274W YLR116W
YOR020C
YIR009W
YDR318W
YER146W
YOR375C
YBR052C
YNL272C
YHR089C
YLR293C
YDR004W
YLR347C
YNL216W
YNL286W
YAL032C
YLR335W
YDL071C
YNL004W YDR432W
YHR098C
YMR308C
YKL015W
YDR386W
YBR211C
YOR098C
Y
SM3
YDL147W
YKR037C
YKL002W
YNL273W
YKL155C
YDL111C
YDL132W
YPR082C
YNL155W
YEL068C
YBR135W
YBR160W
YDR148C
YBR055C
YJR052W
YBR172C
YDR171W
YDR183W
YNL023C YDR192C
L
YDR394W
YKL173W
YPL105C
YFL018WA
YDR139C
YLR419W
YDL110C
YBR275C
YHR035W
YGR158C
YCL066W
YDL030W
YDR315C
YDL013W
YMR047C YDL207W
YGR218W
YNL036W
YBL023C
YPR145W
YIL115C
YFR046C
YER045C
YLR259C
YLR456W
YLR323C
YCR002C
YOR078W
YOL021C
YLR392C
YER009W
YDR179C
YKR060W
YOR206W
YDR132C
YDL155W YJR090C
YOR117W
YER116C
YLR100W
YDR104C YNR052C YAL021C YMR043W YIR025W
YNR069C
YGR108W
YCR073C
YDR328C
YBR114W
YJR074W
YJR117W
YIL125W
YIL063C
YOR319W
YGR232W
YJR091C
YGR269W
YKL148C
YGL249W
YLL041C
YJL194W
YLR345W YJL203W
YGL096W
YGL221C
YLR210W
YLR442CYLR429W
YFL029C
YJL041W
YDR485C
YER107C
YLR128W
YFL009W
YMR025W
YHR057C
YNL189W
YOR057W
YKL130C
YLR352W
YPR054W
YKL101W
YLR224W YML088W
YOL058W
YLR399C
YDR227W
YKR101W
YLR097C
YJL061W
YMR125W
YNL031C
YGL097W
YDL029W
YPL133C
YBR221C
YBR234C
YOR038C
YML065W
YLR006C
YCR039C
YJL218W
YOR185C
YDL216C
YJR065C
YLL049WYNR068C
YBR009C
YBR103W
YIL065C
YBL008W
YDR052C
YCL067C
YLR314C
YPR062W
YMR048W
YJL048C
YDR507C
YBR010W
YMR314W
YDL017W
YER065C
YCR084C
YOR204W
YPL178W
YPR020WYDR503C
YDR313C
YIL112W
YCR097W
YDR002W
YML010W
YNL030W
YGR063C
YGR116W
YKL205W
YFL061W
YDL235C
YLR079W
YLR321C
YNR031C
YPL214C
YKR048C
YMR001C
YMR284W
YER025W
YNR048W
YDR395W
YDL236W
YKL039W YGR099W
YIL147C
YDL042C
YLR368W
YML028W YBR252W
YCL024W YPR120CYPL153C
YPL111W
YKL144C
YMR138W
YOR156C
YCR050C
YFR028C
YMR106C
YJL076W
YHL009C
YFR057W
YDL225W
YJL088W
YHR107C
YMR226C
YDR255C
YJR076C
YLR303W
YLR109W
YLR176C
YCR022C
YDR225W
YDR217C
YDR085C
YDL117W
YNL078W
YOR006C
YKL135C
YDR218C
YBR284W
YPL241C
YIL126W
YIL132C
YOR070C
YOL070C
YOL127WYLR406C
YBR112C
YDL075W
YDR043C
YHR075C
YDR084C
YGL198W
YGL161C
YIL011W
YFL038C
YGR129W
YNL263C
YHR105W
YDR211W
YMR269W
YOR106W
YOR260W
YGR083C
YJR007W
YOR370C
YFR052W
YKL196C
YIL013C
YDR273W
YDR061W
YOR036W
YPL246C
YJR034W
YMR236W
YDR468C
YGR117C
YNL064C
YLR452C
YMR080C YFR033C
YAL005C YGR072W
YNL236W
YDR172W YER105C
YHR077C
YLR182W
YML095C
YGL095C
YPL237W
YNL199C
YMR197C
YBR143C
YOR359W
YOL018C
YBR134W
YOR128C
YGR057C
YEL023C
YPR102C
YGR085C
YLR322W
YLR376C
YFR037C
YMR091C
YDL011C
YMR035W
YPR078C
YHR106W
YPR093C
YDL051W
YGL035C
YMR150C
YHL019C
YPL259C
YHR141C
YGR024C
YHR216W
YPL110C
YPL025C
YDR087C
YNR050C
YKL090W
YLR075W
YIL082W
YOL108C
YMR317W
YIR012W
YPL128C
YDR123C
YNL279W
YKL017C
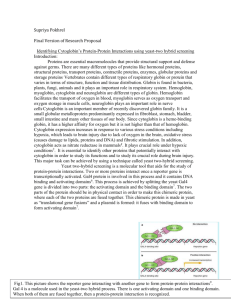
Fig. 2. Integrating protein networks and other biological information. (Bottom) The map of interactions among 1200 yeast proteins represents only
the tip of the information iceberg. Highlighted in blue are proteins involved in galactose regulation, which in the map are found in a topological
cluster (see text). A cluster of cytoskeletal proteins (highlighted in red) is also visible. (Top) Integrating the protein interaction network with the
metabolic network (shown as green compounds and grey enzymes), the gene regulatory network (shown as pink genes and blue transcription factors),
and the signaling network indicated by the Cyclin-dependent kinase Srb10 and its cyclin Srb11 (red). Yellow boxes indicate functional modules that
