View/Open - Scholarworks @ CSU San Marcos
advertisement

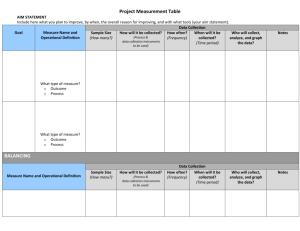
Executive Summary Evaluation of Capital Investments at Cliniqa Corporation: A Systematic Ranking and Evaluation Procedure Cliniqa Corporation Vy Nguyen 14 May 2014 Professional Science Masters in Biotechnology, California State University, San Marcos Management teams are frequently charged with making informed decisions for valid investments and cost-saving ideas for laboratory instruments. Cliniqa Corporation is a research and manufacturing company that produces quality controls and calibrators for a number of in-vitro diagnostic instruments. The semester-in-residence project resulted in an organized investment evaluation procedure for management to consider all the relevant factors before committing to an instrument procurement decision. Purchasing new equipment can extend the capabilities of the company into additional product lines and strategically target desired customers. It was important to establish an expert system to harvest the knowledge of senior management and to potentially improve on the current evaluation techniques. The research methodology included an evaluation of the current instruments in operation to set a baseline on the previous evaluations and to establish a quantitative ranking system. The sensitivity of the factors required the continuous fine-tuning of the analysis tool. The result of the project was a five-step evaluation tool with proof-of-concept pilot testing for the recommendation of instrument investment. The process began with setting constraints based on the needs and requirements that the company can commit to for asset acquisition. Secondly, data was collected for the criteria of: manufacturer, type of analyzer, first year introduced to market, square footage, routine maintenance, assay menu capability, test throughput, ability of being an open channel instrument and continuous runs, requirement for mainline water hookups, and incremental training required for a routine operator. Then, quantitative ranking values (1-10) were applied and compared against the constraints. Third was to calculate the total cost of ownership for qualified instruments. The fourth step was to consider the qualitative factors of: instrument usability, upholding company reputation, customer satisfaction, self-imposed analyzers for strategic contractual purposes, and product line expansion. The last step of the tool was to analyze all the factors involved for the recommendation of new instruments. The pilot testing proposed the purchase of a refurbished Roche cobas e411, Abbott Architect i1000sr, or Siemens Advia Centaur CP to meet the company’s objectives of maintaining quality products and boosting overall profitability. Evaluation of Capital Investments at Cliniqa Corporation: A Systematic Ranking and Evaluation Procedure Vy Nguyen Cliniqa Corporation 14 May 2014 Professional Science Masters, Biotechnology California State University, San Marcos Committee Julie Jameson Mike Hoskins Alan Styles Casey Zace Overview Introduction Background Importance of Project Research methodology Analysis of current situation through pilot testing Discussion Future Directions • • Retrieved from http://www.cliniqa.com/Custom-Manufacturing/OEM-Solutions.aspx What do we do? What are clinical analyzers? • Why do we need them? • How do we get them? Retrieved from http://www.dealermarketing.com/bizdev/business-development/2725how-to-make-a-dealership-stand-out An expert system captures the knowledge from senior management Retrieved from http://www.igcseict.info/theory/7_2/expert/ Research methodology A. Development • Survey key operational personnel, review of technical and support documents for instruments • Identify constraints • Identify relevant/unique criteria for evaluation • Identify all current instruments used by organization • Developed a data collection and analysis approach • Identify all current non-point-of-care (POC) instruments used by organization Research methodology B. Refinement • Collect data for current non-POC equipment • Rank instruments on evaluation criteria • Evaluate current instrument decisions using developed evaluation tool • Identify differences, reconcile or consider reasons for differences • Identify qualitative decision criteria not included in investment evaluation tool • Amend/update investment evaluation tool Research methodology C. Pilot testing • Identify non-POC instruments not currently used by organization (potential investments) • Collect data for potential instruments • Rank instrument on amended evaluation criteria • Identify equipment that exceeds evaluation thresholds • Financial calculations The developed investment evaluation tool consists of five steps Step 1: Determine constraints Step 2: Collect data for potential instrument(s) and quantitatively rank each instrument Step 3: Perform financial calculations Step 4: Consider the qualitative factors Step 5: Propose instrument(s) Step 1 Results Step 2a Results Step 2b Step 2b Results Step 3 Results Step 4 Qualitative factors Step 5 Instrument recommendations http://reacting.ma/detail_siemens_ADVIA%20CENTAUR%20CP.html http://www.sunflowerlab.net/pathology_laboratory_in_mumbai.php http://www.roche-diagnostics.co.in/Products/Pages/COBAS4000platform.aspx Discussion/Future Directions Utility of tool: expert system, methodical tool Determine alternatives for the company: purchase, lease, external testing, maintain capital (pay off credit) Not all variables apply Improve on current analysis techniques Opportunity costs, budget allocation, ROI based on cash flow determine risks/liabilities Limitations of tool: subjectivity and sensitivity of factors Future Directions: requires additional fine-tuning/updating of analysis tool References Byers, S. S., Groth, J. C., Richards, R. M., & Wiley, M. K. (1997). Capital investment analysis for managers. Management Decision, 35(3), 250-257. Retrieved from http://ezproxy.csusm.edu/login?url=http://search.proquest.com/docview/212065320?accountid=10363 CAP Today: Survey of Instruments (2007, June). In American Proficiency Institute. Retrieved March 2, 2014, from http://www.cap.org/apps/docs/cap_today/surveys/0607ImmunoSvy.pdf CAP Today: Survey of Instruments (2003, July). In American Proficiency Institute. Retrieved February 21, 2014, from http://www.cap.org/apps/docs/cap_today/surveys/0703_3252_ChemSvy.PDF Database (2013). In American Proficiency Institute. Retrieved March 2, 2014, from https://www.apipt.com/pds.aspx Drury, C., &Tayles, M. (1997).The misapplication of capital investment appraisal techniques. Management Decision, 35(2), 86-93. Retrieved from http://ezproxy.csusm.edu/login?url=http://search.proquest.com/docview/212053493?accountid=10363 Li,J., K., Min, J, Otake, T., Voorhis, T.V., (2008). Inventory and investment in setup and quality operations under Return On Investment maximization, European Journal of Operational Research, 185(2), 593-605. Retrieved from http://www.sciencedirect.com/science/article/pii/S0377221707000823 Jiambalvo, James. Managerial Accounting. 4th ed. N.p.: John Wiley & Sons, 2010. Print. O'Kane, M. J. (2014, March). Point of Care Testing - Current and Emerging Quality Perspectives. Point of Care, 13(1). Shao, L. P., & Shao, A. T. (1996).Risk analysis and capital budgeting techniques of U.S. multinational enterprises. Managerial Finance, 22(1), 41-57. Retrieved from http://ezproxy.csusm.edu/login?url=http://search.proquest.com/docview/212669275?accountid=10363 Sokoll, L. J., & Chan, D. W. (1999, June 15). Clinical Analyzers. Immunoassays. Analytical Chemistry, 71(1), 356R362R. Retrieved March 1, 2014