involve additional protein interactions within complexes and with other proteins (not shown). Modified after Tucker et al. [12] and Rohde et al. [71].
cellular protein, without regard to the various splice variants
or post-translationally modified forms that may exist. Isoforms
could interact differently from the form that was actually used
in the assay, which in many cases is unclear. This is particularly true for assays that use only one or a small number of
the possible alternative transcripts from each gene. Thus, many
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
so-called ‘‘protein’’ interactions maps are actually gene or locus interaction maps, which tell us only that one or more of
the proteins encoded by one locus is capable of interacting
with one or more of the proteins encoded by another locus.
Nevertheless, such maps have proven to be useful as starting
points for additional studies, particularly if the caveats are
borne in mind.
7. Using the interaction maps for systems biology
A complete systems-level understanding of any biological
process may require more input data than current technologies
can offer. Intuitively, we imagine that we could model a system
best only after knowing all of the molecules involved, their
concentrations, how they fit together, the effect of each individual part on its neighbors, and dynamic parameters such as how
concentrations, interactions, and mechanics change over time.
But this seems unrealistic given the fact that the high throughput technologies for measuring many of these parameters are
still on the drawing board, if they exist at all. Do we really need
to know all of the details of a process to be able to develop a
useful systems-wide understanding or to have a predictive
model? Analysis of protein interaction maps has suggested that
even sparse data can be used to derive initial, rudimentary
models of biological networks.
Topological analyses, for example, initially of metabolic
pathways and subsequently of protein interaction maps, began
to reveal some common properties of biological networks
[51,52]. These initial studies suggested the exciting possibility
that cellular networks may be organized according to some
general principles that could be understood without a detailed
knowledge of all the constituent proteins and interactions
[53,54]. Moreover, analysis of network topology can provide
insights into protein and pathway function. For example, protein networks contain highly connected hub proteins, which
have been shown to correlate with evolutionarily conserved
proteins, and in yeast with proteins encoded by essential genes
[51,55,56]. Thus, a proteinÕs relative position in a network has
implications for its function and importance. Analysis of
topology also reveals clusters of highly interconnected proteins
that correlate with conserved functional modules (Fig. 2), such
as protein complexes or signaling pathways [57–59]. Thus,
even the currently available noisy protein interaction maps
can be used to explore the hierarchical organization of biological networks and to reveal interconnected modules that control specific biological processes. As these modules are
defined and further elaborated, understanding them and their
higher order organization will increasingly rely on advances
in information technology.
1825
a map of interactions [60–62]. These programs allow exploration and ad hoc analyses of interaction data but they rarely
incorporate all of the useful available information about the
molecules and interactions they represent (e.g., see Figs. 1
and 2). Moreover, they usually fail to capture the essential dynamic properties of biological networks. Animated cartoons,
on the other hand, can provide at least a qualitative representation of the dynamics of a process (see, for example, Fig. 3).
However, such oversimplification does not capture the details
of the system or facilitate quantitative modeling. In a way,
visualization of molecular networks is where word processing
was in the early 1980s.
To help us model biological processes, and to visualize and
manipulate those models, we need programs to generate more
dynamic and realistic representations of biological events and
structures. We need what might be called ‘‘interactive CellTV’’ to visualize and manipulate models of cellular events
and behavior. Importantly, iCell-TV must operate across several scales of time and space to allow biologists to navigate all
available relevant information. Such a system, for example,
might allow users to explore the changes in the molecular
structure resulting from a post-translational modification,
zoom out to witness the subsequent changes in network and
pathway dynamics, and then change time scales to observe
organelle movement or cell behavior. The number and complexity of the experiments that must be done to test hypotheses
8. Perspectives: iCell-TV
How can biologists access and integrate the deluge of proteomics data to help them understand biology? While this
information should help drive the generation of hypotheses
and hypothesis-testing research, we may be generating data
faster than we are learning how to use it. Tools for accessing
and analyzing molecular interaction data have just begun to
emerge over the past few years. Several ‘‘visualization’’ tools
and graphing programs, for example, allow users to construct
Fig. 3. Visualizing the dynamics of protein interaction and signaling
networks. The pheromone signaling pathway in yeast is a highly
dynamic process that involves numerous protein interactions, phosphorylation events, and small-molecule interactions involving ATP
and GTP. Typical textbook (i.e., static) representations like this do not
reflect the dynamics of this process. A more realistic representation is
available through animation, as shown at http://www.bioveo.com/
MAPK/MAPk.htm. Simplified from an animation by Tom Dallman,
by permission of the author.
1826
coming from network analyses are likely to be costly and inefficient. The next generation of biological information management systems must, therefore, allow us to do biology truly in
silico. For this to be possible, they must enable the development and manipulation of quantitative models, which are often initially based on a qualitative understanding. However,
it is often the case that about the time we understand a system
well enough to be able to model it, it becomes too hard to
understand in a qualitative sense. A system for navigating
qualitative information based on quantitative data would give
users the ability not only to understand the complexity of biological processes but also to manipulate those processes, to
construct new models, and to test new hypotheses. Zoom in,
change a Kd or a Vmax, then zoom out and watch what happens
to the system. This would be systems biology for the rest of us,
and would open biological inquiry to a vast resource of
creativity.
Acknowledgments: We thank Tom Dallman for permission to use his
pheromone pathway animation.
References
[1] Phizicky, E., Bastiaens, P.I., Zhu, H., Snyder, M. and Fields, S.
(2003) Protein analysis on a proteomic scale. Nature 422, 208–
215.
[2] Fields, S. and Sternglanz, R. (1994) The two-hybrid system: an
assay for protein–protein interactions. Trends Genet. 10, 286–292.
[3] Pandey, A. and Mann, M. (2000) Proteomics to study genes and
genomes. Nature 405, 837–846.
[4] Rain, J.C., et al. (2001) The protein–protein interaction map of
Helicobacter pylori. Nature 409, 211–215.
[5] Bartel, P.L., Roecklein, J.A., SenGupta, D. and Fields, S. (1996)
A protein linkage map of Escherichia coli bacteriophage T7. Nat.
Genet. 12, 72–77.
[6] Uetz, P., Rajagopala, S.V., Dong, Y.A. and Haas, J. (2004) From
ORFeomes to protein interaction maps in viruses. Genome Res.
14, 2029–2033.
[7] Marsischky, G. and LaBaer, J. (2004) Many paths to many
clones: a comparative look at high-throughput cloning methods.
Genome Res. 14, 2020–2028.
[8] Bouwmeester, T., et al. (2004) A physical and functional map of
the human TNF-alpha/NF-kappa B signal transduction pathway.
Nat. Cell Biol. 6, 97–105.
[9] Brajenovic, M., Joberty, G., Kuster, B., Bouwmeester, T. and
Drewes, G. (2004) Comprehensive proteomic analysis of human
Par protein complexes reveals an interconnected protein network.
J. Biol. Chem. 279, 12804–12811.
[10] Walhout, A.J., Boulton, S.J. and Vidal, M. (2000) Yeast twohybrid systems and protein interaction mapping projects for yeast
and worm. Yeast 17, 88–94.
[11] Grigoriev, A. (2003) On the number of protein–protein interactions in the yeast proteome. Nucleic Acids Res. 31, 4157–4161.
[12] Tucker, C.L., Gera, J.F. and Uetz, P. (2001) Towards an
understanding of complex protein networks. Trends Cell Biol.
11, 102–106.
[13] Ito, T., Chiba, T., Ozawa, R., Yoshida, M., Hattori, M. and
Sakaki, Y. (2001) A comprehensive two-hybrid analysis to explore
the yeast protein interactome. Proc. Natl. Acad. Sci. USA 98,
4569–4574.
[14] Stanyon, C.A., et al. (2004) A Drosophila protein-interaction map
centered on cell-cycle regulators. Genome Biol. 5, R96.
[15] Cornell, M., Paton, N.W. and Oliver, S.G. (2004) A critical and
integrated view of the yeast interactome. Comp. Funct. Genom. 5,
382–402.
[16] Edwards, A.M., Kus, B., Jansen, R., Greenbaum, D., Greenblatt,
J. and Gerstein, M. (2002) Bridging structural biology and
genomics: assessing protein interaction data with known complexes. Trends Genet. 18, 529–536.
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
[17] von Mering, C., Krause, R., Snel, B., Cornell, M., Oliver, S.G.,
Fields, S. and Bork, P. (2002) Comparative assessment of largescale data sets of protein–protein interactions. Nature 417, 399–
403.
[18] Tong, A.H., et al. (2004) Global mapping of the yeast genetic
interaction network. Science 303, 808–813.
[19] Tewari, M., et al. (2004) Systematic interactome mapping and
genetic perturbation analysis of a C. elegans TGF-beta signaling
network. Mol. Cell 13, 469–482.
[20] von Mering, C., et al. (2005) STRING: known and predicted
protein–protein associations, integrated and transferred across
organisms. Nucleic Acids Res. 33 (Database Issue), D433–D437.
[21] Jansen, R., Lan, N., Qian, J. and Gerstein, M. (2002) Integration
of genomic datasets to predict protein complexes in yeast. J.
Struct. Funct. Genom. 2, 71–81.
[22] Jansen, R., et al. (2003) A Bayesian networks approach for
predicting protein–protein interactions from genomic data. Science 302, 449–453.
[23] Bergmann, S., Ihmels, J. and Barkai, N. (2004) Similarities and
differences in genome-wide expression data of six organisms.
PLoS Biol. 2, E9.
[24] Stuart, J.M., Segal, E., Koller, D. and Kim, S.K. (2003) A genecoexpression network for global discovery of conserved genetic
modules. Science 302, 249–255.
[25] Pellegrini, M., Marcotte, E.M., Thompson, M.J., Eisenberg, D.
and Yeates, T.O. (1999) Assigning protein functions by comparative genome analysis: protein phylogenetic profiles. Proc. Natl.
Acad. Sci. USA 96, 4285–4288.
[26] Date, S.V. and Marcotte, E.M. (2003) Discovery of uncharacterized cellular systems by genome-wide analysis of functional
linkages. Nat. Biotechnol. 21, 1055–1062.
[27] Bowers, P.M., Cokus, S.J., Eisenberg, D. and Yeates, T.O. (2004)
Use of logic relationships to decipher protein network organization. Science 306, 2246–2249.
[28] Wong, S.L., et al. (2004) Combining biological networks to
predict genetic interactions. Proc. Natl. Acad. Sci. USA 101,
15682–15687.
[29] Marcotte, E.M., Pellegrini, M., Thompson, M.J., Yeates, T.O.
and Eisenberg, D. (1999) A combined algorithm for genome-wide
prediction of protein function. Nature 402, 83–86.
[30] Aloy, P., et al. (2004) Structure-based assembly of protein
complexes in yeast. Science 303, 2026–2029.
[31] Aloy, P. and Russell, R.B. (2002) Interrogating protein interaction networks through structural biology. Proc. Natl. Acad. Sci.
USA 99, 5896–5901.
[32] Lu, L., Arakaki, A.K., Lu, H. and Skolnick, J. (2003) Multimeric
threading-based prediction of protein–protein interactions on a
genomic scale: application to the Saccharomyces cerevisiae proteome. Genome Res. 13, 1146–1154.
[33] Zhang, L.V., Wong, S.L., King, O.D. and Roth, F.P. (2004)
Predicting co-complexed protein pairs using genomic and proteomic data integration. BMC Bioinform. 5, 38.
[34] Enright, A.J., Iliopoulos, I., Kyrpides, N.C. and Ouzounis, C.A.
(1999) Protein interaction maps for complete genomes based on
gene fusion events. Nature 402, 86–90.
[35] Reiss, D.J. and Schwikowski, B. (2004) Predicting protein–
peptide interactions via a network-based motif sampler. Bioinformatics 20 (Suppl 1), I274–I282.
[36] Sharon, R., et al. (2005) Conserved patterns of protein interaction in multiple species. Proc. Natl. Acad. Sci. USA 102, 1974–
1979.
[37] Walhout, A.J., Sordella, R., Lu, X., Hartley, J.L., Temple, G.F.,
Brasch, M.A., Thierry-Mieg, N. and Vidal, M. (2000) Protein
interaction mapping in C. elegans using proteins involved in
vulval development. Science 287, 116–122.
[38] Matthews, L.R., Vaglio, P., Reboul, J., Ge, H., Davis, B.P.,
Garrels, J., Vincent, S. and Vidal, M. (2001) Identification of
potential interaction networks using sequence-based searches for
conserved protein–protein interactions or ‘‘interologs’’. Genome
Res. 11, 2120–2126.
[39] Yu, H., et al. (2004) Annotation transfer between genomes:
protein–protein interologs and protein–DNA regulogs. Genome
Res. 14, 1107–1118.
[40] Lehner, B. and Fraser, A.G. (2004) A first-draft human proteininteraction map. Genome Biol. 5, R63.
P. Uetz, R.L. Finley Jr. / FEBS Letters 579 (2005) 1821–1827
[41] Lee, I., Date, S.V., Adai, A.T. and Marcotte, E.M. (2004) A
probabilistic functional network of yeast genes. Science 306,
1555–1558.
[42] Alfarano, C., et al. (2005) The biomolecular interaction network
database and related tools 2005 update. Nucleic Acids Res. 33
(Database Issue), D418–D424.
[43] Salwinski, L., Miller, C.S., Smith, A.J., Pettit, F.K., Bowie, J.U.
and Eisenberg, D. (2004) The database of interacting proteins:
2004 update. Nucleic Acids Res. 32 (Database issue), D449–D451.
[44] Hermjakob, H., et al. (2004) IntAct: an open source molecular
interaction database. Nucleic Acids Res. 32 (Database issue),
D452–D455.
[45] Hermjakob, H., et al. (2004) The HUPO PSIÕs molecular
interaction format – a community standard for the representation
of protein interaction data. Nat. Biotechnol. 22, 177–183.
[46] Saito, R., Suzuki, H. and Hayashizaki, Y. (2003) Construction of
reliable protein–protein interaction networks with a new interaction generality measure. Bioinformatics 19, 756–763.
[47] Goldberg, D.S. and Roth, F.P. (2003) Assessing experimentally
derived interactions in a small world. Proc. Natl. Acad. Sci. USA
100, 4372–4376.
[48] Deane, C.M., Salwinski, L., Xenarios, I. and Eisenberg, D. (2002)
Protein interactions: two methods for assessment of the reliability
of high throughput observations. Mol. Cell Proteomics 1, 349–
356.
[49] Giot, L., et al. (2003) A protein interaction map of Drosophila
melanogaster. Science 302, 1727–1736.
[50] Bader, J.S., Chaudhuri, A., Rothberg, J.M. and Chant, J. (2004)
Gaining confidence in high-throughput protein interaction networks. Nat. Biotechnol. 22, 78–85.
[51] Jeong, H., Mason, S.P., Barabasi, A.L. and Oltvai, Z.N. (2001)
Lethality and centrality in protein networks. Nature 411, 41–42.
[52] Jeong, H., Tombor, B., Albert, R., Oltvai, Z.N. and Barabasi,
A.L. (2000) The large-scale organization of metabolic networks.
Nature 407, 651–654.
[53] Strogatz, S.H. (2001) Exploring complex networks. Nature 410,
268–276.
[54] Barabasi, A.L. and Oltvai, Z.N. (2004) Network biology: understanding the cellÕs functional organization. Nat. Rev. Genet. 5,
101–113.
[55] Said, M.R., Begley, T.J., Oppenheim, A.V., Lauffenburger, D.A.
and Samson, L.D. (2004) Global network analysis of phenotypic
effects: protein networks and toxicity modulation in Saccharomyces cerevisiae. Proc. Natl. Acad. Sci. USA 101, 18006–18011.
1827
[56] Han, J.D., et al. (2004) Evidence for dynamically organized
modularity in the yeast protein–protein interaction network.
Nature 430, 88–93.
[57] Poyatos, J.F. and Hurst, L.D. (2004) How biologically relevant
are interaction-based modules in protein networks. Genome Biol.
5, R93.
[58] Spirin, V. and Mirny, L.A. (2003) Protein complexes and
functional modules in molecular networks. Proc. Natl. Acad.
Sci. USA 100, 12123–12128.
[59] von Mering, C., Zdobnov, E.M., Tsoka, S., Ciccarelli, F.D.,
Pereira-Leal, J.B., Ouzounis, C.A. and Bork, P. (2003) Genome
evolution reveals biochemical networks and functional modules.
Proc. Natl. Acad. Sci. USA 100, 15428–15433.
[60] Shannon, P., et al. (2003) Cytoscape: a software environment for
integrated models of biomolecular interaction networks. Genome
Res. 13, 2498–2504.
[61] Breitkreutz, B.J., Stark, C. and Tyers, M. (2003) Osprey: a
network visualization system. Genome Biol. 4, R22.
[62] Mrowka, R. (2001) A Java applet for visualizing protein–protein
interaction. Bioinformatics 17, 669–671.
[63] Uetz, P., et al. (2000) A comprehensive analysis of protein–protein
interactions in Saccharomyces cerevisiae. Nature 403, 623–627.
[64] Drees, B.L., et al. (2001) A protein interaction map for cell
polarity development. J. Cell Biol. 154, 549–571.
[65] Fromont-Racine, M., et al. (2000) Genome-wide protein interaction screens reveal functional networks involving Sm-like
proteins. Yeast 17, 95–110.
[66] Gavin, A.C., et al. (2002) Functional organization of the yeast
proteome by systematic analysis of protein complexes. Nature
415, 141–147.
[67] Ho, Y., et al. (2002) Systematic identification of protein complexes in Saccharomyces cerevisiae by mass spectrometry. Nature
415, 180–183.
[68] Li, S., et al. (2004) A map of the interactome network of the
metazoan C. elegans. Science 303, 540–543.
[69] Measday, V., Moore, L., Retnakaran, R., Lee, J., Donoviel, M.,
Neiman, A.M. and Andrews, B. (1997) A family of cyclin-like
proteins that interact with the Pho85 cyclin-dependent kinase.
Mol. Cell. Biol. 17, 1212–1223.
[70] Carroll, A.S. and OÕShea, E.K. (2002) Pho85 and signaling
environmental conditions. Trends Biochem. Sci. 27, 87–93.
[71] Rohde, J.R., Trinh, J. and Sadowski, I. (2000) Multiple signals
regulate GAL transcription in yeast. Mol. Cell. Biol. 20, 3880–
3886.